S-1: General form of registration statement for all companies including face-amount certificate companies
Published on August 14, 2020
Table of Contents
As filed with the Securities and Exchange Commission on August 14, 2020
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
Benitec Biopharma Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 2834 | 84-462-0206 | ||
| (State or other jurisdiction of incorporation or organization) |
(Primary Standard Industrial Classification Code Number) |
(I.R.S. Employer Identification Number) |
3940 Trust Way Hayward, California 94545 (510) 780-0819
(Address, including zip code, and telephone number, including area code, of registrants principal executive offices)
Dr. Jerel Banks
Chief Executive Officer
3940 Trust Way
Hayward, California 94545
(510) 780-0819
(Name, address, including zip code, and telephone number, including area code, of agent for service)
Copies to:
| Ben D. Orlanski, Esq. Matthew S. OLoughlin, Esq. Proskauer Rose LLP 2029 Century Park East, Suite 2400 Los Angeles, CA 90067-3010 (310) 557-2900 (310) 557-2193Facsimile |
Steven M. Skolnick, Esq. Lowenstein Sandler LLP 1251 Avenue of the Americas New York, NY 10020 (212) 262-6700 (212) 262-7402-Facsimile |
Approximate date of commencement of proposed sale to public: As soon as practicable after this Registration Statement is declared effective.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the following box. ☒
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See the definitions of large accelerated filer, accelerated filer, smaller reporting company and emerging growth company in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☐ | |||
| Non-accelerated filer | ☒ | Smaller reporting company | ☒ | |||
| Emerging growth company | ☒ | |||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☒
CALCULATION OF REGISTRATION FEE
|
|
||||
| Title of Each Class of Securities to be Registered |
Proposed Maximum Aggregate Offering Price(1)(2) |
Amount of Registration Fee |
||
| Common stock, $0.0001 par value per share(3) |
$ | $ | ||
| Pre-funded warrants to purchase shares of common stock and common stock issuable upon exercise thereof(3) |
$ | $ | ||
| Total |
$20,000,000 | $2,596 | ||
|
|
||||
|
|
||||
| 1) | Estimated solely for purposes of determining the registration fee in accordance with Rule 457(o) under the Securities Act of 1933, as amended. Pursuant to Rule 416(a) under the Securities Act of 1933, as amended, this registration statement shall also cover an indeterminate number of shares that may be issued and resold resulting from stock splits, stock dividends or similar transactions. |
| 2) | Includes additional shares of common stock that the underwriter has the option to purchase. |
| 3) | The proposed maximum aggregate offering price of the common stock proposed to be sold in the offering will be reduced on a dollar-for-dollar basis based on the aggregate offering price of the pre-funded warrants offered and sold in the offering (plus the aggregate exercise price of the common stock issuable upon exercise of the pre-funded warrants), and as such the proposed aggregate maximum offering price of the common stock and pre-funded warrants (including the common stock issuable upon exercise of the pre-funded warrants), if any, is $20,000,000 (including the underwriters option to purchase additional shares of common stock). |
The Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until the Registration Statement shall become effective on such date as the Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
Table of Contents
The information in this preliminary prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities and it is not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
SUBJECT TO COMPLETION, DATED AUGUST 14, 2020
Preliminary Prospectus

Shares of Common Stock,
Pre-funded Warrants to Purchase Shares of Common Stock
Shares of Common Stock underlying the Pre-funded Warrants
We are offering shares of our common stock. Each share of common stock is being sold at a public offering price of $ per share.
We are also offering to certain purchasers whose purchase of shares of common stock in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if such purchasers so choose, pre-funded warrants in lieu of shares of common stock that would otherwise result in any such purchasers beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock. Each pre-funded warrant will be exercisable for one share of our common stock and will be exercisable at any time after its original issuance until exercised in full. The purchase price of each pre-funded warrant will be equal to the price at which a share of common stock are sold to the public in this offering, minus $0.01, and the exercise price of each pre-funded warrant will be $0.01 per share. This offering also relates to the shares of common stock issuable upon exercise of any pre-funded warrants sold in this offering. For each pre-funded warrant we sell, the number of shares of common stock we are offering will be decreased on a one-for-one basis.
Our common stock is listed on The Nasdaq Capital Market under the symbol BNTC. On August 13, 2020, the last reported sale price of our common stock on The Nasdaq Capital Market was $8.31. There is no established public trading market for the pre-funded warrants, and we do not expect a market to develop. We do not intend to apply for listing of the pre-funded warrants on any securities exchange or other nationally recognized trading system. Without an active trading market, the liquidity of the pre-funded warrants will be limited.
The public offering price per share will be determined between us, the underwriter and purchasers based on market conditions at the time of pricing, and may be at a discount to the current market price of our common stock. Therefore, the recent market price used throughout this prospectus may not be indicative of the actual public offering price.
We are an emerging growth company, as that term is used in the Jumpstart Our Business Startups Act of 2012, and, as such, we have elected to comply with certain reduced public company reporting requirements.
You should read this prospectus, together with additional information described under the heading Where You Can Find More Information, carefully before you invest in any of our securities.
Investing in our securities involves a high degree of risk. See Risk Factors beginning on page 19.
| Per Share of Common Stock |
Per Pre-Funded Warrant |
Total | ||||||||||
| Public offering price |
$ | $ | $ | |||||||||
| Underwriting discounts and commissions(1) |
$ | $ | $ | |||||||||
| Proceeds to us (before expenses) |
$ | $ | $ | |||||||||
| (1) | We have agreed to reimburse certain expenses of the underwriter, including a management fee equal to 1% of the gross proceeds of this offering, which are not included in the table above. See Underwriting for a description of the compensation payable to the underwriter. |
We have granted the underwriter a 30-day option to purchase an aggregate of up to additional shares of our common stock from us at the public offering price per share, less the underwriting discounts and commissions. See Underwriting.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed on the adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense.
Delivery of the securities offered hereby is expected to be made on or about , 2020.
Sole Book-Running Manager
H.C. Wainwright & Co.
The date of this prospectus is , 2020
Table of Contents
| Page | ||||
| 1 | ||||
| 2 | ||||
| 2 | ||||
| 3 | ||||
| 5 | ||||
| 15 | ||||
| 17 | ||||
| 19 | ||||
| 66 | ||||
| 67 | ||||
| 68 | ||||
| Market Price of Our Common Stock and Related Stockholder Matters |
70 | |||
| 70 | ||||
| Managements Discussion and Analysis of Financial Condition and Results of Operations |
71 | |||
| 84 | ||||
| 112 | ||||
| 116 | ||||
| 122 | ||||
| 123 | ||||
| 125 | ||||
| 126 | ||||
| 131 | ||||
| 139 | ||||
| 144 | ||||
| 144 | ||||
| 145 | ||||
i
Table of Contents
This prospectus is part of a registration statement on Form S-1 that we filed with the Securities and Exchange Commission (SEC). You should rely only on the information contained in this prospectus or contained in any free writing prospectus prepared by or on behalf of us or to which we have referred you. We have not, and the underwriter has not, authorized anyone to provide you with information that is different from that contained in such prospectuses. We and the underwriter are offering to sell shares of our common stock and pre-funded warrants which may be exercised for shares of common stock, and seeking offers to buy shares of our common stock and our pre-funded warrants, only in jurisdictions where such offers and sales are permitted. For investors outside the United States: We have not, and the underwriter has not, taken any action that would permit this offering or possession or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than in the United States. Persons outside the United States who come into possession of this prospectus must inform themselves about, and observe any restrictions relating to, the offering of the securities covered hereby and the distribution of this prospectus outside the United States. The information in this prospectus is accurate only as of the date of this prospectus, regardless of the time of delivery of this prospectus or any sale of our common stock.
This prospectus and the information incorporated herein by reference contain summaries of certain provisions contained in some of the documents described herein, but reference is made to the actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred to herein have been filed, will be filed or will be incorporated by reference as exhibits to the registration statement of which this prospectus is a part, and you may obtain copies of those documents as described below under the section entitled Where You Can Find Additional Information. We urge you to read carefully this prospectus, together with the information incorporated herein by reference before deciding whether to participate in the offering hereunder.
We further note that the representations, warranties and covenants made by us in any document that is filed as an exhibit to the registration statement of which this prospectus is a part and in any document that is incorporated by reference herein were made solely for the benefit of the parties to such agreement, including, in some cases, for the purpose of allocating risk among the parties to such agreements, and should not be deemed to be a representation, warranty or covenant to you. Moreover, such representations, warranties or covenants were accurate only as of the date when made. Accordingly, such representations, warranties and covenants should not be relied on as accurately representing the current state of our affairs.
Unless the context otherwise requires, the terms Benitec, the Company, we, us, our and similar terms used in this prospectus refer (i), prior to the Re-domiciliation (as defined herein) to Benitec Biopharma Limited, an Australian corporation, and its subsidiaries, and (ii), following the Re-domiciliation, to Benitec Biopharma Inc., a Delaware corporation, and its subsidiaries (including Benitec Limited). Any references to Benitec Limited refer to Benitec Biopharma Limited, an Australian corporation.
All references to $ in this prospectus refer to U.S. dollars. All references to A$ in this prospectus mean Australian dollars. As of June 30, 2020, the rate of exchange of U.S. dollars to Australian dollars was 1.4489 AUD.
Our fiscal year-end is June 30. References to a particular fiscal year are to our fiscal year ended June 30 of that calendar year.
1
Table of Contents
This prospectus includes information with respect to market and industry conditions and market share from third-party sources or based upon estimates using such sources when available. We believe that such information and estimates are reasonable and reliable. We also believe the information extracted from publications of third-party sources has been accurately reproduced. However, we have not independently verified any of the data from third-party sources. Similarly, our internal research is based upon our understanding of industry conditions, and such information has not been verified by any independent sources.
We have proprietary and licensed rights to trademarks used in this prospectus which are important to our business, many of which are registered under applicable intellectual property laws. These trademarks include:
| | BENITEC BIOPHARMA® |
| | BENITEC® |
| | GIVING DISEASE THE SILENT TREATMENT® |
| | SILENCING GENES FOR LIFE® |
Solely for convenience, trademarks and trade names referred to in this prospectus appear without the ® or symbols, but such references are not intended to indicate, in any way, that we will not assert, to the fullest extent possible under applicable law, our rights or the rights of the applicable licensor to these trademarks and trade names. We do not intend our use or display of other companies trade names, trademarks or service marks to imply a relationship with, or endorsement or sponsorship of us by, any other companies. Each trademark, trade name or service mark of any other company appearing in this prospectus is the property of its respective holder.
2
Table of Contents
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements that are subject to a number of risks and uncertainties, many of which are beyond our control. All statements, other than statements of historical fact included in this prospectus, regarding our strategy, future operations, financial position, projected costs, prospects, plans and objectives of management are forward-looking statements. When used in this prospectus, the words could, believe, anticipate, intend, estimate, expect, may, continue, predict, potential, project, or the negative of these terms, and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain such identifying words. These statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, levels of activity, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. Although we believe that we have a reasonable basis for each forward-looking statement contained in this prospectus, we caution you that these statements are based on a combination of facts and important factors currently known by us and our expectations of the future, about which we cannot be certain.
Forward-looking statements may include statements about:
| | our plans to develop and potentially commercialize our product candidates; |
| | the timing of the initiation and completion of preclinical studies and clinical trials; |
| | the timing of patient enrollment and dosing in any future clinical trials; |
| | the timing of the availability of data from clinical trials; |
| | the timing of expected regulatory filings and approvals; |
| | regulatory developments in the United States; |
| | the development of novel AAV vectors; |
| | expectations about the plans of licensees of our technology; |
| | the clinical utility and potential attributes and benefits of ddRNAi and our product candidates, including the potential duration of treatment effects and the potential for a one shot cure; |
| | potential future out-licenses and collaborations; |
| | our expectations regarding expenses, ongoing losses, future revenue, capital needs and needs for additional financing; |
| | our use of proceeds from this offering; |
| | the length of time over which we expect our cash and cash equivalents to be sufficient to execute on our business plan; |
| | our intellectual property position and the duration of our patent portfolio; and |
| | the impact of the current COVID-19 pandemic, the disease caused by the SARS-CoV-2 virus, which may adversely impact our business and preclinical and future clinical trials. |
as well as other forward-looking statements detailed under the caption Risk Factors in this prospectus and in other reports filed with the SEC.
We have based the forward-looking statements included in this prospectus and in the documents incorporated herein by reference on information available to us on the date of this prospectus or on the date thereof. Except as required by law we undertake no obligation to revise or update any forward-looking statements, whether as a result of new information, future events or otherwise. You are advised to consult any additional disclosures that we may make directly to you or through reports that we, in the future, may file with
3
Table of Contents
the SEC, including annual reports on Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K.
All forward-looking statements included herein or in documents incorporated herein by reference are expressly qualified in their entirety by the cautionary statements contained or referred to elsewhere in this prospectus.
4
Table of Contents
This summary highlights information contained in other parts of this prospectus and in the documents incorporated by reference herein and does not contain all of the information that you should consider in making your investment decision. Before investing in our securities, you should carefully read this entire prospectus and the documents incorporated by reference herein, including our consolidated financial statements and the related notes, and the information set forth under the sections titled Risk Factors. Some of the statements in this prospectus and the documents incorporated by reference herein constitute forward-looking statements that involve risks and uncertainties. See information set forth under the section Cautionary Note Regarding Forward-Looking Statements.
Company Overview
We endeavor to become the leader in discovery, development, and commercialization of therapeutic agents capable of addressing significant unmet medical need via the application of the silence and replace approach to the treatment of genetic disorders.
Benitec Biopharma, Inc. (Benitec or the Company or in the third person, we or our) is a development-stage biotechnology company focused on the advancement of novel genetic medicines with headquarters in Hayward, California. The proprietary platform, called DNA-directed RNA interference, or ddRNAi, combines RNA interference, or RNAi, with gene therapy to create medicines that facilitate sustained silencing of disease-causing genes following a single administration. The Company is developing ddRNAi-based therapeutics for chronic and life-threatening human conditions including Oculopharyngeal Muscular Dystrophy (OPMD), and Chronic Hepatitis B.
BB-301 is the most advanced ddRNAi-based genetic medicine currently under development by Benitec. BB-301 is an internally optimized, adeno-associated virus (AAV) -based gene therapy agent that is designed to both silence the expression of mutated, disease-causing genes (to slow, or halt, the underlying mechanism of disease progression) and replace the mutant genes with normal, wild type genes (to drive restoration of function in diseased cells). This fundamental approach to disease management is called silence and replace and this biological mechanism offers the potential to restore the underlying physiology of the treated tissues and, in the process, improve treatment outcomes for patients suffering from the chronic and, potentially, fatal effects of Oculopharyngeal Muscular Dystrophy (OPMD). BB-301 has been granted Orphan Drug Designation in the United States and the European Union.
Through the combination of the targeted gene silencing effects of RNAi and the durable transgene expression achievable via the use of modified viral vectors, the silence and replace approach has the potential to produce long-term silencing of disease-causing genes along with simultaneous replacement of wild type gene function following a single administration of the proprietary genetic medicine. We believe this novel attribute of the investigational agents under development by Benitec may facilitate the achievement of robust clinical activity while greatly reducing the dosing frequencies traditionally expected for medicines employed for the management of chronic diseases. Additionally, the establishment of chronic gene silencing and gene replacement may significantly reduce the risk of patient non-compliance during the course of medical management of potentially fatal clinical disorders.
We will require additional financing to progress our product candidates through to key inflection points.
5
Table of Contents
Our Strengths
We believe that the combination of our proprietary ddRNAi technology and our deep expertise in the design and development of genetic medicines, and specifically ddRNAi-based therapeutics, will enable us to achieve and maintain a leading position in gene silencing and gene therapy for the treatment of human disease. Our key strengths include:
| | A first mover advantage for ddRNAi-based therapeutics; |
| | A proprietary ddRNAi-based silence and replace technology platform that may potentially enable the serial development of single-administration therapeutics capable of facilitating sustained, long-term silencing of disease-causing genes and concomitant replacement of wildtype gene function; |
| | A proprietary AAV vector technology which improves the endosomal escape capability of virus produced in insect cells using a baculovirus system. This technology has broad application in AAV-based gene therapies; |
| | The capabilities to drive the development of a pipeline of programs focused on chronic diseases with either large patient populations, including Chronic Hepatitis B virus infection, or rare diseases, which may potentially support the receipt of Orphan Drug Designation, including OPMD; and |
| | A growing portfolio of patents protecting improvements to our ddRNAi, and silence and replace, technology and product candidates through at least 2036, with additional patent life anticipated through at least 2040. |
Our Strategy
We endeavor to become the leader in discovery, development, and commercialization of therapeutic agents capable of addressing significant unmet medical need via the application of the silence and replace approach to the treatment of genetic disorders. We apply the following general strategy to drive the Company toward these goals:
| | Selectively develop proprietary and partnered programs; and |
| | Continue to explore and secure research and development partnerships with global biopharmaceutical companies supported by the differentiated nature of our scientific platform and intellectual property portfolio. |
Our senior leadership team will continue to explore partnership opportunities with global biopharmaceutical companies, as we expect that the unique attributes of the proprietary ddRNAi and silence and replace approaches, and the breadth of potential clinical indications amenable to our proprietary methods, to support the formation of collaborations over a broad range of diseases with significant unmet medical need.
We actively protect our intellectual property and proprietary technology. These efforts are central to the growth of our business and include:
| | Seeking and maintaining patents claiming our ddRNAi and silence and replace technologies, modified AAV vectors and manufacture processes, and other inventions relating to our specific products in development or that are otherwise commercially and/or strategically important to the development of our business; |
| | Protecting and enforcing our intellectual property rights; and |
| | Strategically licensing intellectual property from third parties to advance development of our product candidates. |
6
Table of Contents
Our TechnologyddRNAi and Silence and Replace
Our proprietary technology platforms are designated as DNA-directed RNA interference, or ddRNAi, and silence and replace. ddRNAi is designed to produce long-term silencing of disease-causing genes, by combining RNA interference, or RNAi, with viral delivery agents typically associated with the field of gene therapy (i.e. viral vectors). Modified AAV vectors are employed to deliver genetic constructs that encode short hairpin RNAs that are, then, serially expressed and processed, to produce small interfering RNA, or siRNA, molecules within the transduced cell for the duration of the life of the target cell. These newly introduced siRNA molecules drive long-term, and potentially permanent, silencing of the expression of the disease-causing gene. The silence and replace approach further bolsters the biological benefits of long-term silencing of disease-causing genes by incorporating multifunctional genetic constructs within the modified AAV vectors to create an AAV-based gene therapy agent that is designed to both silence the expression of mutated, disease-causing genes (to slow, or halt, the underlying mechanism of disease progression) and, simultaneously, replace the mutant genes with normal, wild type genes (to drive restoration of function in diseased cells). This fundamentally distinct approach to disease management offers the potential to restore the underlying physiology of the treated tissues and, in the process, improve treatment outcomes for patients suffering from the chronic and, potentially, fatal effects of diseases like Oculopharyngeal Muscular Dystrophy (OPMD).
Traditional gene therapy is defined by the introduction of an engineered transgene to correct the pathophysiological derangements derived from mutated or malfunctioning genes. Mutated genes can facilitate the intracellular production of disease-causing proteins or hamper the production of critical, life-sustaining, proteins, and the introduction of a new transgene can facilitate the restoration of production of normal proteins within the diseased cell, thus, restoring natural biological function. Critically, the implementation of this traditional method of gene therapy cannot eliminate the expression, or the potential deleterious effects of, the underlying mutant gene (as mutant proteins may be continually expressed and aggregate or drive the aggregation of other native proteins within the diseased cell). In this regard, the dual capabilities of the proprietary silence and replace approach to silence a disease-causing gene via ddRNAi and simultaneously replace the wildtype activity of a mutant gene via the delivery of an engineered transgene could facilitate the development of differentially efficacious treatments for a range of genetic disorders.
Overview of RNAi and the siRNA Approach
The mutation of a single gene can cause a chronic disease via the resulting intracellular production of a disease-causing protein (i.e. an abnormal form of the protein of interest), and many chronic and/or fatal disorders are known to result from the inappropriate expression of a single gene or multiple genes. In some cases, genetic disorders of this type can be treated exclusively by silencing the intracellular production of the disease-causing protein through well-validated biological approaches like RNA interference (RNAi). RNAi employs small nucleic acid molecules to activate an intracellular enzyme complex, and this biological pathway temporarily reduces the production of the disease-causing protein. In the absence of the disease-causing protein, normal cellular function is restored and the chronic disease that initially resulted from the presence of the mutant protein is partially or completely resolved. RNAi is potentially applicable to over 20,000 human genes and a large number of disease-causing microorganism-specific genes.
7
Table of Contents
Figure 1. The siRNA Approach
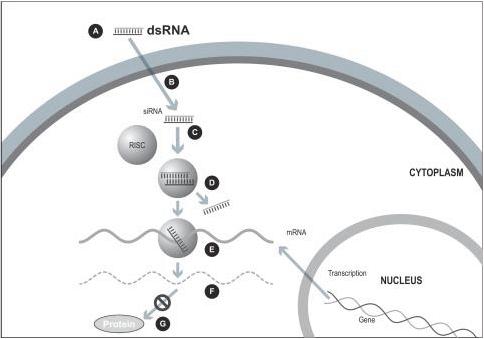
A small double stranded RNA, or dsRNA, molecule (A, Figure 1), comprising one strand known as the sense strand and another strand known as the antisense strand, which are complementary to each other, is synthesized in the laboratory. These small dsRNAs are called small interfering RNAs, or siRNAs. The sequence of the sense strand corresponds to a short region of the target gene mRNA. The siRNA is delivered to the target cell (B, Figure 1), where a group of enzymes, referred to as the RNA-Induced Silencing Complex, or RISC, process the siRNA (C, Figure 1), where one of the strands (usually the sense strand) is released (D, Figure 1). RISC uses the antisense strand to find the mRNA that has a complementary sequence (E, Figure 1) leading to the cleavage of the target mRNA (F, Figure 1). As a consequence, the output of the mRNA (protein production) does not occur (G, Figure 1). Several companies, including Alnylam Pharmaceuticals Inc. (Alnylam), Arbutus Biopharma Corp (Arbutus), and Dicerna Pharmaceuticals Inc. (Dicerna), utilize this approach in their RNAi product candidates.
Importantly, many genetic disorders are not amenable to the traditional gene silencing approach outlined in Figure 1, as the diseased cells may produce a mixture of the wildtype protein of interest and the disease-causing, mutant variant of the protein, and the underlying genetic mutation may be too small to allow for selective targeting of the disease-causing variant of the protein through the use of siRNA-based approaches exclusively. In these cases, it is extraordinarily difficult to selectively silence the disease-causing protein without simultaneously silencing the wildtype intracellular protein of interest whose presence is vital to the conduct of normal cellular functions.
Our proprietary silence and replace technology utilizes the unique specificity and robust gene silencing capabilities of RNAi while overcoming many of the key limitations of siRNA-based approaches to disease management.
8
Table of Contents
Our Approach to the Treatment of Genetic DiseasesddRNAi and Silence and Replace
Our proprietary silence and replace approach to the treatment of genetic diseases combines RNAi with wildtype gene replacement to drive sustained silencing of disease-causing genes and concomitant restoration of functional wildtype genes following a single administration of the therapeutic agent. Benitec employs ddRNAi in combination with classical gene therapy (i.e. transgene delivery via viral vectors) to overcome several of the fundamental limitations of RNAi.
The silence and replace approach to the treatment of genetic disorders employs adeno-associated viral vectors (AAVs) to deliver genetic constructs which may, after a single administration to the target tissues:
| | Chronically express RNAi molecules inside of the target, diseased, cells (to serially silence the intracellular production of mutant, disease-causing, protein and the wildtype protein of interest); |
| | Simultaneously drive the expression of a wildtype variant of the protein of interest (to restore native intracellular biological processes); and |
| | AAV vectors can accommodate the multi-functional DNA expression cassettes containing the engineered wildtype transgenes and the novel genes encoding short hairpinRNA/microRNA molecules (shRNA/miRNA) that are required to support the development of therapeutic agents capable of the achievement of the goals of the silence and replace approach to therapy. |
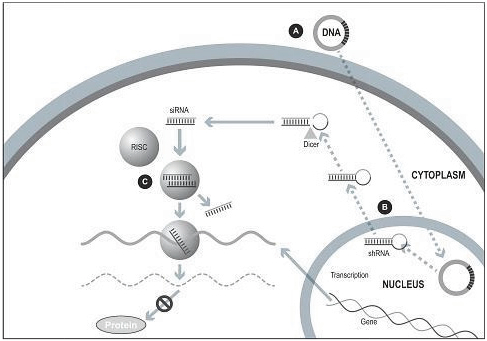
Our silence and replace technology utilizes proprietary DNA expression cassettes to foster continuous production of gene silencing shRNAs and wildtype proteins (via expression of the wildtype transgene). A range of viral and non-viral gene therapy vectors can be used to deliver the DNA construct into the nucleus of the target cell and, upon delivery, shRNA molecules are expressed and subsequently processed by intracellular enzymes into siRNA molecules that silence the expression of the mutant, disease causing protein (Figure 2).
In the silence and replace approach (Figure 2):
| | A DNA construct is delivered to the nucleus of the target cell by a gene therapy vector (A) such as an AAV; |
| | Once inside of the nucleus, the DNA construct drives the continuous production of shRNA molecules (B) which are processed by an enzyme called Dicer into siRNAs (C); and |
| | The processed siRNA is incorporated into RISC and silences the target gene using the same mechanism shown in Figure 1. |
| | When the DNA expression cassette is also comprised of a wildtype transgene, upon entry of the DNA construct into the nucleus of the target cell via the use of the AAV vector, the DNA construct also drives the continuous production of wildtype protein (to restore native intracellular biological processes) |
9
Table of Contents
Figure 2. The Silence and Replace Approach
Our Pipeline
The following table sets forth our current product candidates and their development status:
Table 1. Pipeline: Oculopharyngeal Muscular Dystrophy and Chronic Hepatitis B Virus Infection
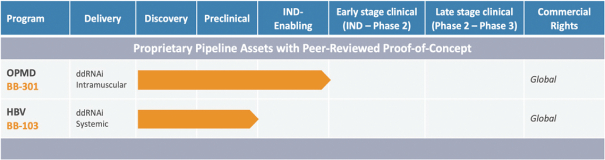
BB-301
BB-301 is a late-stage nonclinical investigational agent currently in development for the treatment of Oculopharyngeal Muscular Dystrophy. BB-301 is the lead pipeline program for Benitec, and IND-enabling studies are currently being conducted. A summary of the BB-301 program is provided in Figure 3.
10
Table of Contents
Figure 3. Overview of the BB-301 Program

BB-103
BB-103 has demonstrated robust nonclinical activity during the evaluation of this agent for the treatment of Chronic Hepatitis B Virus infection. Benitec is currently seeking strategic partners to advance BB-103 through IND-enabling studies.
Risk Factors
An investment in our securities involves a high degree of risk. Any of the factors set forth under Risk Factors may limit our ability to successfully execute our business strategy. You should carefully consider all of the information set forth in this prospectus and, in particular, should evaluate the specific factors set forth under Risk Factors in deciding whether to invest in our securities. These risk factors include, among others:
| | We have incurred significant net losses and anticipate that we will continue to incur significant net losses for the foreseeable future. We may never achieve or maintain profitability; |
| | We have never generated any revenue from product sales and may never be profitable; |
| | Even if this offering is successful, we will need to raise additional funding, which may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us to delay, limit or discontinue our product development efforts or other operations; |
| | Currently, no product candidates utilize ng ddRNAi technology have been approved for commercial sale, and our approach to the development of ddRNAi technology may not result in safe, effective or marketable products; |
| | We are early in our product development efforts and have only one product candidate in early-stage clinical trials. All of our other current product candidates are still in preclinical development. We may |
11
Table of Contents
| not be able to obtain regulatory approvals for the commercialization of some or all of our product candidates; |
| | Issues which may impact ddRNAi delivery into the cell could limit our ability to develop and commercialize product candidates; |
| | If other companies develop technologies or product candidates for our target disease indications more rapidly than we do or if their technologies, including delivery technologies, are more effective, our ability to develop and successfully commercialize product candidates may be compromised; and |
| | If we are unable to obtain or protect intellectual property rights related to our product candidates, we may not be able to obtain exclusivity for our product candidates or prevent others from developing similar competitive products. |
Recent Developments
Re-domiciliation
On April 15, 2020, or the Implementation Date, the re-domiciliation, or the Re-domiciliation, of Benitec Biopharma Limited, a public company incorporated under the laws of the State of Western Australia, or Benitec Limited, was completed in accordance with the Scheme Implementation Agreement, as amended and restated as of January 30, 2020, between Benitec Limited and us. As a result of the Re-domiciliation, our jurisdiction of incorporation was changed from Australia to Delaware, and Benitec Limited became our wholly owned subsidiary.
The Re-domiciliation was effected pursuant to a statutory scheme of arrangement under Australian law, or the Scheme, whereby on the Implementation Date, all of the issued and outstanding ordinary shares of Benitec Limited were exchanged for newly issued shares of our common stock, on the basis of one share of our common stock, par value $0.0001 per share, for every 300 ordinary shares of Benitec Limited issued and outstanding. Holders of Benitec Limiteds American Depository Shares, or ADSs (each of which represented 200 ordinary shares), received two shares of our common stock for every three ADSs held.
Our common stock began trading on The Nasdaq Capital Market, or Nasdaq, at the start of trading on the Implementation Date under the symbol BNTC.
COVID-19 Pandemic
In December 2019, an outbreak of a novel strain of coronavirus was identified in Wuhan, China. This virus continues to spread globally, has been declared a pandemic by the World Health Organization and has spread to nearly every country, including Australia and the United States. The impact of this pandemic has been and will likely continue to be extensive in many aspects of society, which has resulted in and will likely continue to result in significant disruptions to businesses and capital markets around the world. The extent to which the coronavirus impacts us will depend on future developments, which are highly uncertain and cannot be predicted, including new information which may emerge concerning the severity of the coronavirus and the actions to contain the coronavirus or treat its impact, among others.
Certain of our research and development efforts are conducted globally, including the ongoing development of our silence and replace therapeutic for the treatment of Oculopharyngeal Muscular Dystrophy (OPMD), and will be dependent upon our ability to initiate preclinical and clinical studies despite the ongoing COVID-19 pandemic. As we continue to actively advance our preclinical programs, including our ongoing tissue transduction studies for BB-301, we are in close contact with our principal investigators and preclinical trial sites, which are primarily located in the France, and are assessing the impact of COVID-19 on our studies and the expected development timelines and costs of all of our product candidates, on an ongoing basis. In light of recent developments relating to the COVID-19 global pandemic, the focus of healthcare providers and hospitals on
12
Table of Contents
fighting the virus, and consistent with the FDAs updated industry guidance for conducting clinical trials issued on March 18, 2020, we have experienced delays to the original timeline regarding the initiation and anticipated completion of the ongoing BB-301 IND-enabling development work. The initiation of the BB-301 tissue transduction study, which represents a key component of the IND-enabling work, was delayed by several months, however, the study has been recently initiated and the dosing of the initial preclinical cohorts has proceeded without incident. We will continue to evaluate the impact of the COVID-19 pandemic on our business and expect to reevaluate the timing of our anticipated preclinical and clinical milestones as we learn more and the impact of COVID-19 on our industry becomes more clear.
We had also implemented work-from-home measures for the majority of our employees between March 2020 and June 2020, resulting in a reduction of laboratory work and a halt of non-essential business travel. As we transition our employees back to our premises, there is a risk that COVID-19 infections occur at our offices or laboratory facilities and significantly affect our operations. Additionally, if any of our critical vendors are impacted, our business could be affected if we become unable to timely procure essential equipment, supplies or services in adequate quantities and at acceptable prices.
Corporate Information
We were incorporated as a Delaware corporation on April 13, 2020 in connection with the Re-domiciliation completed on April 15, 2020. Our predecessor, Benitec Limited, was incorporated under the laws of Australia in 1995. Our stock is traded on The Nasdaq Capital Market under the symbol BNTC. Our principal executive offices are located at 3940 Trust Way, Hayward, California 94545. Our telephone number is (510) 780-0819, and our Internet website is www.benitec.com. The information on, or that can be accessed through, our website is not part of this prospectus and is not incorporated by reference herein.
Implications of Being an Emerging Growth Company and a Smaller Reporting Company
We are an emerging growth company as defined in the Jumpstart Our Business Startups Act of 2012, as amended (JOBS Act). We will remain an emerging growth company until the earliest to occur of: the last day of the fiscal year in which we have more than $1.07 billion in annual revenue; the date we qualify as a large accelerated filer, with at least $700 million of equity securities held by non-affiliates; the issuance, in any three-year period, by us of more than $1.0 billion in non-convertible debt securities; and June 30, 2021. As a result of this status, we have taken advantage of reduced reporting requirements in this prospectus and may elect to take advantage of other reduced reporting requirements in our future filings with the SEC. In particular, in this prospectus, we have not included all of the executive compensation related information that would be required if we were not an emerging growth company. In addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards, delaying the adoption of these accounting standards until they would apply to private companies. We have irrevocably elected not to avail ourselves of this exemption and, as a result, we will adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required for other public companies that are not emerging growth companies. We intend to rely on other exemptions provided by the JOBS Act, including without limitation, not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act.
We are also a smaller reporting company and will remain a smaller reporting company while either (i) the market value of our stock held by non-affiliates was less than $250 million as of the last business day of our most recently completed second fiscal quarter or (ii) our annual revenue was less than $100 million during our most recently completed fiscal year and the market value of our stock held by non-affiliates was less than $700 million as of the last business day of our most recently completed second fiscal quarter. If we are a smaller reporting
13
Table of Contents
company at the time we cease to be an emerging growth company, we may continue to rely on exemptions from certain disclosure requirements that are available to smaller reporting companies, including many of the same exemptions from disclosure requirements as those that are available to emerging growth companies, such as reduced disclosure obligations regarding executive compensation in our registration statements, prospectus and our periodic reports and proxy statements. For so long as we remain a smaller reporting company, we are permitted and intend to rely on exemptions from certain disclosure and other requirements that are applicable to other public companies that are not smaller reporting companies.
14
Table of Contents
| Common stock offered by us |
shares (or shares, if the underwriters option to purchase additional shares is exercised in full). |
| Pre-funded warrants offered by us |
We are also offering to certain purchasers whose purchase of shares of common stock in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if such purchasers so choose, pre-funded warrants in lieu of shares of common stock that would otherwise result in any such purchasers beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock. The exercise price of each pre-funded warrant will equal $0.01 per share. Each pre-funded warrant will be exercisable upon issuance and will not expire prior to exercise. We are offering up to shares of common stock and up to pre-funded warrants, in the aggregate not exceeding more than a total of shares of common stock and pre-funded warrants. For each pre-funded warrant we sell, the number of shares of common stock we are offering will be decreased on a one-for-one basis. This prospectus also relates to the offering of the shares of common stock issuable upon exercise of the pre-funded warrants. |
| Option to purchase additional shares |
We have granted the underwriter a 30-day option to purchase an aggregate of up to additional shares of our common stock from us at the public offering price per share, less the underwriting discounts and commissions. See Underwriting. |
| Common stock to be outstanding immediately following this offering |
shares (or shares, if the underwriters option to purchase additional shares is exercised in full) assuming we sell only shares of common stock in this offering. |
| Use of proceeds |
We estimate that the net proceeds from this offering will be approximately $ million ($ million if the underwriters option to purchase additional shares is exercised in full), based on an assumed public offering price per share of common stock of $ , the last reported sale price of our common stock on the Nasdaq Capital Market on , 2020, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and assuming we sell only shares of common stock in this offering. We intend to use the net proceeds from this financing for the continued advancement of development activities for our product pipeline, general corporate purposes, and strategic growth opportunities. See Use of Proceeds. |
| Dividend policy |
For the foreseeable future, we currently intend to retain all available funds and any future earnings to support our operations and to finance the growth and development of our business. |
15
Table of Contents
| Risk factors |
An investment in our securities involves a high degree of risk. You should read the Risk Factors section of this prospectus for a discussion of factors to consider carefully before deciding to invest in our securities. |
| Nasdaq symbol |
BNTC. |
| No listing of pre-funded warrants |
We do not intend to apply for listing of the pre-funded warrants on any national securities exchange or trading system. |
The number of shares of common stock to be outstanding after this offering is based on 1,108,374 shares of common stock outstanding at August 10, 2020 and excludes as of such date the following:
| | 70,162 shares of common stock issuable upon exercise of stock options outstanding as of August 10, 2020 at a weighted-average exercise price of $60.82 per share; and |
| | 215,099 shares of common stock issuable upon the exercise of warrants exercisable for shares of common stock outstanding as of August 10, 2020 at a weighted-average exercise price of $32.31 per share. |
Unless otherwise indicated, all information in this prospectus assumes no purchaser elects to purchase pre-funded warrants and no exercise by the underwriter of its option to purchase additional shares.
16
Table of Contents
SUMMARY CONSOLIDATED FINANCIAL DATA
The following summary consolidated financial data as of and for the years ended June 30, 2019 and 2018 are derived from our audited consolidated financial statements included in this prospectus. The summary consolidated financial data as of and for the nine months ended March 31, 2020 and 2019 have been derived from our unaudited consolidated financial statements included in this prospectus. These unaudited consolidated financial statements have been prepared on a basis consistent with our audited consolidated financial statements and, in our opinion, contain all adjustments, consisting only of normal recurring adjustments, necessary for a fair presentation of such consolidated financial data. You should read this data together with our consolidated financial statements and related notes included elsewhere in this prospectus. Our historical results are not necessarily indicative of our future results, and are not necessarily indicative of the results that may be expected for any interim periods, or any future year or period.
| Nine months ended March 31, | Year ended June 30, | |||||||||||||||
| (in thousands) | 2020 | 2019 | 2019 | 2018 | ||||||||||||
| Statements of operations data: |
||||||||||||||||
| Revenues: |
||||||||||||||||
| Revenues from customers |
$ | 137 | $ | 10,955 | $ | 11,551 | $ | 292 | ||||||||
| Government research and development grants |
| 601 | 648 | 3,098 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total revenues |
137 | 11,556 | 12,199 | 3,390 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Expenses: |
||||||||||||||||
| Royalties and license fees |
(233 | ) | 405 | 435 | 349 | |||||||||||
| Research and development |
2,095 | 3,304 | 4,567 | 7,704 | ||||||||||||
| General and administrative |
3,669 | 3,754 | 4,614 | 4,539 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total expenses |
5,531 | 7,463 | 9,616 | 12,592 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Income (Loss) from Operations |
(5,394 | ) | 4,093 | 2,583 | (9,202 | ) | ||||||||||
| Other Income (Loss): |
||||||||||||||||
| Foreign currency transaction gain (loss) |
4 | (75 | ) | (75 | ) | (30 | ) | |||||||||
| Finance income, net |
52 | 80 | 122 | 187 | ||||||||||||
| Unrealized gain (loss) on investment |
(1 | ) | (20 | ) | (21 | ) | (32 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total other income (loss) |
55 | (15 | ) | 26 | 125 | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net Income (Loss) |
(5,339 | ) | 4,078 | 2,609 | (9,077 | ) | ||||||||||
| Other Comprehensive Income (Loss): |
||||||||||||||||
| Unrealized foreign currency translation gain (loss) |
(869 | ) | (36 | ) | (531 | ) | (219 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total Other Comprehensive Income (Loss) |
(869 | ) | (36 | ) | (531 | ) | (219 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total Comprehensive Income (Loss) |
$ | (6,208 | ) | $ | 4,042 | $ | 2,078 | $ | (9,296 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net Income (Loss) Available to Common Stockholders |
$ | (5,339 | ) | $ | 4,078 | $ | 2,609 | $ | (9,077 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Basic and Diluted Weighted-Average Number of Shares Outstanding |
995,246 | 856,765 | 856,765 | 701,516 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Basic and Diluted Income (Loss) Per Share of Common Stock |
$ | (5.36 | ) | $ | 4.76 | $ | 3.05 | $ | (12.94 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
17
Table of Contents
| March 31, | June 30, | |||||||||||||||
| 2020 | 2019 | 2019 | 2018 | |||||||||||||
| Balance sheet data: |
||||||||||||||||
| Cash and cash equivalents |
$ | 11,400 | $ | 17,677 | $ | 15,718 | $ | 11,879 | ||||||||
| Working capital |
11,747 | 18,011 | 16,115 | 13,552 | ||||||||||||
| Total assets |
13,580 | 20,244 | 19,235 | 15,760 | ||||||||||||
| Total long-term liabilities |
263 | 6 | | 36 | ||||||||||||
| Total stockholders equity |
12,336 | 18,453 | 16,594 | 13,844 | ||||||||||||
18
Table of Contents
Investing in our securities involves a high degree of risk. You should carefully consider the risks and uncertainties described below together with all of the other information contained in this prospectus, including our consolidated financial statements and the related notes, before deciding to invest in our securities. The risks and uncertainties described below and in the documents incorporated by reference herein are not the only ones we face. Additional risks and uncertainties that we are unaware of, or that we currently believe are not material, may also become important factors that affect us. If any of the following risks actually occurs, our business, financial condition, results of operations and prospects could be materially and adversely affected, the trading price of our common stock could decline and you could lose all or part of your investment.
Risks Related to Our Financial Condition, Capital Requirements and Largest Shareholder
We have incurred significant losses since inception and anticipate that we will continue to incur losses for the foreseeable future. If we are unable to achieve or sustain profitability, the market value of our common stock will likely decline.
As of March 31, 2020 and June 30, 2019, we had accumulated losses of $113.8 million and $108.9 million, respectively. We have devoted most of our financial resources to research and development, including our clinical and preclinical development activities. To date, we and our predecessor have financed our operations primarily through the issuance of equity securities, research and development grants from the Australian government and payments from our collaboration partners. We do not expect to generate any significant revenue for the foreseeable future, and we expect to incur significant operating losses for the foreseeable future due to the cost of research and development, preclinical studies and clinical trials and the regulatory approval process for product candidates. The amount of our future net losses is uncertain and will depend, in part, on the rate of our future expenditures. Our ability to continue operations will depend on, among other things, our ability to obtain funding through equity or debt financings, strategic collaborations or additional grants.
We expect to continue to incur significant expenses, and we may incur operating losses for the foreseeable future. We anticipate that our expenses will increase substantially if and as we:
| | continue our research and preclinical development of our product candidates; |
| | expand the scope of our current preclinical studies for our product candidates or initiate clinical, additional preclinical or other studies for product candidates; |
| | seek regulatory and marketing approvals for any of our product candidates that successfully complete clinical trials; |
| | further develop the manufacturing process for our product candidates; |
| | change or add additional manufacturers or suppliers; |
| | seek to identify and validate additional product candidates; |
| | acquire or in-license other product candidates and technologies, which may or may not include those related to our ddRNAi technology and delivery vectors for our therapeutic candidates; |
| | maintain, protect and expand our intellectual property portfolio; |
| | create additional infrastructure to support our operations as a public company in the United States and our product development and future commercialization efforts; and |
| | experience any delays or encounter issues with any of the above. |
The net losses we incur may fluctuate significantly from quarter to quarter and year to year, such that a period-to-period comparison of our results of operations may not be a good indication of our future performance. In any particular quarter or quarters, our operating results could be below the expectations of securities analysts or investors, which could cause the price of our common stock to decline.
19
Table of Contents
We have never generated any revenue from product sales and may never be profitable.
Our ability to generate significant revenue and achieve profitability depends on our ability to, alone or with strategic collaboration partners, successfully complete the development of and obtain the regulatory approvals for our product candidates, to manufacture sufficient supply of our product candidates, to establish a sales and marketing organization or suitable third-party alternative for the marketing of any approved products and to successfully commercialize any approved products on commercially reasonable terms. All of these activities will require us to raise sufficient funds to finance business activities.
In the past we received milestone payments from our collaborative partners. However, we do not anticipate generating revenue from commercializing product candidates for the foreseeable future, if ever.
Our ability to generate future revenues from commercializing product candidates depends heavily on our success in:
| | establishing proof of concept in preclinical studies and clinical trials for our product candidates; |
| | successfully initiating and completing clinical trials of our product candidates; |
| | obtaining regulatory and marketing approvals for product candidates for which we complete clinical trials; |
| | maintaining, protecting and expanding our intellectual property portfolio, and avoiding infringing on intellectual property of third parties; |
| | establishing and maintaining successful licenses, collaborations and alliances with third parties; |
| | developing a sustainable, scalable, reproducible and transferable manufacturing process for our product candidates; |
| | establishing and maintaining supply and manufacturing relationships with third parties that can provide products and services adequate, in amount and quality, to support clinical development and commercialization of our product candidates, if approved; |
| | launching and commercializing any product candidates for which we obtain regulatory and marketing approval, either by collaborating with a partner or, if launched independently, by establishing a sales, marketing and distribution infrastructure; |
| | obtaining market acceptance of any product candidates that receive regulatory approval as viable treatment options; |
| | obtaining favorable coverage and reimbursement rates for our products from third-party payers; |
| | addressing any competing technological and market developments; |
| | identifying and validating new product candidates; and |
| | negotiating favorable terms in any collaboration, licensing or other arrangements into which we may enter. |
The process of developing product candidates for ddRNAi-based and antisense RNA-based therapeutics contains a number of inherent risks and uncertainties. For example, with regard to ddRNAi, it may not be possible to identify a target region of a disease-associated gene that has not been previously identified and/or patented by others, resulting in restrictions on freedom to operate for that target sequence. Silencing the target gene may not ultimately result in curing the disease as there may be more factors contributing to the development of the disease than the target gene. Silencing the target gene using ddRNAi may lead to short-term or long-term adverse effects that were not predicted or observed in preclinical studies. The delivery of the DNA construct to the target cells may not be possible, or complete or adequate to provide sufficient therapeutic benefit.
20
Table of Contents
Even if one or more of our product candidates is approved for commercial sale, we may incur significant costs associated with commercializing any approved product candidate. As one example, our expenses could increase beyond expectations if we are required by the United States Food and Drug Administration (FDA) or other regulatory agencies, domestic or foreign, to perform clinical and other studies in addition to those that we currently anticipate. Even if we are able to generate revenues from the sale of any approved products, we may not become profitable and may need to obtain additional funding to continue operations, which could have an adverse effect on our business, financial condition, results of operations and prospects.
Even if this offering is successful, we will need to continue our efforts to raise additional funding, which may not be available on acceptable terms, or at all. Failure to obtain capital when needed may negatively impact our ability to continue as a going concern.
Developing ddRNAi products is expensive, and we expect our research and development expenses to increase substantially in connection with our ongoing activities, particularly as we advance our product candidates in preclinical studies and in future clinical trials and as we undertake preclinical studies of new product candidates.
As of March 31, 2020 and June 30, 2019, our cash and cash equivalents were $11.4 million and $15.7 million, respectively. We estimate that the net proceeds from this offering will be approximately $ million ($ million assuming the full exercise of the underwriters option to purchase additional common stock), assuming a public offering price of $ per share, the last reported sale price of our common stock on the Nasdaq Capital Market on , 2020, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us, and assuming we sell only shares of common stock in this offering. We estimate that these net proceeds, together with our existing cash and cash equivalents, will be sufficient to fund our operations until approximately the first quarter of 2022. However, our operating plan may change as a result of many factors currently unknown to us, and we may need to seek additional funds sooner than planned, through public or private equity or debt financings, government grants or other third-party funding, strategic alliances and licensing arrangements or a combination of these approaches. In addition, because the length of time and activities associated with successful development of our product candidates is highly uncertain, we are unable to estimate the actual funds we will require for development and any approved marketing and commercialization activities. In any event, we will require additional capital to obtain regulatory approval for our product candidates and to commercialize any product candidates that receive regulatory approval.
Any additional fundraising efforts may divert our management from their day-to-day activities, which may compromise our ability to develop and commercialize our product candidates. In addition, we cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable to us, if at all. Moreover, the terms of any financing may adversely affect the holdings or the rights of our shareholders, and the issuance of additional securities, whether equity or debt, by us, or the possibility of such issuance, may cause the market price of our common stock to decline. If we incur indebtedness we may be required to agree to restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions that could compromise our ability to conduct our business. We could also seek financing through arrangements with collaborative partners at an earlier stage than would otherwise be desirable and we may be required to relinquish rights to some or all of our technologies or product candidates or otherwise agree to terms unfavorable to us.
If we are unable to obtain funding on a timely basis or on acceptable terms, we may be required to significantly curtail, delay or discontinue one or more of our research or development programs or the commercialization of any approved product candidates.
21
Table of Contents
Nant Capital, LLC is our largest shareholder and may have interests that are different from ours and our other shareholders.
Nant Capital, LLC, or Nant Capital, holds 26.4% of our outstanding common stock as of August 10, 2020. Nant Capital appointed Dr. Jerel Banks to the board of directors of Benitec Limited, and Dr. Banks remains on our Board of Directors (the Board) and is Chief Executive Officer of the Company. Prior to joining the Company, Dr. Banks was the Chief Investment Officer of Nant Capital.
We also entered into an exclusive sublicense agreement with an affiliate of Nant Capital and a research collaboration agreement with Nant Capital pursuant to which we agreed to develop and commercialize an epidermal growth factor receptor, or EGFR, antisense RNA product candidate for head and neck squamous cell carcinoma (HNSCC), which we call BB-401. We terminated the exclusive sublicense agreement for convenience, with the termination effective as of June 2020, because the BB-401 program has been terminated. Prior to termination of the exclusive sublicense agreement, we were required to make periodic payments to Nant Capitals affiliate for so long as the agreement remained in effect, as well as contingent milestone and royalty payments in connection with the development and sale of the licensed technology.
As a result of its ownership of our common stock and its nomination of a director to our Board, Nant Capital may exert influence over us, including election of our Board, corporate transactions and strategic decisions. The interests of Nant Capital may differ from or conflict with the interests of our other shareholders. In the future, if Nant Capital does not support a merger, tender offer, sale of assets or other business combination because it judges that transaction to be inconsistent with Nant Capitals investment strategy, we may be unable to enter into or consummate a transaction that would enable other shareholders to realize a premium over the then-prevailing market prices for our common stock. Furthermore, if Nant Capital sells substantial amounts of our common stock to enhance its liquidity position, fund alternative investments or for other reasons, the trading price of our common stock could decline significantly, and other shareholders may be unable to sell their shares of our common stock at favorable prices. We cannot predict or control how Nant Capital may use the influence it has as a result of its common stock holdings.
We may be unable to conduct and conclude clinical trials for our product candidate for OPMD if we are unable to raise additional financing apart from this offering.
We have sufficient funds at this time to complete the pre-clinical studies for BB-301 and to initiate the clinical studies for BB-301, however, we expect that we will need to raise additional funds to complete the clinical studies. If we are unable to raise additional funds, and thus unable to continue our studies, our business operations may be adversely affected.
Risks Related to the Product Development and Regulatory Approval of Our Product Candidates
Our product candidates are based on ddRNAi and silence and replace technology. Currently, no product candidates utilizing ddRNAi technology or silence and replace technology have been approved for commercial sale and our approach to the development of ddRNAi technology and silence and replace technology may not result in safe, effective or marketable products.
We have concentrated our product research and development efforts on our ddRNAi technology and silence and replace technology, and our future success depends on successful clinical development of these technologies. We plan to progress our product candidates using our ddRNAi technology and our silence and replace technology for the treatment of the life-threatening conditions of OPMD and hepatitis B.
The scientific research that forms the basis of our efforts to develop product candidates is based on the therapeutic use of ddRNAi, and the identification, optimization and delivery of ddRNAi-based product candidates is relatively new. The scientific evidence to support the feasibility of successfully developing therapeutic treatments based on ddRNAi is preliminary and limited. There can be no assurance that any
22
Table of Contents
development and technical problems we experience in the future will not cause significant delays or unanticipated costs, or that such development problems can be solved. We may be unable to reach agreement on favorable terms, or at all, with providers of vectors needed to optimize delivery of our product candidates to target disease cells and we may also experience unanticipated problems or delays in expanding our manufacturing capacity or transferring our manufacturing process to commercial partners, any of which may prevent us from completing our preclinical trials, initiating clinical trials or commercializing our products on a timely or profitable basis, if at all.
Only a few product candidates based on ddRNAi have been tested in either animals or humans, and a number of clinical trials conducted by other companies using other forms of RNAi technologies have not been successful. We may discover that application of ddRNAi does not possess properties required for a therapeutic benefit, such as the ability to continually express shRNAs for the period of time required to be maximally effective or the ability of viral vectors or other technologies to effectively deliver ddRNAi constructs to target cells in therapeutically relevant concentrations. In addition, application of ddRNAi-based products in humans may result in safety issues. We currently have only limited data, and no conclusive evidence, to suggest that we can effectively produce effective therapeutic treatments using our ddRNAi technology.
We are early in our product development efforts and our current product candidates are still in preclinical development. We may not be able to obtain regulatory approvals for the commercialization of our product candidates.
The research, testing, manufacturing, labeling, approval, selling, marketing and distribution of biologics is subject to extensive regulation by the FDA and other regulatory authorities, and these regulations differ from country to country. We do not have any products on the market and are early in our development efforts. All of our ddRNAi product candidates and our silence and replace product candidates are in preclinical development. Our current product candidates are subject to the risks of failure typical for development of biologics. The development and approval process is expensive and can take many years to complete, and its outcome is inherently uncertain. In addition, the outcome of preclinical testing and early clinical trials may not be predictive of the success of later clinical trials, and interim results of a clinical trial do not necessarily predict final results.
We have not submitted a marketing application, or received marketing approval, for any of our product candidates and will not submit any applications for marketing approval for several years. We have limited experience in conducting and managing clinical trials necessary to obtain regulatory approvals, including marketing approval by the FDA. To receive marketing approval, we must, among other things, demonstrate with evidence from clinical trials that the product candidate is both safe and effective for each indication for which approval is sought, and failure can occur in any stage of development. Satisfaction of the marketing approval requirements typically takes several years and the time needed to satisfy them may vary substantially, based on the type, complexity and novelty of the biopharmaceutical product. We cannot predict if or when we might receive regulatory approvals for any of our product candidates currently under development.
The FDA and foreign regulatory authorities also have substantial discretion in the biopharmaceutical product approval process. The numbers, types and sizes of preclinical studies and clinical trials that will be required for regulatory approval varies depending on the product candidate, the disease or condition that the product candidate is designed to address and the regulations applicable to any particular product candidate. Approval policies, regulations or the type and amount of clinical data necessary to gain marketing approval may change during the course of a product candidates clinical development and may vary among jurisdictions, and there may be varying interpretations of data obtained from preclinical studies or clinical trials, any of which may cause delays or limitations in the marketing approval or the decision not to approve an application. Regulatory agencies can delay, limit or deny marketing approval of a product candidate for many reasons, including:
| | the FDA or comparable foreign regulatory authorities may disagree with the design or implementation of any future clinical trials; |
23
Table of Contents
| | we may be unable to demonstrate to the satisfaction of the FDA or comparable foreign regulatory authorities that a product candidate is safe and effective for its proposed indication; |
| | the results of any future clinical trials may not meet the level of statistical or clinical significance required by the FDA or comparable foreign regulatory authorities for approval; |
| | the patients recruited for a particular clinical program may not be sufficiently broad or representative to assure safety and effectiveness in the full population for which we seek approval; |
| | the results of any future clinical trials may not confirm the positive results from earlier preclinical studies or clinical trials; |
| | we may be unable to demonstrate that a product candidates clinical and other benefits outweigh its safety risks; |
| | the FDA or comparable foreign regulatory authorities may disagree with our interpretation of data from preclinical studies or clinical trials; |
| | the data collected from any future clinical trials of our product candidates may not be sufficient to the satisfaction of FDA or comparable foreign regulatory authorities to support the submission of a biologics license application, or BLA, or other comparable submission in foreign jurisdictions or to obtain regulatory approval in the United States or elsewhere; |
| | the FDA or comparable foreign regulatory authorities may only agree to approve a product candidate under conditions that are so restrictive that the product is not commercially viable; |
| | regulatory agencies might not approve or might require changes to our manufacturing processes or facilities; or |
| | regulatory agencies may change their approval policies or adopt new regulations in a manner rendering our clinical data insufficient for approval. |
Any delay in obtaining or failure to obtain required approvals could materially and adversely affect our ability to generate revenue from the particular product candidate, which likely would result in significant harm to our financial position and adversely impact the price of our common stock. Furthermore, any regulatory approval to market a product may be subject to limitations on the indicated uses for which we may market the product. These limitations may limit the size of the market for the product.
We are not permitted to market our product candidates in the United States or in other countries until we receive approval of a BLA from the FDA or marketing approval from applicable regulatory authorities outside the United States. Obtaining approval of a BLA can be a lengthy, expensive and uncertain process. If we fail to obtain FDA approval to market our product candidates, we will be unable to sell our product candidates in the United States, which will significantly impair our ability to generate revenues. In addition, failure to comply with FDA and non-U.S. regulatory requirements may, either before or after product approval, if any, subject us to administrative or judicially imposed sanctions, including:
| | restrictions on our ability to conduct clinical trials, including full or partial clinical holds on ongoing or planned trials; |
| | restrictions on the products, manufacturers or manufacturing process; |
| | warning letters; |
| | civil and criminal penalties; |
| | injunctions; |
| | suspension or withdrawal of regulatory approvals; |
| | product seizures, detentions or import bans; |
24
Table of Contents
| | voluntary or mandatory product recalls and publicity requirements; |
| | total or partial suspension of production; |
| | imposition of restrictions on operations, including costly new manufacturing requirements; and |
| | refusal to approve pending BLAs or supplements to approved BLAs. |
Even if we do receive regulatory approval to market a product candidate, any such approval may be subject to limitations on the indicated uses for which we may market the product. It is possible that none of our existing product candidates or any product candidates we may seek to develop in the future will ever obtain the appropriate regulatory approvals necessary for us or our collaborators to commence product sales. Any delay in obtaining, or an inability to obtain, applicable regulatory approvals would prevent us from commercializing our product candidates, generating revenues and achieving and sustaining profitability.
Because our ddRNAi and silence and replace product candidates are considered gene therapies, it is difficult to predict the time and cost of product candidate development as well as subsequently obtaining regulatory approval.
The clinical trial requirements of the FDA and comparable foreign regulatory authorities and the criteria these regulators use to determine the safety and efficacy of a product candidate vary substantially according to the type, complexity, novelty and intended use and market of such product candidates. The regulatory approval process for novel product candidates such as ours can be more expensive and take longer than for other pharmaceutical product candidates. The FDA and comparable foreign regulatory authorities have relatively limited experience with ddRNAi-based and silence and replace-based therapeutics, which makes it difficult to determine how long it will take or how much it will cost to obtain regulatory approvals for our product candidates in the United States or other countries. We and our current collaborators, or any future collaborators, may never receive approval to market and commercialize any product candidate. Even if we or a collaborator obtain regulatory approval, the approval may be for disease indications or patient populations that are not as broad as we intended or desired and may require labeling that includes significant use or distribution restrictions or safety warnings.
Regulatory requirements governing gene and cell therapy products have changed frequently and may continue to change in the future. Also, before a clinical trial can begin at any institution, that institutions institutional review board, or IRB, and its institutional biosafety committee, or IBC, if it has one, have to review the proposed clinical trial to assess the safety of the trial. In addition, adverse developments in clinical trials of gene therapy products conducted by others may cause the FDA or comparable foreign regulatory bodies to change the requirements for approval of any of our product candidates.
These committees and advisory groups and the new guidelines they promulgate and new requirements they may impose may lengthen the clinical development and regulatory review process, require us to perform additional studies, increase our development costs, lead to changes in regulatory positions and interpretations, delay or prevent approval and commercialization of these product candidates or lead to significant post-approval limitations or restrictions. As we advance our product candidates, we will be required to consult with these regulatory committees and advisory groups, and comply with applicable guidelines and requirements as they may change from time to time. If we fail to do so, we may be required to delay or discontinue development of our product candidates. These additional processes may result in a development, review and approval process that is longer than we otherwise would have expected for our product candidates. Delay or failure to obtain, or unexpected costs in obtaining, the regulatory approval necessary to bring a potential product to market would delay or prevent us from commercializing our product candidates, generating revenues and achieving and sustaining profitability.
25
Table of Contents
Positive results from preclinical studies of our product candidates are not necessarily predictive of the results of our planned clinical trials of our product candidates.
Positive results in preclinical proof of concept and animal studies of our product candidates may not result in positive results in clinical trials in humans. Many companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in clinical trials after achieving positive results in preclinical development or early stage clinical trials, and we cannot be certain that we will not face similar setbacks. These setbacks have been caused by, among other things, preclinical findings made while clinical trials were underway or safety or efficacy observations made in clinical trials, including adverse events. Moreover, preclinical and clinical data are often susceptible to varying interpretations and analyses, and many companies that believed their product candidates performed satisfactorily in preclinical studies and clinical trials nonetheless failed to obtain FDA or other regulatory authority approval. If we fail to produce positive results in our clinical trials of our product candidates, the development timeline and regulatory approval and commercialization prospects for our product candidates, and, correspondingly, our business and financial prospects, would be negatively impacted.
Issues that may impact delivery of our therapeutics to the cell could adversely affect or limit our ability to develop and commercialize product candidates.
Successful clinical development of ddRNAi-based therapeutics and silence and replace-based therapeutics is largely dependent on using the appropriate delivery methodologies, including viral vectors, to obtain therapeutically relevant concentrations of the DNA constructs that express the shRNAs and engineered transgenes in the appropriate target cells. To develop effective product candidates, we will need to license delivery technologies from third parties or develop delivery technologies with research collaborators. Although delivery technologies, including AAV vectors, have been identified and are well defined for specific tissue types, we continue to seek vectors with ideal delivery properties for indications we are pursuing, including OPMD. The tissue tropism and other physical properties of AAV vectors are limited and may not be effective for other product candidates or delivery into a wide array of tissues types. AAV vectors can also trigger immune responses in some patients, and those patients will not derive clinical benefit from administration of a product candidate unless steps are taken to clinically address the issue. If we or our collaborators are not successful in identifying effective delivery methodologies to achieve a therapeutically relevant concentration for our product candidates in the target tissues, we may not succeed in developing marketable products. In addition, if we are unable to reach agreement on favorable terms, or at all, with providers of any effective vectors that we do identify, we may not succeed in completing our clinical trials or commercializing our products on a timely or profitable basis, if at all.
We use AAV vectors as part of our ddRNAi and silence and replace approaches for several indications. As such, we require licenses and the ability to manufacture large quantities of AAV particles under the FDAs current good manufacturing practices, or cGMP, requirements and those of comparable foreign regulatory authorities in order to commercialize a product candidate using an AAV vector.
We may find it difficult to enroll patients in any future clinical trials, and patients could discontinue their participation in our current and any future clinical trials, which could delay or prevent our current and any future clinical trials of our product candidates and make those trials more expensive to undertake.
Identifying and qualifying patients to participate in current and any future clinical trials of our product candidates is critical to our success. The timing of our clinical trials depends on the speed at which we can recruit patients to participate in testing our product candidates. Patients may be unwilling to participate in any future clinical trials because of negative publicity from adverse events in the biotechnology, RNAi or gene therapy industries. Patients may be unavailable for other reasons, including competitive clinical trials for similar patient populations, and the timeline for recruiting patients, conducting trials and obtaining regulatory approval of potential products may be delayed. If we have difficulty enrolling a sufficient number of patients to conduct any future clinical trials as planned, we may need to delay, limit or discontinue those clinical trials. Clinical trial delays could result in increased costs, slower product development, setbacks in testing the safety and effectiveness of our technology or discontinuation of the clinical trials altogether.
26
Table of Contents
We may not be able to identify, recruit and enroll a sufficient number of patients, or those with required or desired characteristics to achieve diversity in a trial, to complete any future clinical trials in a timely manner. Patient enrollment is affected by factors including:
| | finding and diagnosing patients; |
| | severity of the disease under investigation; |
| | design of the clinical trial protocol; |
| | size and nature of the patient population; |
| | eligibility criteria for the trial in question; |
| | perceived risks and benefits of the product candidate under study; |
| | proximity and availability of clinical trial sites for prospective patients; |
| | availability of competing therapies and clinical trials; |
| | clinicians and patients perceptions of the potential advantages of the product being studied in relation to other available therapies, including any new products that may be approved for the indications we are investigating; |
| | patient referral practices of physicians; |
| | ability to monitor patients adequately during and after treatment; and |
| | the COVID-19 pandemic. |
We or any potential collaborators plan to seek initial marketing approval for our product candidates in the United States, Canada and Europe. We may not be able to initiate or continue any future clinical trials in a timely manner if we cannot enroll a sufficient number of eligible patients to participate in the clinical trials required by the FDA or comparable foreign regulatory agencies. Our ability to successfully initiate, enroll and complete a clinical trial in any foreign country is subject to numerous risks unique to conducting business in foreign countries, including:
| | difficulty in establishing or managing relationships with contract research organizations, or CROs, clinical sites and physicians; |
| | different standards for the conduct of clinical trials; |
| | our inability to locate and engage qualified local consultants, physicians and partners; and |
| | the potential burden of complying with a variety of foreign laws, medical standards and regulatory requirements, including the regulation of biopharmaceutical and biotechnology products and treatments. |
In addition, patients enrolled in current and any future clinical trials may discontinue their participation at any time during the trial as a result of a number of factors, including experiencing adverse events, which may or may not be judged related to our product candidates under evaluation. The discontinuation of patients in any one of our trials may cause us to delay or discontinue our clinical trial, or cause the results from that trial not to be positive or sufficient to support either partnering with a pharmaceutical or biotechnology company to further develop and commercialize the product candidate or filing for regulatory approval of the product candidate.
We may encounter substantial delays in any future clinical trials or we may fail to demonstrate safety and efficacy to the satisfaction of applicable regulatory authorities.
Before obtaining marketing approval from regulatory authorities for the sale of any of our product candidates, we must conduct extensive clinical trials to demonstrate the safety and efficacy of the product
27
Table of Contents
candidates in humans. Clinical trials are expensive and time-consuming, and their outcome is uncertain. We may experience delays in initiating and completing any clinical trials that we are conducting or intend to conduct as a result of the COVID-19 pandemic, and we do not know whether our ongoing or planned clinical trials will begin or progress on schedule, need to be redesigned, enroll patients on time or be completed on schedule, or at all. We cannot guarantee that any clinical trials will be initiated or conducted as planned or completed on schedule, if at all. A failure of one or more clinical trials can occur at any stage of testing. Events that may prevent successful or timely completion of clinical development include:
| | inability to generate sufficient preclinical, toxicology or other data to support the initiation of human clinical trials; |
| | delays in reaching consensus with regulatory agencies on trial design; |
| | identifying, recruiting and training suitable clinical investigators; |
| | delays in reaching agreement on acceptable terms with prospective CROs and clinical trial sites; |
| | delays in obtaining required IRB or IBC approval at each clinical trial site; |
| | delays in recruiting suitable patients to participate in our clinical trials, including as a result of the current COVID-19 pandemic; |
| | imposition of a clinical hold by regulatory agencies, including after an inspection of our clinical trial operations or trial sites; |
| | failure by our CROs, other third parties or us to adhere to clinical trial requirements; |
| | failure to perform in accordance with the FDAs good clinical practices, or GCP, or applicable regulatory requirements in other countries; |
| | inability to manufacture, test, release, import or export for use sufficient quantities of our product candidates for use in clinical trials; |
| | failure to manufacture our product candidate in accordance with cGMP requirements or applicable regulatory guidelines in other countries; |
| | delays in the testing, validation and manufacturing of our product candidates; |
| | delays in the delivery of our product candidates to the clinical trial sites; |
| | delays in having patients complete participation in a trial or return for post-treatment follow-up; |
| | clinical trial sites dropping out of a trial; |
| | occurrence of serious adverse events associated with the product candidate that are viewed to outweigh its potential benefits; |
| | changes in regulatory requirements and guidance that require amending or submitting new clinical trial protocols; |
| | clinical trials of our product candidates may produce negative or inconclusive results, and we may decide, or regulators may require us, to conduct additional clinical trials or discontinue product development programs; or |
| | the severity, duration and impact of the COVID-19 pandemic. |
Further, a clinical trial may be suspended or discontinued by us, our collaborators, the IRBs or the IBCs at the sites in which such trials are being conducted, the data safety monitoring board, or DSMB, for such trial, or by the FDA or comparable foreign regulatory authorities due to a number of factors, including the imposition of a clinical hold or termination of a trial due to failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, unforeseen safety issues or adverse side effects of our product candidate,
28
Table of Contents
or a product candidate from another company that shares similar properties, failure to demonstrate adequate benefit from using a product candidate, changes in governmental regulations or administrative actions or lack of adequate funding to continue the clinical trial. If we experience discontinuation of, or delays in the completion of, any clinical trial of our product candidates, the commercial prospects of our product candidates may be harmed, and our ability to generate product revenues from any of these product candidates may be eliminated or delayed. Furthermore, many of the factors that lead to a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of regulatory approval of our product candidates.
In addition, if we or our third-party collaborators make significant manufacturing or formulation changes to our product candidates, we or they may need to conduct additional studies to bridge the modified product candidates to earlier versions to ensure comparability, safety and efficacy of the two different product candidates.
Any inability to successfully complete preclinical and clinical development could result in additional costs to us or impair our ability to commercialize our programs and product candidates. Clinical trial delays could also shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to bring products to market before we do, which could impair our ability to successfully commercialize our product candidates.
If the results of our current or any future clinical trials are inconclusive or if there are safety concerns or adverse events associated with our product candidates, we may:
| | fail to obtain, or be delayed in obtaining, marketing approval for our product candidates; |
| | obtain approval for indications or patient populations that are not as broad as intended or desired; |
| | obtain approval with labeling that includes significant use or distribution restrictions or safety warnings; |
| | need to change the way the product is administered; |
| | be required to perform additional clinical trials to support approval or be subject to additional post-marketing testing requirements; |
| | have regulatory authorities withdraw their marketing approval of the product after granting it; |
| | have regulatory authorities impose restrictions on distribution of the product in the form of a risk evaluation and mitigation strategy, or REMS, or modified REMS, that limit our ability to commercialize the product; |
| | be subject to the addition of labeling statements, such as warnings or contraindications; |
| | be sued and held liable for harm caused to patients; or |
| | experience damage to our reputation. |
Any of these events could prevent us from achieving or maintaining market acceptance of our product candidates and impair our ability to commercialize our product candidates.
In addition, principal investigators for our clinical trials may serve as scientific advisors or consultants to us from time to time and may receive cash or equity compensation in connection with such services. If these relationships and any related compensation result in perceived or actual conflicts of interest, or the FDA concludes that the financial relationship may have affected the interpretation of any particular study, the integrity of the data generated at the applicable clinical trial site may be questioned and the utility of the clinical trial itself may be jeopardized, which could result in the delay or rejection of any BLA we submit to the FDA or any comparable foreign regulating authorities. Any such delay or rejection could prevent us from commercializing our product candidates.
29
Table of Contents
Our product candidates and the process for administering our product candidates may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval, limit the commercial profile of an approved label, or result in significant negative consequences following any potential marketing approval.
Treatment with our product candidates may produce undesirable side effects or adverse reactions or events. For example, the AAV vector and related capsid protein, which we are currently using to deliver many of our ddRNAi and silence and replace product candidates, could cause adverse immunological side effects due to preexisting and/or recall responses to the naturally occurring virus from which the vector is engineered, or to the DNA construct product itself. These responses may also interfere with therapeutic efficacy if not identified and managed optimally. Preexisting immune responses to AAV manifesting as neutralizing antibodies are common within the general population and may be a limitation to the enrollment of patients in gene therapy clinical trials using AAV vectors, the successful use of AAV vectors in gene therapy clinical trials and the market acceptance of product candidates, if approved, that are delivered using AAV vectors. Patients with neutralizing antibodies to AAV will not derive clinical benefit from administration of such a product candidate unless steps are taken to clinically address the issue and those treatments themselves may cause adverse effects. In previous clinical trials undertaken by other companies involving systemic administration of AAV viral vectors for gene therapy, some subjects experienced adverse events, including the development of a negative T cell response against the AAV capsid protein. If our vectors cause similar adverse events, we may be required to delay or discontinue further clinical development of our product candidates. It is also possible that we may discover new adverse events related to AAV or other vectors, which could potentially enhance the risk to patients who use our product candidates delivered with that vector. Additionally, the procedure used to administer the treatment could result in undesirable health effects.
If any such adverse events occur, our current and any future clinical trials could be suspended or discontinued and the FDA or comparable foreign regulatory authorities could order us to cease further development or deny approval of our product candidates for any or all targeted indications. The product-related side effects could affect patient recruitment or the ability of enrolled patients to complete the trial. If we elect or are required to delay, suspend or discontinue any clinical trial of any of our product candidates, the commercial prospects of such product candidates will be harmed and our ability to generate product revenues from any of these product candidates will be delayed or eliminated. Any of these occurrences may harm our business, financial condition and prospects significantly.
Additionally, if any of our biopharmaceutical product candidates receive marketing approval, the FDA could require us to adopt a REMS to ensure that the benefits of the product outweigh its risks, which may include, among other things, a medication guide outlining the risks of gene therapies for distribution to patients and a communication plan to patients and healthcare practitioners. Other elements to assure safe use in a mandated REMS could include, but are not limited to, restrictions upon distribution and prescribing, additional prescriber training, establishment of patient registries and other measures that could limit commercialization of the product. Comparable foreign regulating authorities might require adoption of measures similar to a REMS. Furthermore, if we or others later identify undesirable side effects caused by our product candidate, a number of potentially significant negative consequences could result, including:
| | regulatory authorities may withdraw approvals of such product candidate; |
| | regulatory authorities may require additional warnings on the label; |
| | we may be required to create a medication guide outlining the risks of such side effects for distribution to patients; |
| | we may be required to change the way a product candidate is administered or conduct additional clinical trials; |
| | we could be sued and held liable for harm caused to patients; and |
| | our reputation may suffer. |
30
Table of Contents
Any of these events could prevent us from achieving or maintaining market acceptance of our product candidates and could significantly harm our business, prospects, financial condition and results of operations.
If we are unable to successfully develop related diagnostics for our therapeutic product candidates, or experience significant delays in doing so, we may not achieve marketing approval or realize the full commercial potential of our therapeutic product candidates.
We may develop related diagnostics for some of our therapeutic product candidates. Such related diagnostics are subject to regulation by the FDA and typically to comparable foreign regulatory authorities as medical devices and typically require separate regulatory approval or clearance prior to commercialization. Marketing approval or clearance of the diagnostic will require sufficient data to support its safety and efficacy. In addition, at least in some cases, the FDA and comparable foreign regulatory authorities may require the development and regulatory approval or clearance of a related diagnostic as a condition to approving our therapeutic product candidates. While we have some limited experience in developing diagnostics, we plan to rely in large part on third parties to perform these functions. We may seek to enter into arrangements with one or more third parties to create a related diagnostic for use with our current or future product candidates.
If we or any third parties that we engage to assist us are unable to successfully develop or obtain marketing approval or clearance for related diagnostics for our therapeutic product candidates, or experience delays in doing so:
| | the development of relevant product candidates may be delayed or impaired altogether if we are unable to appropriately select patients for enrollment in our clinical trials; |
| | our relevant therapeutic product candidate may not receive marketing approval if its effective use depends on a related diagnostic in the regulatory authoritys judgment; and |
| | we may not realize the full commercial potential of any therapeutic product candidates that receive marketing approval if, among other reasons, we are unable to appropriately identify patients with the specific genetic alterations targeted by our therapeutic product candidates. |
If any of these events were to occur, our business would be harmed.
Even if we complete the necessary preclinical studies and clinical trials, we cannot predict when or if we will obtain regulatory approval to commercialize a product candidate or the approval may be for a more narrow indication than we expect.
We cannot commercialize a product until the appropriate regulatory authorities have reviewed and approved the product candidate. Even if our product candidates demonstrate safety and efficacy in clinical trials, the regulatory agencies may not complete their review processes in a timely manner, or we may not be able to obtain regulatory approval. Additional delays may result if an FDA Advisory Committee or comparable foreign regulatory authority recommends non-approval or restrictions on approval. In addition, we may experience delays or rejections based upon additional government regulation from future legislation or administrative action, or changes in regulatory agency policy during the period of product development, clinical trials and the review process. Regulatory agencies may also approve a treatment candidate for fewer or more limited indications than requested, may not approve the price we intend to charge for our product candidate, may limit our ability to promote the product, may impose significant limitations upon the approval of the product, including, but not limited to, narrow indications, significant warnings, precautions or contraindications with respect to conditions of use, or may grant approval subject to the performance of post-marketing studies. In addition, regulatory agencies may not approve the labeling claims that are necessary or desirable for the successful commercialization of our product candidates. The FDA or comparable foreign regulatory authorities may impose a REMS or other conditions upon our approval that limit our ability to commercialize the product candidate.
31
Table of Contents
Inadequate funding for the FDA and other government agencies could hinder their ability to hire and retain key leadership and other personnel, prevent new products and services from being developed or commercialized in a timely manner or otherwise prevent those agencies from performing normal business functions on which the operation of our business may rely, which could negatively impact our business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory, and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies on which our operations may rely, including those that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, in recent years, including in 2018 and 2019, the U.S. government shut down several times and certain regulatory agencies, such as the FDA, had to furlough critical employees and stop critical activities. Separately, in response to the COVID-19 pandemic, the FDA postponed most inspections of foreign manufacturing facilities and products, postponed routine surveillance inspections of domestic manufacturing facilities and is conducting only teleconference meetings. Regulatory authorities outside the United States may adopt similar restrictions or other policy measures in response to the COVID-19 pandemic. If a prolonged government shutdown occurs, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business. Further, upon completion of this offering and in our operations as a public company, future government shutdowns could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
Even if we obtain regulatory approval for a product candidate, our products will remain subject to regulatory scrutiny.
Even if we obtain regulatory approval in a jurisdiction, the regulatory authority may still impose significant restrictions on the indicated uses or marketing of our product candidates, or impose ongoing requirements for potentially costly post-approval studies or post-market surveillance. For example, the holder of an approved BLA is obligated to monitor and report to the FDA adverse events and any failure of a product to meet the specifications in the BLA. FDA guidance advises that patients treated with some types of gene therapy undergo follow-up observations for potential adverse events for as long as 15 years. The holder of an approved BLA must also submit new or supplemental applications and obtain FDA approval for certain changes to the approved product, product labeling or manufacturing process. Advertising and promotional materials must comply with FDA rules and are subject to FDA review, in addition to other potentially applicable foreign, federal and state laws.
In addition, product manufacturers and their establishments, products and applications are subject to payment of user fees/or and continual review and periodic inspections by the FDA and comparable foreign regulatory authorities for compliance with cGMP and comparable foreign requirements, and adherence to commitments made in the BLA. If we or a regulatory agency discover previously unknown problems with a product such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, a regulatory agency may impose restrictions relative to that product or the manufacturing facility, including requiring recall or withdrawal of the product from the market or suspension of manufacturing.
If we fail to comply with applicable regulatory requirements following approval of any of our product candidates, a regulatory agency may:
| | issue a warning letter asserting that we are in violation of the law; |
| | seek an injunction or impose civil or criminal penalties or monetary fines; |
32
Table of Contents
| | suspend or withdraw regulatory approval; |
| | suspend any ongoing clinical trials; |
| | refuse to permit government reimbursement of our product by government-sponsored third-party payers; |
| | refuse to approve a pending BLA or supplements to a BLA submitted by us for other indications or new product candidates; |
| | seize our product; or |
| | refuse to allow us to enter into or continue supply contracts, including government contracts. |
Any government investigation of alleged violations of law could require us to expend significant time and resources in response and could generate negative publicity. The occurrence of any event or penalty described above may inhibit our ability to commercialize our product candidates and generate revenues.
Even if we obtain and maintain approval for our product candidates from the FDA, we may never obtain approval for our product candidates outside of the United States, which would limit our market opportunities and adversely affect our business.
Approval of a product candidate in the United States by the FDA does not ensure approval of such product candidate by regulatory authorities in other countries or jurisdictions, and approval by one foreign regulatory authority does not ensure approval by regulatory authorities in other foreign countries or by the FDA. Sales of our product candidates outside of the United States will be subject to foreign regulatory requirements governing clinical trials and marketing approval. Even if the FDA grants marketing approval for a product candidate, comparable regulatory authorities of foreign countries must also approve the manufacturing and marketing of the product candidates in those countries. Approval procedures vary among jurisdictions and can involve requirements and administrative review periods different from, and greater than, those in the United States, including additional preclinical studies or clinical trials. In many countries outside the United States, a product candidate must be approved for reimbursement before it can be approved for sale in that country. In some cases, the price that we intend to charge for our products, if approved, is also subject to approval. Even if a product candidate is approved, the FDA or comparable regulatory authorities in other countries, as the case may be, may limit the indications for which the product may be marketed, require extensive warnings on the product labeling or require expensive and time-consuming clinical trials or reporting as conditions of approval. Regulatory authorities in countries outside of the United States also have requirements for approval of product candidates with which we must comply prior to marketing in those countries. Obtaining foreign regulatory approvals and compliance with foreign regulatory requirements could result in significant delays, difficulties and costs for us and could delay or prevent the introduction of our product candidates in certain countries.
Further, clinical trials conducted in one country may not be accepted by regulatory authorities in other countries. Regulatory approval of a product candidate in one country does not ensure approval in any other country, but a failure or delay in obtaining regulatory approval in one country may have a negative effect on the regulatory approval process in other countries. Also, regulatory approval for any of our product candidates may be withdrawn based on adverse events reported or regulatory decisions made in other countries. If we fail to comply with the regulatory requirements in international markets and/or fail to receive applicable marketing approvals, our target market will be reduced, our ability to realize the full market potential of our product candidates will be compromised and our business may be adversely affected.
Our future prospects may also depend on our collaborators ability to successfully develop a pipeline of additional product candidates, and we and our collaborators may not be successful in efforts to use our platform technologies to identify or discover additional product candidates.
The success of our business depends primarily upon our ability to identify, develop and commercialize products based on our platform technology. We do not have any products on the market and are early in our
33
Table of Contents
development efforts. All of our product candidates are in preclinical development. Our product candidates derived from our platform technology may not successfully complete investigational new drug, or IND, enabling studies, and our research programs may fail to identify other potential product candidates for clinical development for a number of reasons. Our and our collaborators research methodology may be unsuccessful in identifying potential product candidates, our potential product candidates may not demonstrate the necessary preclinical outcomes to progress to clinical studies, or our product candidates may be shown to have harmful side effects or may have other characteristics that may make the products unmarketable or unlikely to receive marketing approval.
If any of these events occur, we may be forced to discontinue our development efforts for a program or programs. Research programs to identify new product candidates require substantial technical, financial and human resources. We may focus our efforts and resources on potential programs or product candidates that ultimately prove to be unsuccessful.
We may not be able to obtain orphan drug exclusivity, where relevant, in all markets for our product candidates.
Of our current pipeline product candidates, only our silence and replace therapeutic for the treatment of OPMD has been designated with orphan drug status. In January 2018, the FDA granted such designation after our candidate for the treatment of OPMD had been designated an orphan drug in January 2017 by the European Commission. Regulatory authorities in some jurisdictions, including the United States, may designate drugs or biological products for relatively small patient populations as orphan drugs. Under the Orphan Drug Act, the FDA may designate a product as an orphan drug if it is a product intended to treat a rare disease or condition, which is generally defined as any disease or condition which affects less than 200,000 individuals in the United States. The FDA may also designate a product as an orphan drug if it is intended to treat a disease or condition which affects more than 200,000 individuals in the United States and there is no reasonable expectation that the cost of developing and making a drug or biological product available in the United States for this type of disease or condition will be recovered from sales of the product candidate. Generally, if a product with an orphan drug designation subsequently receives the first marketing approval for the indication for which it has such designation, the product is entitled to a period of marketing exclusivity, which precludes the FDA from approving another marketing application for the same drug for such indication for that time period. The applicable period is seven years in the United States. Orphan drug exclusivity may be lost if the FDA or the European Medicines Agency, or the EMA, determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the drug to meet the needs of patients with the rare disease or condition. While there is no guarantee, FDA orphan drug designation may provide a range of benefits, including a potential fast track process for clinical regulatory approval, potential tax credits for qualified clinical trials and an exemption from FDA application user fees.
Even if we obtain orphan drug exclusivity for a product in the United States or for additional products in the European Union, that exclusivity may not effectively protect the product from competition because different drugs can be approved for the same condition, and the same drug could be approved for a different condition. Even after an orphan drug is approved, the FDA can subsequently approve the same drug, made by a competitor, for the same condition if the FDA concludes that the competitive product is clinically superior in that it is shown to be safer, more effective or makes a major contribution to patient care. In the European Union, the EMA can approve a competitive product if the orphan drug no longer meets the criteria for orphan designation (including sufficient profitability), if the competitive product is safer, more effective or otherwise clinically superior, or if the orphan drug cannot be supplied in sufficient quantities.
34
Table of Contents
Risks Related to Our Reliance on Third Parties
Our prospects for successful development and commercialization of our products are dependent to varying degrees upon the research, development, commercialization, and marketing efforts of any potential collaborators.
We rely on third parties for certain aspects of the research, development, commercialization and marketing of our current and any future product candidates. Other than as provided for in our collaboration agreements, we have no control over the resources, time and effort that our collaborators may devote to the development of product candidates. We are dependent on our collaborators to conduct some aspects of the research and development of each of our product candidates, and expect to need access to them to facilitate and/or to complete the regulatory process. We will likely rely on a pharmaceutical company for the successful marketing and commercialization of any such product candidates for which they/we receive approval, if any. There can be no guarantee at this stage that we will conclude a partnership with such a company on favorable terms, or at all, nor even if we do so, that success will be achieved.
Our ability to recognize revenues from successful potential collaborations may be impaired by multiple factors including:
| | a collaborator may shift its priorities and resources away from our programs due to a change in business strategies, or a merger, acquisition, sale or downsizing of its company or business unit; |
| | a collaborator may cease development in an area that is the subject of a collaboration agreement; |
| | a collaborator may change the success criteria for a particular program or product candidate in development, thereby delaying or ceasing development of such program or product candidate in development; |
| | a collaborator with development or commercialization obligations may not commit sufficient financial or human resources to the development, marketing, distribution or sale of a product, or may otherwise fail in development or commercialization efforts; |
| | a collaborator with manufacturing responsibilities may encounter regulatory, resource or quality issues and be unable to meet demand requirement; |
| | a collaborator could independently develop, or develop with unrelated parties, products that compete directly or indirectly with our product candidates; |
| | a collaborator may exercise its rights under the agreement to discontinue our collaboration; |
| | a dispute may arise between us and a collaborator concerning the development or commercialization of a product candidate, resulting in a delay in milestones, royalty payments, or discontinuation of a program and possibly resulting in costly litigation or arbitration that may divert management attention and resources; |
| | a collaborator may not adequately protect the intellectual property rights associated with a product candidate; |
| | a collaborator may use our proprietary information or intellectual property in such a way as to expose us actual or threatened litigation from a third party, patent office proceedings or other risks that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential liability; and |
| | a collaborator may own or co-own, or have a license to use, intellectual property rights associated with a product candidate that results from our collaborating with them, and in such cases, we would not have the exclusive right to commercialize such intellectual property rights. |
If our potential collaborators do not perform in the manner we expect or fulfill their responsibilities in a timely manner, or at all, the development, regulatory and commercialization process could be delayed or
35
Table of Contents
discontinued or otherwise be unsuccessful. Conflicts between us and our collaborators may arise. In the event of discontinuation of one or more of our collaboration agreements, it may become necessary for us to assume the responsibility for any such product candidates at our own expense or seek new collaborators. In that event, we likely would be required to limit the size and scope of one or more of our independent programs or increase our expenditures and seek additional funding, which may not be available on acceptable terms or at all, and our business may be harmed.
We rely on third parties to conduct our preclinical studies and clinical trials.
We do not have the ability to conduct all aspects of our preclinical testing or any future clinical trials ourselves. We are dependent on third parties to conduct the preclinical studies for our product candidates and will depend on third parties to conduct any future clinical trials for our product candidates, and therefore the timing of the initiation and completion of these trials and studies is reliant on third parties and may occur at times substantially different from our estimates or expectations.
In the case of clinical trials, we expect to rely on CROs and third-party collaborators to conduct any future clinical trials in accordance with our clinical protocols and regulatory requirements. We expect our CROs, investigators and third-party collaborators will play a significant role in the conduct of these trials and subsequent collection and analysis of data. There is no guarantee that any CROs, investigators or the other third-party collaborators on which we rely for administration and conduct of our future clinical trials will devote adequate time and resources to such trials or perform as contractually required. If any of these third parties fails to meet expected deadlines, fails to adhere to our clinical protocols, fails to meet regulatory requirements or otherwise performs in a substandard manner, any future clinical trials may be extended, delayed or terminated. If our current or any of our future clinical trial sites terminates for any reason, we may lose all of the information on subjects enrolled in any such clinical trials.
If we cannot contract with acceptable third parties on commercially reasonable terms, or if these third parties do not carry out their contractual duties, satisfy legal and regulatory requirements for the conduct of preclinical studies or clinical trials or meet expected deadlines, our clinical development programs could be delayed or discontinued.
In all events, we are responsible for ensuring that each of our preclinical studies, and our future clinical trials are conducted in accordance with the general investigational plan and protocols for the study or trial. The FDA requires clinical trials to be conducted in accordance with current GCP, including for conducting, recording and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights and confidentiality of clinical trial participants are protected. Our reliance on third parties that we do not control does not relieve us of these responsibilities and requirements, and any failure to satisfy these responsibilities and requirements, whether caused by us or by third parties upon whom we rely, could have a material adverse effect on our business, financial condition, results of operations and prospects.
Because we rely on third-party manufacturing and supply partners, our supply of research and development, preclinical and clinical development materials may become limited or interrupted or may not be of satisfactory quantity or quality.
We rely on third-party supply and manufacturing partners to manufacture and supply the materials for our research and development and preclinical and clinical study supplies. We do not own manufacturing facilities or supply sources for such materials.
There can be no assurance that our supply of research and development, preclinical and clinical development biologics and other materials will not be limited, interrupted or restricted in certain geographic regions, be of satisfactory quality or continue to be available at acceptable prices. Replacement of a third-party manufacturer could require significant effort, cost and expertise because there may be a limited number of qualified replacements.
36
Table of Contents
The manufacturing process for a product candidate is subject to FDA and foreign regulatory authority review. Suppliers and manufacturers must meet applicable manufacturing requirements and undergo rigorous facility and process validation tests required by regulatory authorities in order to comply with regulatory standards, such as cGMP. In the event that any of our suppliers or manufacturers fails to comply with such requirements or to perform its obligations to us in relation to quality, timing or otherwise, or if our supply of components or other materials becomes limited or interrupted for other reasons, we may be forced to manufacture the materials ourselves or enter into an agreement with another third party, which would be costly and delay any future clinical trials.
In some cases, the technical skills or technology required to manufacture our product candidates may be unique or proprietary to the original manufacturer and we may have difficulty, or there may be contractual restrictions prohibiting us from, transferring such skills or technology to another third party. These factors increase our reliance on our manufacturers and may require us to obtain a license from a manufacturer in order to have another third-party manufacture our product candidates. If we are required to change manufacturers for any reason, we will be required to verify that the new manufacturer maintains facilities and procedures that comply with quality standards and with all applicable regulations and guidelines of the FDA and comparable foreign regulatory authorities. The delays and costs associated with the verification of a new manufacturer could increase our costs and delay the development of our product candidates.
We expect to continue to rely on third-party manufacturers for preclinical and clinical grade product candidates and if we receive regulatory approval for any product candidate. To the extent that we have existing, or enter into future, manufacturing arrangements with third parties, we will depend on these third parties to perform their obligations in a timely manner consistent with contractual and regulatory requirements, including those related to quality control and assurance. If we are unable to obtain or maintain third-party manufacturing for product candidates, or to do so on commercially reasonable terms, we may not be able to develop and commercialize our product candidates successfully. Our or a third partys failure to execute on our manufacturing requirements could adversely affect our business in a number of ways, including:
| | an inability to conduct necessary preclinical studies to progress our product candidates to clinical trials; |
| | an inability to initiate or continue any future clinical trials of product candidates under development; |
| | delay in submitting regulatory applications, or receiving regulatory approvals, for product candidates; |
| | loss of the cooperation of a collaborator; |
| | subjecting our product candidates to additional inspections by regulatory authorities; |
| | requirements to cease distribution or to recall batches of our product candidates; and |
| | in the event of approval to market and commercialize a product candidate, an inability to meet commercial demands for our products. |
Our third-party manufacturers may be subject to damage or interruption from, among other things, fire, natural or man-made disaster, power loss, telecommunications failure, unauthorized entry, computer viruses, denial-of-service attacks, acts of terrorism, human error, vandalism or sabotage, financial insolvency, bankruptcy and similar events. These risks include the impact of the current COVID-19 pandemic. The extent to which COVID-19 may impact our third party partners will depend on future developments, which are highly uncertain and cannot be predicted, including new information which may emerge concerning the severity of the coronavirus, the geographic spread of the disease, the duration of the outbreak, and the actions to contain the coronavirus or treat its impact, including travel and other social distancing restrictions in the United States and other countries, among others.
37
Table of Contents
We and our licensees or collaborators may disagree over our right to receive payments under any potential collaboration agreements with them, potentially resulting in costly litigation and loss of reputation.
Our ability to receive payments under any out-license and collaboration agreements depends on our ability to clearly delineate our rights under those agreements. We have licensed and may license additional portions of our intellectual property to our collaborators with the intent that our collaborators will develop product candidates based on our ddRNAi or other technology to address specific conditions. However, a collaborator may use our intellectual property without our permission, dispute our ownership of intellectual property rights, or argue that our intellectual property does not cover, or add value to, any product candidates they develop. If a dispute arises, it may result in costly patent office procedures and litigation, and our collaborator may refuse to pay us while the dispute is ongoing. Furthermore, regardless of any resort to legal action, a dispute with a collaborator over intellectual property rights may damage our relationship with that collaborator and may also harm our reputation in the industry. Even if we are entitled to payments from our collaborators, we may not actually receive these payments, or we may experience difficulties in collecting the payments to which we believe we are entitled. After our collaborators launch commercial products containing our licensed traits, we will need to rely on the good faith of our collaborators to report to us the sales they earn from these products and to accurately calculate the payments we are entitled to, a process that will involve complicated and difficult calculations. Although we seek to address these concerns in our license and collaboration agreements by reserving our right to audit financial records, such provisions may not be effective.
We have only limited experience in regulatory affairs and intend to rely on consultants and other third parties for regulatory matters, which may affect our ability or the time we require to obtain necessary regulatory approvals.
We have limited experience in filing and prosecuting the applications necessary to gain regulatory approvals for gene therapy or ddRNAi product candidates. Moreover, the product candidates that are likely to result from our development programs are based on novel technologies that have not been extensively tested in humans. The regulatory requirements governing these types of product candidates may be less well defined or more rigorous than for conventional products. As a result, we may experience a longer regulatory process in connection with obtaining regulatory approvals of any products that we develop. We intend to rely on independent consultants for purposes of our regulatory compliance and product development and approvals in the United States and elsewhere. Any failure by our consultants to properly advise us regarding, or properly perform tasks related to, regulatory compliance requirements could compromise our ability to develop and seek regulatory approval of our product candidates.
Our reliance on third parties requires us to share our trade secrets, which increases the possibility that a competitor will discover them or that our trade secrets will be misappropriated or disclosed.
Because we rely on third parties to manufacture our product candidates, and because we collaborate with various organizations and academic institutions on the advancement of our technology and product candidates, we may, at times, share trade secrets with them. We seek to protect our proprietary technology in part by entering into confidentiality agreements and, if applicable, material transfer agreements, collaborative research agreements, consulting agreements or other similar agreements with our collaborators, advisors, employees and consultants prior to beginning research or disclosing proprietary information. These agreements typically limit the rights of the third parties to use or disclose our confidential information, such as trade secrets. Despite these contractual provisions, the need to share trade secrets and other confidential information increases the risk that such trade secrets become known by potential competitors, are inadvertently incorporated into the technology of others, or are disclosed or used in violation of these agreements. Given that our proprietary position is based, in part, on our know-how and trade secrets, discovery by a third party of our trade secrets or other unauthorized use or disclosure would impair our intellectual property rights and protections in our product candidates.
In addition, these agreements typically restrict the ability of our collaborators, advisors, employees and consultants to publish data potentially relating to our trade secrets. Our academic collaborators typically have
38
Table of Contents
rights to publish data, provided that we are notified in advance and may delay publication for a specified time in order to secure our intellectual property rights arising from the collaboration. In other cases, publication rights are controlled exclusively by us or we may share these rights with other parties. Despite our efforts to protect our trade secrets, our competitors may discover our trade secrets, either through breach of these agreements, independent development or publication of information including our trade secrets in cases where we do not have proprietary or otherwise protected rights at the time of publication.
Risks Related to Commercialization of Our Product Candidates
We have not entered into agreements with any third-party manufacturers to support commercialization of our product candidates.
We have not yet secured manufacturing capabilities for commercial quantities of our product candidates or established facilities in the desired locations to support commercialization of our product candidates. We intend to rely on third-party manufacturers for commercialization, but have not entered into any agreements with such manufacturers to support our product candidates currently in development. We may be unable to negotiate agreements with third-party manufacturers to support our commercialization activities on commercially reasonable terms.
We may encounter technical or scientific issues related to manufacturing or development that we may be unable to resolve in a timely manner or with available funds. Currently, we do not have the capacity to manufacture our product candidates on a commercial scale. In addition, our product candidates are novel, and only one manufacturer currently has experience producing our product candidates on a large scale. If we are unable to engage manufacturing partners to produce our product candidates on a larger scale on reasonable terms, our commercialization efforts will be harmed.
Even if we timely develop a manufacturing process and successfully transfer it to the third-party manufacturers of our product candidates, if such third-party manufacturers are unable to produce the necessary quantities of our product candidates, or do so in compliance with cGMP or with pertinent foreign regulatory requirements, and within our planned time frame and cost parameters, the development and sales of our product candidates, if approved, may be impaired. In some jurisdictions, approval of the manufacturer may be required. There is no guarantee such approval can be obtained.
If we are unable to enter into agreements with third parties to commercialize our product candidates or establish sales and marketing capabilities to market and sell our product candidates, we may be unable to generate any revenues.
We currently have no sales and marketing organization and have no experience selling and marketing pharmaceutical products. To successfully commercialize any product candidates that may be approved, we will need to develop these capabilities, either through our relationships with collaborators or our own. We may seek to enter into collaborations with other entities to utilize their marketing and distribution capabilities, but we may be unable to enter into marketing agreements on favorable terms, if at all. If our future collaborators do not commit sufficient resources to commercialize our future products, if any, and we are unable to develop the necessary marketing capabilities on our own, we will be unable to generate sufficient product revenue to sustain our business. The establishment and development of our own sales force or the establishment of a contract sales force to market any products we may develop will be expensive and time-consuming and could delay any product launch. Moreover, we cannot be certain that we will be able to successfully develop this capability. We will be competing with many companies that currently have extensive and well-funded marketing and sales operations to recruit, hire, train and retain marketing and sales personnel. We also face competition in our search for third parties to assist us with the sales and marketing efforts of our product candidates. Without an internal team or the support of a third party to perform marketing and sales functions, we may be unable to compete successfully against these more established companies.
39
Table of Contents
Physicians, patients, third-party payers or others in the medical community may not be receptive to our product candidates, and we may not generate any future revenue from the sale or licensing of our product candidates.
Even if we obtain approval for a product candidate, we may not generate or sustain revenue from sales of the product if the product cannot be sold at a competitive cost or if it fails to achieve market acceptance by physicians, patients, third-party payers or others in the medical community. These market participants may be hesitant to adopt a novel treatment based on ddRNAi or silence and replace technology, and we may not be able to convince the medical community and third-party payers to accept and use, or to provide favorable reimbursement for, any product candidates developed by us or our existing or future collaborators. Market acceptance of our product candidates will depend on, among other factors:
| | the safety and efficacy of our product candidates; |
| | our ability to offer our products for sale at competitive prices; |
| | the relative convenience and ease of administration of our product candidates; |
| | the prevalence and severity of any adverse side effects associated with our product candidates; |
| | the terms of any approvals and the countries in which approvals are obtained; |
| | limitations or warnings contained in any labeling approved by the FDA or comparable foreign regulatory authorities; |
| | conditions upon the approval imposed by FDA or comparable foreign regulatory authorities, including, but not limited to, a REMS; |
| | the willingness of patients to try new treatments and of physicians to prescribe these treatments; |
| | the availability of government and other third-party payer coverage and adequate reimbursement; and |
| | the availability of alternative effective treatments for the disease indications our product candidates are intended to treat and the relative risks, benefits and costs of those treatments. |
Since we are focused on the emerging therapeutic modality of ddRNAi, and silence and replace these risks may increase if new competitors are able to market ddRNAi-based therapeutics or silence and replace-based therapeutics or if these treatments become less favored in the commercial marketplace. In addition, we believe that one of the benefits of our ddRNAi and silence and replace technologies is the expected length of time of the effects. If our treatments do not have a long-term effect after administration, such a development would likely significantly and adversely affect market acceptance of our product candidates, if approved.
Additional risks apply in relation to any disease indications we pursue which are classified as rare diseases and allow for orphan drug designation by regulatory agencies in major commercial markets, such as the United States or European Union. If pricing is not approved or accepted in the market at an appropriate level for any approved product for which we pursue and receive an orphan drug designation, such product may not generate enough revenue to offset costs of development, manufacturing, marketing and commercialization despite any benefits received from the orphan drug designation, such as market exclusivity, for a period of time. Orphan exclusivity could temporarily delay or block approval of one of our products if a competitor obtains orphan drug designation for its product first. However, even if we obtain orphan exclusivity for one of our products upon approval, our exclusivity may not block the subsequent approval of a competitive product that is shown to be clinically superior to our product.
Market size is also a variable in disease indications not classified as rare. Our estimates regarding potential market size for any indication may be materially different from what we discover to exist at the time we commence commercialization, if any, for a product, which could result in significant changes in our business plan and have a material adverse effect on our business, financial condition, results of operations and prospects.
40
Table of Contents
The COVID-19 pandemic and other public health crises may materially and adversely affect our business, financial condition and results of operations.
The COVID-19 pandemic and other public health crises may materially and adversely affect our business, financial condition and results of operations. The development and commercialization of our products is dependent on the efforts of our third-party collaborators, as we rely on third-parties to conduct our preclinical studies and clinical trials, and for our supply of research and development, preclinical and clinical development biologics and other materials. Accordingly, our operations and supply chain could be subject to significant disruptions to the extent that our third-party partners are impacted by the COVID-19 pandemic or the associated governmental actions. There may be related restrictions or delays with respect to clinical trials and preclinical studies, as well as the export, import or shipment of raw materials, drug substance or drug product that could materially delay our business or clinical trials.
Further, certain of our research and development efforts are conducted globally, including our ongoing development of our silence and replace therapeutic for the treatment of Oculopharyngeal Muscular Dystrophy (OPMD), and will be dependent upon our ability to initiate clinical sites and enroll patients despite the ongoing COVID-19 pandemic.
We had also implemented work-from-home measures for the majority of our employees between March 2020 and June 2020, resulting in a reduction of laboratory work and a halt of non-essential business travel. As we transition our employees back to our premises, there is a risk that COVID-19 infections occur at our offices or laboratory facilities and significantly affect our operations. Additionally, if any of our critical vendors are impacted, our business could be affected if we become unable to timely procure essential equipment, supplies or services in adequate quantities and at acceptable prices.
Moreover, the COVID-19 outbreak has caused significant disruption to the global economy and in the global and U.S. financial markets. This could impact our ability to raise additional funding, which may not be available on acceptable terms, if at all.
The extent to which COVID-19 impacts our results will depend on future developments, which are highly uncertain and cannot be predicted, including new information which may emerge concerning the severity of COVID-19 and the actions to contain COVID-19 or treat its impact, among others. In addition, due to the speed with which the COVID-19 situation is developing and evolving, there is uncertainty around its ultimate impact on public health, business operations and the overall economy; therefore, the negative impact on our financial position, operating results and liquidity cannot be reasonably estimated at this time, but the impact may be material.
We face competition from entities that have developed or may develop product candidates for our target disease indications, including companies developing novel treatments and technology platforms based on modalities and technology similar to ours.
The development and commercialization of pharmaceutical products is highly competitive. We compete with a variety of multinational pharmaceutical companies and specialized biotechnology companies, as well as technology being developed at universities and other research institutions. Our competitors have developed, are developing or could develop product candidates and processes competitive with our product candidates. Competitive therapeutic treatments include those that have already been approved and accepted by the medical community, patients and third-party payers, and any new treatments that enter the market.
There may be a significant number of products that are currently under development, and may become commercially available in the future, for the treatment of conditions for which we are developing, and may in the future try to develop, product candidates. This increasingly competitive landscape may compromise the development of our product candidates.
41
Table of Contents
We are aware of multiple companies that are working in the field of RNAi therapeutics, including Alnylam, Arbutus, Arrowhead, Silence Therapeutics plc, RXi Pharmaceuticals Corporation, Quark Pharmaceuticals, Inc., Marina Biotech, Inc. and Dicerna. Arrowhead, Arbutus and Alnylam are all developing siRNA-based therapeutics for Chronic Hepatitis B Virus Infection, or HBV. Some of our current product candidates, if approved, would compete with approved and currently marketed treatments.
In addition, our ddRNAi-based product candidates would compete with antisense and other RNA-based pharmaceutical products currently under development. Like RNAi therapeutics, antisense products target mRNA with the objective of suppressing the activity of specific genes. The development of antisense products is more advanced than that of RNAi therapeutics, and antisense technology may become the preferred technology for products that target mRNAs.
Many of our competitors have significantly greater financial, technical, manufacturing, marketing, sales and supply resources and experience than we have. If we successfully obtain approval for any product candidate, we will face competition based on many different factors, including the safety and effectiveness of our products, the ease with which our products can be administered and the extent to which patients accept relatively new routes of administration, the timing and scope of regulatory approvals for these products, the availability and cost of manufacturing, marketing and sales capabilities, price, reimbursement coverage and patent position.
Competing products could present superior treatment alternatives, including by being more effective, safer, less expensive or marketed and sold more effectively than any products we may develop. Competitive products may make any products we develop obsolete or non-competitive before we recover the expense of developing and commercializing our product candidates. Such competitors could also recruit our employees, which could negatively impact our level of expertise and our ability to execute our business plan.
A variety of risks outside of our control associated with international operations could adversely affect our business.
If any of our product candidates are approved for commercialization, it is our current intention to market them on a worldwide basis, either alone or in collaboration with others. In addition, we conduct development activities in various jurisdictions throughout the world. We expect that we will be subject to additional risks related to engaging in international operations, including:
| | different regulatory requirements for approval of biopharmaceutical products in foreign countries; |
| | reduced protection for intellectual property rights; |
| | unexpected changes in tariffs, trade barriers and regulatory requirements; |
| | economic weakness, including inflation, or political instability in particular foreign economies and markets; |
| | compliance with tax, employment, immigration and labor laws for employees living or traveling abroad; |
| | foreign currency fluctuations, which could result in increased operating expenses and reduced revenue, and other obligations incident to doing business in another country; |
| | workforce uncertainty in countries where labor unrest is more common than in Australia or the United States; |
| | production shortages resulting from any events affecting raw material supply or manufacturing capabilities abroad; and |
| | business interruptions resulting from geopolitical actions, including war and terrorism, natural disasters, including earthquakes, typhoons, floods and fires and disease pandemics and epidemics, including the current COVID-19 pandemic. |
42
Table of Contents
The insurance coverage and reimbursement status of newly approved products is uncertain.
The availability of coverage and adequate reimbursement by governmental and private payers is essential for most patients to be able to afford expensive treatments. Sales of our product candidates will depend substantially, both in the United States and abroad, on the extent to which the costs of our product candidates will be paid by health maintenance, managed care, pharmacy benefit and similar healthcare management organizations, or reimbursed by third-party payers. If reimbursement is not available, or is available only to limited levels, we may not be able to successfully commercialize our product candidates. Even if coverage is provided, the reimbursement amounts approved by third-party payers may not be high enough to allow us to establish or maintain pricing sufficient to realize a return on our investment.
There is significant uncertainty related to the insurance coverage and reimbursement of newly approved products. In the United States, the principal decisions about reimbursement for new medicines are typically made by the Centers for Medicare & Medicaid Services, or CMS, an agency within the U.S. Department of Health and Human Services, as CMS decides whether and to what extent a new medicine will be covered and reimbursed under Medicare. Private payers tend to follow CMS to a substantial degree. It is difficult to predict what CMS will decide with respect to reimbursement for fundamentally novel products such as ours, as there is no body of established practices and precedents for these new products.
The intended use of a biopharmaceutical product by a physician can also affect pricing. For example, CMS could initiate a National Coverage Determination administrative procedure, by which the agency determines which uses of a therapeutic product would and would not be reimbursable under Medicare. This determination process can be lengthy, thereby creating a long period during which the future reimbursement for a particular product may be uncertain.
Outside the United States, international operations are generally subject to extensive governmental price controls and other market regulations, and we believe the increasing emphasis on cost-containment initiatives in Europe, Canada and other countries is likely to put pressure on the pricing and usage of any of our product candidates that may be approved for marketing in the future. In many countries, the prices of medical products are subject to varying price control mechanisms as part of national health systems. In general, the prices of medicines under such systems can be substantially lower than in the United States. Other countries allow companies to fix their own prices for medicines, but monitor and control company profits. In some countries, we or our collaborators may be required to conduct a clinical trial or other studies that compare the cost-effectiveness of our product candidates to other available therapies in order to obtain or maintain reimbursement or pricing approval. Additional foreign price controls or other changes in pricing regulation could restrict the amount that we are able to charge for our product candidates. Accordingly, in markets outside the United States, the reimbursement for our products may be reduced compared with the United States and may be insufficient to generate commercially reasonable revenue and profits.
Moreover, increasing efforts by governmental and other third-party payers, in the United States and abroad, to cap or reduce healthcare costs, have resulted in legislation and reforms such as, in the United States, the Patient Protection and Affordable Care Act of 2010, as amended by the Health Care Education Reconciliation Act, or the ACA. The ACA may cause such organizations to limit both coverage and level of reimbursement for new products approved and, as a result, they may not cover or provide adequate payment for our product candidates. In addition, other legislative changes have been adopted since the ACA was enacted. These changes included aggregate reductions to Medicare payments to providers of up to 2% per fiscal year effective April 2013 that, due to subsequent legislative amendments to the statute, will stay in effect through 2029 unless additional Congressional action is taken. Note, however, the Coronavirus Aid, Relief, and Economic Security Act (CARES Act), which was signed into law on March 27, 2020, designed to provide financial support and resources to individuals and businesses affected by the COVID-19 pandemic, suspended the 2% Medicare sequester from May 1, 2020 through December 31, 2020, and extended the sequester by one year, through 2030.
43
Table of Contents
Since January 2017, President Trump has signed Executive Orders and other directives designed to delay the implementation of certain provisions of the ACA or otherwise circumvent some of the requirements for health insurance mandated by the ACA. Concurrently, Congress has considered legislation that would repeal or repeal and replace all or part of the ACA. While Congress has not passed comprehensive repeal legislation, several bills affecting the implementation of certain taxes under the ACA have passed. On December 22, 2017, President Trump signed into law federal tax legislation commonly referred to as the Tax Cuts and Jobs Act (the Tax Act), which includes a provision repealing, effective January 1, 2019, the tax-based shared responsibility payment imposed by the ACA on certain individuals who fail to maintain qualifying health coverage for all or part of a year that is commonly referred to as the individual mandate. The 2020 federal spending package permanently eliminated, effective January 1, 2020, the ACA-mandated Cadillac tax on high-cost employer-sponsored health coverage and medical device tax and, effective January 1, 2021, also eliminates the health insurer tax. The Bipartisan Budget Act of 2018, among other things, amended the ACA, effective January 1, 2019, to close the coverage gap in most Medicare Part D drug plans. In December 2018, the Centers for Medicare & Medicaid Services (CMS) published a final rule permitting further collections and payments to and from certain ACA-qualified health plans and health insurance issuers under the ACA risk adjustment program in response to the outcome of federal district court litigation regarding the method CMS uses to determine this risk adjustment.
On December 14, 2018, a Texas U.S. District Court Judge ruled that the ACA is unconstitutional in its entirety because the individual mandate was repealed by Congress as part of the Tax Act. Additionally, on December 18, 2019, the U.S. Court of Appeals for the 5th Circuit upheld the District Court ruling that the individual mandate was unconstitutional and remanded the case back to the District Court to determine whether the remaining provisions of the ACA are invalid as well. On March 2, 2020, the United States Supreme Court granted the petitions for writs of certiorari to review this case, although it is unclear when the Supreme Court will make a decision. It is also unclear how such litigation and other efforts to repeal and replace the ACA will impact the ACA and our business.
In addition, other legislative changes have been proposed and adopted in the United States since the ACA was enacted. In January 2013, former President Obama signed into law the American Taxpayer Relief Act of 2012, which, among other things, reduced Medicare payments to several providers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. This law may result in additional reductions in Medicare and other healthcare funding, which could have a material adverse effect on customers for our product candidates, if approved, and accordingly, our financial operations.
We expect to experience pricing pressures in connection with the sale of any of our product candidates due to the trend toward managed healthcare, the increasing influence of health maintenance organizations and additional legislative changes. The downward pressure on healthcare costs in general, particularly prescription pharmaceutical products and surgical procedures and other treatments, has become very intense. As a result, increasingly high barriers are being erected to the entry of new products. We cannot predict what healthcare reform initiatives may be adopted in the future. It is possible that additional governmental action is taken to address the COVID-19 pandemic. Further federal, state and foreign legislative and regulatory developments are likely, and we expect ongoing initiatives to increase pressure on biopharmaceutical product pricing. Such reforms could depress pricing for any product candidates that we may successfully develop and for which we may obtain regulatory approval and may negatively affect our overall financial condition and ability to develop additional product candidates.
Changes in U.S. tax law may materially adversely affect our financial condition, results of operations and cash flows.
The recently enacted the Tax Act has significantly changed the U.S. federal income taxation of U.S. businesses, including by reducing the U.S. corporate income tax rate, limiting interest deductions, permitting immediate expensing of certain capital expenditures, modifying or repealing many business deductions and credits.
44
Table of Contents
The CARES Act modifies certain provisions of the Tax Act, including increasing the amount of interest expense that may be deducted.
The Tax Act as modified by the CARES Act is unclear in many respects and could be subject to potential amendments and technical corrections, as well as interpretations and implementing regulations by the Treasury and IRS, any of which could lessen or increase certain adverse impacts of the legislation. In addition, it is unclear how these U.S. federal income tax changes will affect state and local taxation, which often uses federal taxable income as a starting point for computing state and local tax liabilities. Our analysis and interpretation of this legislation is preliminary and ongoing. Some of the changes made by the tax legislation may adversely affect us. We continue to work with our tax advisors and auditors to determine the full impact that the recent tax legislation as a whole will have on us. We urge our investors to consult with their legal and tax advisors with respect to such legislation and its potential effect on an investment in our common stock and pre-funded warrants.
Our relationships with third-party payers, healthcare professionals and customers in the United States and elsewhere may be subject to anti-kickback, fraud and abuse, false claims, transparency, health information privacy and security and other healthcare laws and regulations, which could expose us to significant penalties.
Our relationships with third-party payers, healthcare professionals and customers may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations, including, without limitation, the federal Anti-Kickback Statute and the federal False Claims Act, that may constrain the business or financial arrangements and relationships through which we sell, market and distribute any biopharmaceutical products for which we obtain marketing approval. In addition, we may be subject to transparency laws and patient privacy regulation by the federal government and by the U.S. states and foreign jurisdictions in which we conduct our business. The applicable federal, state and foreign healthcare laws and regulations that may affect our ability to operate include, but are not limited to, the following:
| | the federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made under federal and state healthcare programs such as Medicare and Medicaid; |
| | federal civil and criminal false claims laws and civil monetary penalty laws, including the federal False Claims Act, which impose criminal and civil penalties, including civil whistleblower or qui tam actions, against individuals or entities for knowingly presenting, or causing to be presented, to the federal government, including the Medicare and Medicaid programs, claims for payment that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government; |
| | the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which imposes criminal and civil liability for, among other things, executing a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters; |
| | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, or HITECH, and their respective implementing regulations, which impose obligations on covered healthcare providers, health plans and healthcare clearinghouses, as well as their business associates that create, receive, maintain or transmit individually identifiable health information for or on behalf of a covered entity, with respect to safeguarding the privacy, security and transmission of individually identifiable health information; |
| | the federal Open Payments program, created under the ACA, and its implementing regulations, which requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Childrens Health Insurance Program, with specific exceptions, to report annually to the CMS information related to payments or other transfers of value |
45
Table of Contents
| made to physicians, and applicable group purchasing organizations to report annually to CMS ownership and investment interests held by the physicians and their immediate family members by the 90th day of each subsequent calendar year, and disclosure of such information will be made by CMS on a publicly available website; and |
| | analogous state and foreign laws and regulations, such as state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payers, including private insurers; state and foreign laws that require biopharmaceutical or biotechnology companies to comply with the industry voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government or otherwise restrict payments that may be made to healthcare providers; state and foreign laws that require biopharmaceutical or biotechnology manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures; and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. |
Efforts to ensure that our business arrangements with third parties will comply with applicable healthcare laws and regulations may involve substantial costs. It is possible that governmental authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, including, without limitation, damages, fines, disgorgement, imprisonment, exclusion from participation in government healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations, which could have a material adverse effect on our business. If any of the physicians or other healthcare providers or entities with whom we expect to do business is found not to be in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from participation in government healthcare programs, which could also materially affect our business.
Negative public opinion and increased regulatory scrutiny of gene therapy and genetic research may damage public perception of our product candidates or compromise our ability to conduct our business or obtain regulatory approvals for our product candidates.
Gene therapy remains a novel technology and no gene therapy product utilizing ddRNAi or silence and replace has been approved to date in the United States. Public perception may be influenced by claims that gene therapy is unsafe, and gene therapy may not gain the acceptance of the public or the medical community. In particular, our success will depend upon physicians specializing in the treatment of those diseases that our product candidates target prescribing treatments that involve the use of our product candidates in lieu of, or in addition to, existing treatments they are already familiar with and for which greater clinical data may be available. More restrictive government regulations or negative public opinion would have a negative effect on our business or financial condition and may delay or impair the development and commercialization of our product candidates or demand for any products we may develop. Our product candidates, including our viral delivery systems, could produce adverse events. Adverse events in our clinical trials or following approval of any of our product candidates, even if not ultimately attributable to our product candidates, could result in increased governmental regulation, unfavorable public perception, potential regulatory delays in the testing or approval of our product candidates, stricter labeling requirements for those product candidates that are approved and a decrease in demand for any such product candidates.
46
Table of Contents
Risks Related to Our Business Operations
We may not successfully engage in strategic transactions or enter into new collaborations, which could adversely affect our ability to develop and commercialize product candidates, impact our cash position, increase our expenses and present significant distractions to our management.
From time to time, we may consider additional strategic transactions, such as collaborations, acquisitions, asset purchases or sales and out- or in-licensing of product candidates or technologies. In particular, we will evaluate and, if strategically attractive, seek to enter into additional collaborations, including with major biotechnology or pharmaceutical companies. The competition for collaborators is significant, and the negotiation process is time-consuming and complex. Any new collaboration may be on terms that are not optimal for us, and we may not be able to maintain any new or existing collaboration if, for example, development or approval of a product candidate is delayed, sales of an approved product candidate do not meet expectations or the collaborator discontinues the collaboration. Any such collaboration, or other strategic transaction, may require us to incur non-recurring or other charges, increase our expenditures, pose significant integration or implementation challenges or disrupt our management or business.
These transactions would entail numerous operational and financial risks, including exposure to unknown liabilities, incurrence of substantial debt or dilutive issuances of equity securities to pay transaction consideration or costs, higher than expected collaboration, acquisition or integration costs, write-downs of assets or goodwill or impairment charges, increased amortization expenses, difficulty and cost in facilitating the collaboration or combining the operations and personnel of any acquired business, impairment of relationships with key suppliers, manufacturers or customers of any acquired business due to changes in management and ownership and the inability to retain key employees of any acquired business.
Accordingly, although there can be no assurance that we will undertake or successfully complete any additional transactions of the nature described above, any transactions that we do complete may be subject to the foregoing or other risks and have a material adverse effect on our business, results of operations, financial condition and prospects. Conversely, any failure to enter any collaboration or other strategic transaction that would be beneficial to us could delay and make more expensive the development and potential commercialization of our product candidates and have a negative impact on the competitiveness of any product candidate that reaches market.
Any inability to attract and retain qualified key management and technical personnel would impair our ability to implement our business plan.
Our success largely depends on the continued service of key management and other specialized personnel, including Dr. Jerel Banks (Chief Executive Officer) and Ms. Megan Boston (Executive Director). The loss of one or more members of our management team or other key employees or advisors, if not adequately replaced, could delay or increase the cost of our research and development programs and materially harm our business, financial condition, results of operations and prospects. The relationships that our key managers have cultivated within our industry make us particularly dependent upon their continued employment with us. We are dependent on the continued service of our technical personnel because of the highly technical nature of our product candidates and the specialized nature of the regulatory approval process for our product candidates. Because our management team and key employees are not obligated to provide us with continued service, they could terminate their employment with us at any time without penalty. We do not maintain key person life insurance policies on any of our management team members or key employees. Our future success will depend in large part on our continued ability to attract and retain other highly qualified scientific, technical and management personnel, as well as personnel with expertise in clinical testing, manufacturing, governmental regulation and commercialization. We face competition for personnel from other companies, universities, public and private research institutions, government entities and other organizations.
47
Table of Contents
Our collaborations with outside scientists and consultants may be subject to restriction and change.
We work with medical experts, chemists, biologists and other scientists at academic and other institutions, and consultants who assist us in our research, development and regulatory efforts, including the members of our scientific advisory board. In addition, these scientists and consultants have provided, and we expect that they will continue to provide, valuable advice regarding our programs and regulatory approval processes. These scientists and consultants are not our employees and may have other commitments that would limit their future availability to us.
If a conflict of interest arises between their work for us and their work for another entity, we may lose their services. In addition, we are limited in our ability to prevent them from establishing competing businesses or developing competing products. For example, if a key scientist acting as a principal investigator in any of our future clinical trials identifies a potential product or compound that is more scientifically interesting to his or her professional interests, his or her availability to remain involved in any future clinical trials could be restricted or eliminated.
We may experience difficulties in managing our growth and expanding our operations.
We have limited experience in development and commercialization of pharmaceutical products. As our product candidates continue to advance through preclinical studies and any future clinical trials and potentially toward regulatory approval and commercial sale, we will need to expand our development, regulatory, manufacturing and sales capabilities or contract with other organizations to provide these capabilities for us. In the future, we expect to have to manage additional relationships with collaborators or partners, suppliers and other organizations. Our ability to manage our operations and future growth will require us to continue to improve our operational, financial and management controls, reporting systems and procedures. We may not be able to implement improvements to our management information and control systems in an efficient or timely manner and may discover deficiencies in existing systems and controls.
Our employees, independent contractors, principal investigators, CROs, consultants and vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and insider trading.
We are exposed to the risk of fraud or other misconduct by our employees, independent contractors, principal investigators, CROs, consultants, commercial partners and vendors. Misconduct by these parties could include intentional failures to comply with the regulations of the FDA and comparable foreign regulators, provide accurate information to the FDA and comparable foreign regulators, comply with healthcare fraud and abuse laws and regulations in the United States and abroad, report financial information or data accurately or disclose unauthorized activities to us. In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Such misconduct could also involve the improper use of information obtained in the course of any future clinical trials, which could result in regulatory sanctions and cause serious harm to our reputation. We have adopted a code of conduct applicable to all of our employees, but it is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of civil, criminal and administrative penalties, damages, monetary fines, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
48
Table of Contents
We could face potential product liability and, if successful claims are brought against us, we may incur substantial liability and costs.
The use of our product candidates in clinical trials and the sale of any products for which we may in the future obtain marketing approval exposes us to the risk of product liability claims. Product liability claims might be brought against us by consumers, healthcare providers, pharmaceutical companies or others selling or otherwise coming into contact with our product candidates. There is a risk that our product candidates may induce adverse events. If we cannot successfully defend against product liability claims, we could incur substantial liability and costs. In addition, regardless of merit or eventual outcome, product liability claims may result in:
| | impairment of our business reputation; |
| | withdrawal of clinical trial participants; |
| | costs due to related litigation; |
| | distraction of managements attention from our primary business; |
| | substantial monetary awards to patients or other claimants; |
| | the inability to commercialize our product candidates; |
| | decreased demand for our product candidates, if approved for commercial sale; and |
| | increased cost, or impairment of our ability, to obtain or maintain product liability insurance coverage. |
We carry combined general public and products liability (including human clinical trials extension) insurance of $20 million (per any one occurrence) and $10 million (per any one occurrence and in the aggregate), respectively. We believe our product liability insurance coverage is sufficient in light of our current clinical programs. However, we may not be able to maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to liability. If we obtain marketing approval for any product candidates, we intend to expand our insurance coverage to include the sale of commercial products, but we may not be able to obtain or maintain product liability insurance on commercially reasonable terms or in adequate amounts. On occasion, large judgments have been awarded against other pharmaceutical companies in class action lawsuits based on pharmaceutical products, or medical treatments that had unanticipated adverse effects. A successful product liability claim or series of claims brought against us could cause the price of our common stock to decline and, if judgments exceed our insurance coverage, could materially and adversely affect our financial position. During the course of treatment, patients may suffer adverse events, including death, for reasons that may be related to our product candidates. Such events could subject us to costly litigation, require us to pay substantial amounts of money to injured patients, delay, negatively impact or end our opportunity to receive or maintain regulatory approval to market our products, or require us to suspend or discontinue our commercialization efforts. Even in a circumstance in which we do not believe that an adverse event is related to our product candidate, the investigation into the circumstance may be time-consuming or inconclusive. These investigations may harm our reputation, delay our regulatory approval process, limit the type of regulatory approvals our product candidates receive or maintain, and compromise the market acceptance of any of our product candidates that may in the future receive regulatory approval. As a result of these factors, a product liability claim, even if successfully defended, could hurt our business and impair our ability to generate revenue.
We and our development partners, third-party manufacturers and suppliers use biological materials and may use hazardous materials, and any claims relating to improper handling, storage or disposal of these materials could be time consuming or costly.
We and our development partners, third-party manufacturers and suppliers may use hazardous materials, including chemicals and biological agents and compounds that could be dangerous to human health and safety or the environment. Our operations and the operations of our third-party manufacturers and suppliers also produce
49
Table of Contents
hazardous waste products. National, state and local laws and regulations in the United States and other countries govern the use, generation, manufacture, storage, handling and disposal of these materials and wastes. Compliance with applicable environmental laws and regulations may be expensive, and current or future environmental laws and regulations may impair our product development and commercialization efforts. In addition, we cannot entirely eliminate the risk of accidental injury or contamination from these materials or wastes. We do not carry specific biological or hazardous waste insurance coverage, and our property, casualty and general liability insurance policies specifically exclude coverage for damages and fines arising from biological or hazardous waste exposure or contamination. Accordingly, in the event of contamination or injury, we could be held liable for damages or be penalized with fines in an amount exceeding our resources, and any future clinical trials, regulatory approvals or product commercialization progress could be suspended.
We may use our limited financial and human resources to pursue a particular research program or product candidate and fail to capitalize on programs or product candidates that may be more profitable or for which there is a greater likelihood of success.
Because we have limited resources, we may forego or delay pursuit of opportunities with certain programs or product candidates or for indications that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable commercial products or profitable market opportunities. Our spending on current and future research and development programs for product candidates may not yield any commercially viable products. If we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights to that product candidate through strategic collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to such product candidate, or we may allocate internal resources to a product candidate in a therapeutic area in which it would have been more advantageous to enter into a collaboration arrangement.
Our internal computer and information technology systems, or those of our collaborators and other development partners, third-party CROs or other contractors or consultants, may fail or suffer security breaches, which could result in a disruption of our product development programs.
Despite the implementation of security measures, our internal computer and information technology systems and those of our current and any future CROs and other contractors, consultants and collaborators are vulnerable to damage from computer viruses, cyber-attacks, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. Such events could cause interruptions of our operations. While we have not experienced any material system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a disruption of our development programs and our business operations, whether due to a loss of our trade secrets or other similar disruptions. For example, the loss of clinical trial data from ongoing or future clinical trials or data from preclinical studies could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. Likewise, we rely on third parties to manufacture our product candidates and will rely on third parties to conduct future clinical trials, and similar events relating to their computer systems could also have similar consequences to our business. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development and commercialization of our product candidates could be delayed and become more expensive. As a result of COVID-19, we may face increased cybersecurity risks due to our reliance on internet technology and the number of our employees that are working remotely, which may create additional opportunities for cybercriminals to exploit vulnerabilities.
Cyber-attacks, breaches, interruptions or other data security incidents could result in legal claims or proceedings, liability under federal or state laws that protect the privacy of personal information, regulatory penalties, significant remediation costs, disrupt key business operations and divert attention of management and key information technology resources. In the United States, notice of breaches must be made to affected
50
Table of Contents
individuals, the U.S. Secretary of the Department of Health and Human Services, or HHS, and for extensive breaches, notice may need to be made to the media or U.S. state attorneys general. Such a notice could harm our reputation and our ability to compete. HHS has the discretion to impose penalties without attempting to resolve violations through informal means. In addition, U.S. state attorneys general are authorized to bring civil actions seeking either injunctions or damages in response to violations that threaten the privacy of state residents. There can be no assurance that we, our collaborators, CROs, vendors, and any other business counterparties will be successful in efforts to detect, prevent, protect against or fully recover systems or data from all break-downs, service interruptions, attacks or breaches of systems. In addition, we do not maintain standalone cyber-security insurance and have limited insurance coverage in the event of any breach or disruption of our or our collaborators, CROs, or vendors systems, including any unauthorized access or loss of any personal data that we may collect, store or otherwise process. The costs related to significant security breaches or disruptions could be material and exceed the limits of any insurance coverage we may have. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or systems, or inappropriate disclosure of confidential or proprietary information, including data related to our personnel, we could incur liability and the further development and commercialization of our product candidates could be delayed and our business and operations could be adversely affected and/or could result in the loss or disclosure of critical or sensitive data, which could result in financial, legal, business or reputational harm to us.
Our current laboratory operations are concentrated in one location and any events affecting this location may seriously compromise our ability to operate our business and continue the development of our product candidates.
Our current laboratory operations are located in our facility situated in Hayward, California. Any unplanned event, such as flood, fire, explosion, earthquake, extreme weather condition, medical epidemics, including the current COVID-19 pandemic, power shortage, telecommunication failure or other natural or manmade accidents or incidents that result in us being unable to fully utilize the facility, may compromise our ability to operate our business, particularly on a daily basis, cause us financial losses and inhibit or delay our continued development of our product candidates. Loss of access to this facility may result in increased costs, delays in the development of our product candidates or interruption of our business operations. As part of our risk management policy, we maintain insurance coverage at levels that we believe are appropriate for our business. However, in the event of an accident or incident at this facility, we cannot assure you that the amounts of insurance will be sufficient to satisfy any damages and losses. If our facility is unable to operate because of an accident or incident or for any other reason, even for a short period of time, any or all of our research and development programs may be harmed. Any business interruption may have a material adverse effect on our business, financial position, results of operations and prospects.
The investment of our cash and cash equivalents is subject to risks which may cause losses and affect the liquidity of these investments.
As of March 31, 2020 and June 30, 2019, we had $11.4 million and $15.7 million, respectively, in cash and cash equivalents. We historically have invested substantially all of our available cash and cash equivalents in cash deposits meeting the criteria of our investment policy, which is focused on the preservation of our capital. These investments are subject to general credit, liquidity, market and interest rate risks. We may realize losses in the fair value of these investments, which would have a negative effect on our financial results. In addition, should our investments cease paying or reduce the amount of interest paid to us, our interest income would suffer. The market risks associated with our investment portfolio may have an adverse effect on our results of operations, liquidity and financial condition.
51
Table of Contents
Risks Related to Our Intellectual Property
If we are unable to obtain or protect sufficient intellectual property rights related to our product candidates, we may not be able to obtain exclusivity for our product candidates or prevent others from developing similar competitive products.
We rely upon a combination of patents, know-how, trade secret protection and confidentiality agreementsboth that we own or possess or that are owned or controlled by our licensors and licensed to usto protect the intellectual property related to our technology and product candidates.
The patent application process, also known as patent prosecution, is expensive and time-consuming, and we and our current or future licensors and licensees may not be able to prepare, file, and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we or our current licensors or licensees, or any future licensors or licensees, will fail to identify or obtain sufficient protection for patentable aspects of inventions made in the course of development and commercialization activities before it is too late to obtain patent protection on them. Therefore, our patents and patent applications may not be prosecuted and enforced in a manner consistent with the best interests of our business. It is possible that defects of form in the preparation or filing of our patents or patent applications may exist, or may arise in the future, for example with respect to proper priority claims, inventorship, claim scope, patent term adjustments, etc., although we are unaware of any such defects. If we or our current licensors or licensees, or any future licensors or licensees, fail to establish, maintain or protect such patents and other intellectual property rights, such rights may be reduced or eliminated. If our current licensors or licensees, or any future licensors or licensees, are not fully cooperative or disagree with us as to the prosecution, maintenance or enforcement of any patent rights, such patent rights could be compromised. If there are material defects in the form or preparation of our patents or patent applications, such patents or applications may be invalid and unenforceable in circumstances where it is not possible to remedy those material defects. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business.
The strength of patents in the biotechnology and pharmaceutical field involves complex legal and scientific questions and can be uncertain. The patent applications that we own or in-license may fail to result in issued patents with claims that cover our product candidates in the United States or other jurisdictions. In addition, we cannot guarantee that any patents will issue from any pending or future patent applications owned by or licensed to us. There is no assurance that all of the potentially relevant prior art relating to our patents and patent applications has been found. If such prior art exists, it can invalidate a patent or prevent a patent from issuing from a pending patent application. Even if patents do successfully issue and even if such patents cover our product candidates, third parties may initiate opposition, interference, re-examination, post-grant review, inter partes review, nullification or derivation action in court or before patent offices or similar proceedings challenging the validity, enforceability or scope of such patents, which may result in the patent claims being narrowed or invalidated. Furthermore, even if our patents and patent applications are unchallenged, they may not adequately protect our intellectual property, provide exclusivity for our product candidates or prevent others from designing around our claims. Any of these outcomes could impair our ability to prevent competition from third parties.
If the patent applications we hold or have in-licensed with respect to our programs or product candidates fail to issue, or are revoked, if the breadth or strength of our patent protection is threatened, or if our patent portfolio fails to provide meaningful exclusivity for our product candidates, it could dissuade companies from collaborating with us to develop product candidates and threaten our ability to commercialize future products. Any successful opposition to any patents owned by or licensed to us could deprive us of rights necessary for the successful commercialization of any product candidates that we may develop. Further, if we encounter delays in regulatory approvals, the period of time during which we could market a product candidate under patent protection could be reduced. Since patent applications in the United States and most other countries are confidential for a period of time (up to 18 months) after filing, we cannot be certain that we or our licensors were the first to file any patent application related to a product candidate. Furthermore, if third parties have filed such
52
Table of Contents
patent applications before March 16, 2013, an interference proceeding in the United States can be initiated by such third parties to determine who was the first to invent any of the subject matter covered by the patent claims of our applications. If third parties have filed such applications on or after March 16, 2013, a derivation proceeding in the United States can be initiated by such third parties to determine whether our invention was derived from theirs. Even where we have a valid and enforceable patent, we may not be able to exclude others from practicing our invention where the other party can show that they used the invention in commerce before our filing date or the other party benefits from a compulsory license. In addition, patents have a limited lifespan. In the United States, and in most other jurisdictions with a patent system, the natural expiration of a patent is generally 20 years after its filing date. Various extensions may be available, but the life of a patent, and the protection it affords, is limited. Even if patents covering our product candidates are obtained, once the patent life has expired for a product, we may be open to competition from competitive medications, including biosimilar or generic medications. This risk is material in light of the length of the development process of our products and lifespan of our current patent portfolio.
In addition to the protection afforded by patents, we rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or that we elect not to patent, processes for which patents are difficult to enforce and any other elements of our product candidate discovery and development processes that involve proprietary know-how, information or technology that is not covered by patents. However, trade secrets can be difficult to protect. What constitutes a trade secret and what protections are available for trade secrets varies from state to state in the United States and country by country worldwide. We seek to protect our proprietary technology and processes, in part, by entering into confidentiality agreements with our employees, consultants, scientific advisors and contractors. We also seek to preserve the integrity and confidentiality of our data and trade secrets by measures designed to maintain the physical security of our premises and physical and electronic security of our information technology systems. Security measures may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known to or be independently discovered by competitors. Although we expect all of our employees and consultants to assign their inventions to us, and all of our employees, consultants, advisors and any third parties who have access to our proprietary know-how, information or technology to enter into confidentiality agreements, we cannot provide any assurances that all such agreements have been duly executed, that our trade secrets and other confidential proprietary information will not be disclosed or that competitors will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. Misappropriation or unauthorized disclosure of our trade secrets can be difficult to detect, could impair our competitive position and may have a material adverse effect on our business. Additionally, if the steps taken to maintain our trade secrets are deemed inadequate, we may have insufficient recourse against third parties for misappropriating the trade secret. In addition, others may independently discover our trade secrets and proprietary information. For example, the FDA, as part of its Transparency Initiative, is currently considering whether to make additional information publicly available on a routine basis, including information that we may consider to be trade secrets or other proprietary information, and it is not clear at the present time how the FDAs disclosure policies may change in the future, if at all.
Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. Further, the laws of some foreign countries such as India and China do not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, we may encounter significant problems in protecting and defending our intellectual property both in the United States and abroad. If we are unable to prevent material disclosure of the non-patented intellectual property related to our technologies to third parties, and there is no guarantee that we will have any such enforceable trade secret protection, we may not be able to establish or maintain a competitive advantage in our markets.
53
Table of Contents
We rely on license relationships with a number of third parties for portions of our intellectual property, including platform technology patents relating to our ddRNAi technology.
We have in-licensed certain intellectual property from third parties, including technology related to ddRNAi and antisense RNA. In some cases, our licenses to intellectual property are non-exclusive and the licensors may license the technology to our competitors in the same field, which may result is significant competition for us. In other cases, our licenses to intellectual property are exclusive only for a specific field of use (such as human therapeutics), and the licensors retain rights to practice the licensed intellectual property themselves and to grant licenses to third parties in other fields. Such third parties may develop improvements to the licensed intellectual property that are not licensed to us, which could block our ability to continue developing the product candidate covered by the licensed intellectual property, unless we negotiate a license. Such third parties may also disclose competitively-sensitive information about the licensed intellectual property that diminishes its value. In other cases, our licenses are for research purposes only. Upon regulatory marketing approval of our product candidates it may be necessary for us to obtain a broader license in order to commercialize. We cannot guarantee the availability of the broader license or that it can be obtained on commercially reasonable terms.
We rely on some of these third party licensors to file and prosecute patent applications and maintain patents and otherwise protect the intellectual property we license. We have not had and do not have primary control over these activities for certain of our patents or patent applications and other intellectual property rights we license, and therefore cannot guarantee that these patents and applications will be prosecuted or enforced in a manner consistent with the best interests of our business. We cannot be certain that such activities by third parties have been or will be conducted in compliance with applicable laws and regulations or will result in valid and enforceable patents and other intellectual property rights. Additionally, we may not be able to control the publication or other disclosures of research carried out by our licensors relating to technology that could otherwise prove patentable. Pursuant to the terms of some of our license agreements with third parties, some of our third-party licensors have the right, but not the obligation, to control enforcement of our licensed patents or defense of any claims asserting the invalidity of these patents. Even if we are permitted to pursue such enforcement or defense, we will require the cooperation of our licensors, and cannot guarantee that we would receive it and on what terms. We cannot be certain that our licensors will allocate sufficient resources or prioritize their or our prosecution or enforcement of such patents or defense of such claims to protect our interests in the licensed patents. If we cannot obtain patent protection, or enforce existing or future patents against third parties, our competitive position and our financial condition could suffer.
If we fail to comply with our obligations in the agreements under which we license intellectual property rights from third parties or otherwise experience disruptions to our business relationships with our licensors, we could lose license rights that are important to our business.
We hold rights under license or sublicense agreements with third parties that are important to our business. Under our existing license and sublicense agreements, we are subject to various obligations, including diligence obligations with respect to development and commercialization activities and payment obligations. In spite of our efforts, our licensors may conclude that we have materially breached our obligations under such license agreements and terminate the license agreements, thereby removing or limiting our ability to develop and commercialize product candidates and technology covered by these license agreements.
We may need to obtain additional licenses from third parties to advance our research or allow commercialization of our product candidates, such as if we identify new technology that would advance our programs or if an existing license agreement is terminated. We may fail to obtain any of these licenses at a reasonable cost or on reasonable terms, if at all. In that event, we may be required to expend significant time and resources to develop or license replacement technology. If we are unable to do so, we may be unable to develop or commercialize the affected product candidates.
54
Table of Contents
In many cases, patent prosecution of our in-licensed technology is controlled solely by the licensor. If our licensors fail to obtain and maintain patent or other protection for the proprietary intellectual property we license from them, we could lose our rights to the intellectual property or our exclusivity with respect to those rights, and our competitors could market competing products using the intellectual property. In some cases, we control the prosecution of patents resulting from licensed technology. In the event we breach any of our obligations related to such prosecution, we may incur significant liability to our licensing partners and the value of the licensed patents may be adversely affected.
Licensing of intellectual property is of critical importance to our business and involves complex legal, business and scientific issues and is complicated by the rapid pace of scientific discovery in our industry. Disputes may arise regarding intellectual property subject to a licensing agreement, including:
| | the scope of rights granted under the license agreement and other interpretation-related issues; |
| | the extent to which our technology and processes infringe on intellectual property of a licensor that is not subject to the licensing agreement; |
| | the sublicensing of patent and other rights under any collaborative relationships we might enter into in the future; |
| | our diligence obligations under the license agreement and what activities satisfy those diligence obligations; |
| | the ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our partners; and |
| | the priority of invention of patented technology. |
If disputes over intellectual property that we have licensed prevent or impair our ability to maintain our current licensing arrangements on acceptable terms, we may be unable to successfully develop and commercialize the affected product candidates.
Our results of operations will be affected by the level of royalty payments that we are required to pay to third parties.
We are a party to license agreements that require us to remit royalty payments and other payments related to in-licensed intellectual property. Under our in-license agreements, we may pay up-front fees and milestone payments and be subject to future royalties. We cannot precisely predict the amount, if any, of royalties we will owe in the future, and if our calculations of royalty payments are incorrect, we may owe additional royalties, which could negatively affect our results of operations. As our product sales increase, we may, from time to time, disagree with our third-party collaborators as to the appropriate royalties owed, and the resolution of such disputes may be costly, may consume managements time, and may damage our relationship with our collaborators. Furthermore, we may enter into additional license agreements in the future, which may also include royalty, milestone and other payments.
The licenses we may grant to our collaborators and other licensees to use our ddRNAi and other technology may be exclusive to the development of product candidates for certain conditions.
Some of the out-licenses we may grant to our collaborators to use our ddRNAi and other technology may be exclusive to the development of product candidates for certain conditions, so long as our collaborators comply with certain requirements. That means that once our ddRNAi technology is licensed to a collaborator for a specified condition, we are generally prohibited from developing product candidates for that condition and from licensing the ddRNAi to any third party for that condition. The limitations imposed by these exclusive licenses could prevent us from expanding our business and increasing our development of product candidates with new collaborators, both of which could adversely affect our business and results of operations.
55
Table of Contents
Third-party claims of intellectual property infringement may prevent or delay our development and commercialization efforts.
Our commercial success depends in part on our avoiding infringement of the patents and proprietary rights of third parties. There is a substantial amount of litigation, both within and outside the United States, involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries, including patent infringement lawsuits, interferences, oppositions and inter partes review proceedings before the USPTO, and corresponding foreign patent offices. Numerous U.S. and foreign issued patents and pending patent applications, which are owned by third parties, exist in the fields in which we are pursuing development candidates. As the biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that our product candidates may be subject to claims of infringement of the patent rights of third parties.
Third parties may assert that we are employing their proprietary technology without authorization. There may be third-party patents or patent applications with claims to materials, formulations, methods of manufacture or methods for treatment related to the use or manufacture of our product candidates. Because patent applications can take many years to issue, there may be currently pending patent applications which may later result in issued patents that our product candidates may be accused of infringing. In addition, third parties may obtain patents in the future and claim that use of our technologies infringes upon these patents. If any third-party patents were held by a court of competent jurisdiction to cover the manufacturing process, methods of use or formulations of any of our product candidates, any DNA constructs formed during the manufacturing process or any final product itself, the holders of any such patents may be able to block our ability to commercialize such product candidate unless we obtained a license under the applicable patents, or until such patents expire. In either case, such a license may not be available on commercially reasonable terms or at all.
Parties making claims against us may obtain injunctive or other equitable relief, which could effectively block our ability to further develop and commercialize one or more of our product candidates. Defense of these claims, regardless of their merit, would involve substantial litigation expense and would be a substantial diversion of management and employee resources from our business. In the event of a successful claim of infringement against us, we may have to pay substantial damages, including treble damages and attorneys fees for willful infringement, pay royalties, redesign our infringing products or obtain one or more licenses from third parties, which may be impossible or require substantial time and monetary expenditure. Even if a license can be obtained on acceptable terms, the rights may be non-exclusive, which could give our competitors access to the same technology or intellectual property rights licensed to us. If we fail to obtain a required license, we may be unable to effectively market product candidates based on our technology, which could limit our ability to generate revenue or achieve profitability and possibly prevent us from generating revenue sufficient to sustain our operations.
We may not be successful in obtaining or maintaining necessary rights to gene therapy product components and processes for our development pipeline through acquisitions and in-licenses.
Presently, we have certain rights to intellectual property to develop our current gene therapy product candidates. However, our product candidates may require specific formulations to work effectively and efficiently and rights to such formulations may be held by others. In addition, we may need additional intellectual property rights as we develop future product candidates. In particular, we are aware of a third party patent directed to AAV vectors that expires in 2026. In the event we receive regulatory marketing approval before the expiration date it may be necessary for us to obtain a license to the patent in order to commercialize. We cannot guarantee the availability of the license or that it can be obtained on commercially reasonable terms.
We may be unable to acquire or in-license any compositions, methods of use, processes or other third-party intellectual property rights from third parties that we identify on terms that we find acceptable, or at all. The licensing and acquisition of third-party intellectual property rights is a competitive area, and a number of more established companies are also pursuing strategies to license or acquire third-party intellectual property rights
56
Table of Contents
that we may consider attractive. These established companies may have a competitive advantage over us due to their size, cash resources and greater clinical development and commercialization capabilities.
For example, we sometimes collaborate with U.S. and foreign academic institutions to accelerate our preclinical research or development under written agreements with these institutions. Typically, these institutions provide us with an option to negotiate a license to any of the institutions rights in technology resulting from the collaboration. Regardless of such right of first negotiation for intellectual property, we may be unable to negotiate a license within the specified time frame or under terms that are acceptable to us. If we are unable to do so, the institution may offer the intellectual property rights to other parties, potentially blocking our ability to pursue our program.
In addition, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. We also may be unable to license or acquire third-party intellectual property rights on terms that would allow us to make an appropriate return on our investment. If we are unable to successfully obtain rights to required third-party intellectual property rights, our business, financial condition and prospects for growth could suffer.
We may be involved in lawsuits to protect or enforce our patents or the patents of our licensors, which could be expensive, time-consuming and unsuccessful, and issued patents covering our product candidates could be found invalid or unenforceable if challenged in court.
Competitors may infringe our patents or the patents of our licensors. To counter infringement or unauthorized use, we may be required to file infringement claims, which can be expensive and time-consuming. In addition, in an infringement proceeding, a court may decide that a patent of ours or our licensors is not valid, is unenforceable or is not infringed, or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. An adverse result in any litigation or defense proceedings could put one or more of our patents at risk of being invalidated or interpreted narrowly and could put our patent applications at risk of not issuing.
For example, in patent litigation in the United States, defendant counterclaims alleging invalidity or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, for example, lack of novelty, obviousness or non-enablement. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant information from the USPTO, or made a misleading statement, during prosecution. The outcome following legal assertions of invalidity and unenforceability during patent litigation is unpredictable. With respect to the validity question, for example, we cannot be certain that there is no invalidating prior art, of which we and the patent examiner were unaware during prosecution. Third parties may also raise similar claims before administrative bodies in the United States or abroad, even outside the context of litigation. Such mechanisms include re-examination, post grant review, and equivalent proceedings in foreign jurisdictions, such as opposition proceedings. Such proceedings could result in revocation, amendments to our patent claims or statements being made on the record such that our claims may no longer be construed to cover our product candidates. Outcomes or statements on the record in one country could have a disadvantageous effect on prosecution or enforcement of a patent or patent application in another country. The outcome following legal assertions of invalidity and unenforceability is unpredictable. With respect to the validity question, for example, we cannot be certain that no invalidating prior art exists or that the patent examiner was aware of all material prior art during prosecution. Moreover, in some circumstances, we do not have the right to control the preparation, filing and prosecution of patent applications, or to maintain, enforce or defend the patents, covering technology that we license from third parties. Therefore, these patents and applications may not be prosecuted, enforced and defended in a manner consistent with the best interests of our business. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, we could lose at least part, and perhaps all, of the patent protection on one or more of our products or certain aspects of our platform technology. Such a loss of patent protection could have a material adverse impact on our business. Patents and other intellectual property rights also will not protect our technology if competitors design around our protected technology without legally infringing our patents or other intellectual
57
Table of Contents
property rights. Even if resolved in our favor, litigation or other legal proceedings relating to intellectual property claims may cause us to incur significant expenses and could distract our technical and management personnel from their normal responsibilities. In addition, enforcement of a favorable decision by a court can depend on cooperation of a governmental authority which may or may not be available in every jurisdiction. Such litigation or proceedings could substantially increase our operating losses and reduce the resources available for development activities or any future sales, marketing or distribution activities.
For our patents and patent applications filed in the United States before March 16, 2013, interference proceedings provoked by third parties or brought by us may be necessary to determine the priority of inventions with respect to our patents or patent applications or those of our licensors. An unfavorable outcome could require us to cease using the related technology or to attempt to license rights to it from the prevailing party. Our business could be harmed if the prevailing party does not offer us a license on commercially reasonable terms. Litigation or interference proceedings may result in a decision adverse to our interests and, even if successful, may result in substantial costs and distract our management and other employees. We may not be able to prevent, alone or with our licensors, misappropriation of our intellectual property rights, particularly in countries where the laws may not protect those rights as fully as in the United States.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could cause the trading price of our common stock to fall.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties or that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
Certain of our key employees and personnel are or were previously employed at universities, medical institutions or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees, consultants and independent contractors do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that we or our employees, consultants or independent contractors have inadvertently or otherwise used or disclosed intellectual property, including trade secrets or other proprietary information, of any of our employees former employer or other third parties. Litigation may be necessary to defend against these claims. Furthermore, universities or medical institutions who employ some of our key employees and personnel in parallel to their engagement by us may claim that intellectual property developed by such person is owned by the respective academic or medical institution under the respective institution intellectual property policy or applicable law. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in defending against such claims, litigation could result in substantial costs, be a distraction to management and other employees, and damage our relationships with the academic and medical institutions.
We may be subject to claims challenging the inventorship or ownership of our patents and other intellectual property.
We may be subject to claims that former employees, collaborators or other third parties have an ownership interest in our patents or other intellectual property. We may in the future have ownership disputes arising, for example, from conflicting obligations of consultants or others who are involved in developing our product candidates. Litigation may be necessary to defend against these and other claims challenging inventorship or ownership. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights, such as exclusive ownership of, or right to use, valuable intellectual property. Such an outcome could have a material adverse effect on our business. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees.
58
Table of Contents
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance fees, renewal fees, annuity fees and various other governmental fees on patents and applications are required to be paid to the USPTO and various governmental patent agencies outside of the United States in several stages over the lifetime of the patents and applications. The USPTO and various corresponding governmental patent agencies outside of the United States require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process and after a patent has issued. There are situations in which non-compliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction.
Changes in U.S. patent law could diminish the value of patents in general, thereby impairing our ability to protect our product candidates.
As is the case with other biotechnology companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biotechnology industry involve both technological and legal complexity, and it therefore is costly, time-consuming and inherently uncertain. In addition, on September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act includes a number of significant changes to U.S. patent law, including provisions that affect the way patent applications will be prosecuted and may also affect patent litigation.
An important change introduced by the Leahy-Smith Act is that, as of March 16, 2013, the United States transitioned to a first-to-file system for deciding which party should be granted a patent when two or more patent applications are filed by different parties claiming the same invention. A third party that files a patent application in the USPTO after that date but before us could therefore be awarded a patent covering an invention of ours even if we had made the invention before it was made by the third party. This will require us to be cognizant going forward of the time from invention to filing of a patent application. It is not clear what, if any, impact the Leahy-Smith Act will have on the operation of our business. However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents.
Among some of the other changes introduced by the Leahy-Smith Act are changes that limit where a patentee may file a patent infringement suit and providing opportunities for third parties to challenge any issued patent in the USPTO. This applies to all of our U.S. patents, even those issued before March 16, 2013. Because of a lower evidentiary standard in USPTO proceedings compared to the evidentiary standard in U.S. federal court necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a district court action. Accordingly, a third party may attempt to use the USPTO procedures to invalidate our patent claims that would not have been invalidated if first challenged by the third party as a defendant in a district court action.
Recent U.S. Supreme Court rulings such as Association for Molecular Pathology v. Myriad Genetics, Inc. (Myriad I); BRCA1- & BRCA2-Based Hereditary Cancer Test Patent Litig. (Myriad II); and Promega Corp. v. Life Technologies Corp. have also narrowed the scope of patent protection available in certain circumstances and weakened the rights of patent owners in some situations. In addition to increasing uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents and patents that we might obtain in the future.
59
Table of Contents
Our success depends, in part, on our ability to protect our intellectual property and our technologies outside the United States.
Our commercial success depends, in part, on our ability to obtain and maintain patent and trade secret protection for our technologies, our traits, and their uses, as well as our ability to operate without infringing upon the proprietary rights of others outside the United States. If we do not adequately protect our intellectual property, competitors may be able to use our technologies and erode or negate any competitive advantage we may have, which could harm our business and ability to achieve profitability.
Filing, prosecuting and defending patents on product candidates in all countries around the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States can be less extensive than those in the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States. In addition, we may at times in-license third-party technologies for which limited international patent protection exists and for which the time period for filing international patent applications has passed. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Potential competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and further, may export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States. These products may compete with our product candidates, if approved, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, trade secrets and other intellectual property protection, particularly those relating to biotechnology products, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
Risks Related to an Investment in Our Common Sock
The market price and trading volume of our common stock may be volatile and may be affected by economic conditions beyond our control.
The market price of our common stock may be highly volatile and subject to wide fluctuations. In addition, the trading volume of our common stock may fluctuate and cause significant price variations to occur. If the market price of our common stock declines significantly, you may be unable to resell your shares of our common stock at or above your purchase price, if at all. We cannot assure you that the market price of our common stock will not fluctuate or significantly decline in the future.
Some specific factors that could negatively affect the price of our common stock or result in fluctuations in its price and trading volume include:
| | results of our clinical trials; |
| | regulatory actions; |
| | actual or expected fluctuations in our operating results; |
60
Table of Contents
| | changes in market valuations of similar companies; |
| | changes in our key personnel; |
| | changes in financial estimates or recommendations by securities analysts; |
| | strategic decisions by us or our competitors, such as acquisitions, collaborations, divestitures, spin-offs, joint ventures, strategic investments or changes in business strategy; |
| | the passage of legislation or other regulatory developments in the United States and other countries affecting us or our industry |
| | changes in trading volume of our common stock on Nasdaq; |
| | sales of our common stock by us, our executive officers or our shareholders in the future; and |
| | conditions in the financial markets or changes in general economic conditions, including as a result of the current COVID-19 pandemic. |
In addition, the stock market has experienced and is currently experiencing significant volatility, particularly with respect to pharmaceutical, biotechnology and other life sciences company stocks. The volatility of pharmaceutical, biotechnology and other life sciences company stocks often does not relate to the operating performance of the companies represented by the stock.
An active trading market for our common stock may not continue to develop or may not be liquid enough for you to sell your shares of our common stock quickly or at market price.
Although our common stock is listed on Nasdaq, if an active public market in the United States for our common stock does not continue to develop, the market price and liquidity of our common stock may be adversely affected. The price of our common stock may decline, which means you may experience a decrease in the value of your shares of our common stock regardless of our operating performance or prospects. In the past, following periods of volatility in the market price of a companys securities, shareholders often instituted securities class action litigation against that company. If we were involved in a class action suit, it could divert the attention of senior management and, if adversely determined, could cause us significant financial harm.
If securities or industry analysts do not continue to publish research or reports, or if they publish adverse or misleading research or reports, regarding us, our business or our market, our stock price and trading volume could decline.
The trading market for our common stock will be influenced by the research and reports that securities or industry analysts publish about us, our business or our market. If no additional securities or industry analysts commence coverage of us, our stock price could be negatively impacted. If any of the analysts who cover us issue adverse or misleading research or reports regarding us, our business model, our intellectual property, our stock performance or our market, or if our operating results fail to meet the expectations of analysts, our stock price would likely decline. If one or more of these analysts cease coverage of us or fail to publish reports on us regularly, we could lose visibility in the financial markets, which in turn could cause our stock price or trading volume to decline.
If we fail to establish and maintain proper internal financial reporting controls, our ability to produce accurate financial statements or comply with applicable regulations could be impaired.
Section 404(a) of the Sarbanes-Oxley Act requires that our management assess and report annually on the effectiveness of our internal controls over financial reporting and identify any material weaknesses in our internal controls over financial reporting. Although Section 404(b) of the Sarbanes-Oxley Act requires our independent registered public accounting firm to issue an annual report that addresses the effectiveness of our internal
61
Table of Contents
controls over financial reporting, we rely on the exemption provided in the JOBS Act, and consequently will not be required to comply with SEC rules that implement Section 404(b) of the Sarbanes-Oxley Act until such time as we are no longer an emerging growth company and are an accelerated or large accelerated filer.
The presence of any material weaknesses in our internal control over financial reporting could result in financial statement errors which, in turn, could lead to errors in our financial reports and/or delays in our financial reporting, which could require us to restate our operating results. We might not identify one or more material weaknesses in our internal controls in connection with evaluating our compliance with Section 404(a) of the Sarbanes-Oxley Act. In order to maintain and improve the effectiveness of our disclosure controls and procedures and internal controls over financial reporting, we will need to expend significant resources and provide significant management oversight. Implementing any appropriate changes to our internal controls may require specific compliance training of our directors and employees, entail substantial costs in order to modify our existing accounting systems, take a significant period of time to complete and divert managements attention from other business concerns. These changes may not, however, be effective in maintaining the adequacy of our internal control.
If we are unable to conclude that we have effective internal controls over financial reporting, investors may lose confidence in our operating results, the price of our common stock could decline and we may be subject to litigation or regulatory enforcement actions. In addition, if we are unable to meet the requirements of Section 404 of the Sarbanes-Oxley Act, our common stock may not be able to remain listed on Nasdaq.
We have never declared or paid dividends on our common stock and we do not anticipate paying dividends in the foreseeable future.
We have never declared or paid cash dividends on our common stock. For the foreseeable future, we currently intend to retain all available funds and any future earnings to support our operations and to finance the growth and development of our business. Any future determination to declare cash dividends will be made at the discretion of our Board, subject to compliance with applicable laws and covenants under current or future credit facilities, which may restrict or limit our ability to pay dividends, and will depend on our financial condition, operating results, capital requirements, general business conditions and other factors that our Board may deem relevant. We do not anticipate paying any cash dividends on our common stock in the foreseeable future. As a result, a return on your investment in our securities will only occur if the price of our common stock appreciates.
Future sales and issuances of our common stock or rights to purchase common stock could result in substantial dilution to the percentage ownership of our stockholders.
We expect that significant additional capital will be needed in the future to continue our planned operations. To raise capital, we may sell common stock or other securities convertible into or exchanged for our common stock in one or more transactions, and in a manner we determine from time to time and at prices that may not be the same as the price per share paid by other investors, and dilution to our stockholders could result. The price per share at which we sell additional shares of our common stock, or securities convertible or exchangeable into common stock, in future transactions may be higher or lower than the price per share paid by other investors. New investors could also receive rights, preferences and privileges senior to those of existing holders of our common stock. In addition, in the event of stock dividends, stock splits, reorganizations or similar events affecting our common stock, we may be required to proportionally adjust the conversion price, exercise price or number of shares issuable upon exercise of any outstanding warrants.
We are an emerging growth company as defined in the JOBS Act and the reduced disclosure requirements applicable to emerging growth companies may make our common stock less attractive to investors and, as a result, adversely affect the price of our common stock and result in a less active trading market for our common stock.
We are an emerging growth company as defined in the JOBS Act, and we may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not
62
Table of Contents
emerging growth companies. For example, we have elected to rely on an exemption from the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act relating to internal control over financial reporting, and we will not provide such an attestation from our auditors for so long as we qualify as an emerging growth company. We have also elected to rely on an exemption that permits an emerging growth company to include only two years of audited financial statements and only two years of related managements discussion and analysis of financial condition and results of operations disclosure, and we have therefore only included two years of audited financial statements, selected financial data and managements discussion and analysis of financial condition and results of operations in this prospectus.
We may avail ourselves of these disclosure exemptions until we are no longer an emerging growth company. We cannot predict whether investors will find our common stock less attractive because of our reliance on some or all of these exemptions. If investors find our common stock less attractive, it may cause the trading price of our common stock to decline and there may be a less active trading market for our common stock.
We will cease to be an emerging growth company upon the earliest of:
| | the end of the fiscal year in which the fifth anniversary of the completion of our initial public offering occurs; |
| | the end of the first fiscal year in which the market value of our ordinary shares held by non-affiliates exceeds $700 million as of the end of the second quarter of such fiscal year; |
| | the end of the first fiscal year in which we have total annual gross revenues of at least $1.07 billion; and |
| | the date on which we have issued more than $1 billion in non-convertible debt securities in any rolling three-year period. |
Our corporate governance structure may prevent our acquisition by another company at a premium over the public trading price of our shares.
It is possible that the acquisition of a majority of our outstanding voting stock by another company could result in our stockholders receiving a premium over the public trading price for our shares. Provisions of our restated certificate of incorporation and our amended and restated bylaws, each as amended, and of Delaware corporate law could delay or make more difficult an acquisition of our company by merger, tender offer or proxy contest, even if it would create an immediate benefit to our stockholders. Furthermore, our certificate of incorporation also provides for a classified board of directors with directors divided into three classes serving staggered terms. These provisions may have the effect of delaying or preventing a change in control of us without action by our stockholders and, therefore, could adversely affect the price of our stock or the possibility of sale of shares to an acquiring person.
As a result of the Re-domiciliation, we are required to comply with the domestic reporting regime under the Exchange Act and will incur significant legal, accounting and other expenses, and our management will be required to devote substantial additional time to new compliance initiatives and corporate governance matters.
As a result of the Re-domiciliation, we no longer qualified as a foreign private issuer under the rules and regulations of the SEC. While we were a foreign private issuer, we were exempt from compliance with certain laws and regulations of the SEC, including the proxy rules, the short-swing profits recapture rules and certain governance requirements, such as independent director oversight of the nomination of directors and executive compensation. In addition, we were not required to file annual, quarterly and current reports and financial statements with the SEC as frequently or as promptly as U.S. companies registered under the Exchange Act. As a result of becoming a U.S. domestic company, we are no longer entitled to foreign private issuer exemptions and must report as a domestic U.S. filer, including filing quarterly reports on Form 10-Q, current reports on Form 8-K and proxy statements under Section 14 of the Exchange Act. In addition, our insiders are now subject to
63
Table of Contents
the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act and we are no longer exempt from the requirements of Regulation FD promulgated by the SEC under the Exchange Act. Moreover, as a domestic filer, we are required to comply with the corporate governance obligations imposed by Nasdaq and no longer have the option to follow our home country rules in lieu of such obligations.
The regulatory and compliance costs associated with the reporting and governance requirements applicable to U.S. domestic issuers may be significantly higher than the costs we previously incurred as a foreign private issuer. As a result, we expect that the loss of foreign private issuer status will increase our legal and financial compliance costs and will make some activities highly time-consuming and costly. In addition, we need to develop our reporting and compliance infrastructure and may face challenges in complying with the new requirements applicable to us.
The market price and trading volume of our common stock may be volatile and may be affected by economic conditions beyond our control.
The market price of our common stock may be highly volatile and subject to wide fluctuations. In addition, the trading volume of our common stock may fluctuate and cause significant price variations to occur. If the market price of our common stock declines significantly, you may be unable to resell your shares of common stock at or above your purchase price, if at all. We cannot assure you that the market price of our common stock will not fluctuate or significantly decline in the future.
Some specific factors that could negatively affect the price of our common stock or result in fluctuations in their price and trading volume include:
| | actual or expected fluctuations in our operating results; |
| | changes in market valuations of similar companies; |
| | changes in our key personnel; |
| | changes in financial estimates or recommendations by securities analysts; |
| | changes in trading volume of shares of our common stock on Nasdaq; |
| | sales of shares of our common stock by us, our executive officers or our shareholders in the future; and |
| | conditions in the financial markets or changes in general economic conditions. |
Although we are required to use our reasonable best efforts to have an effective registration statement covering the issuance of shares of common stock underlying certain of our outstanding warrants at the time that holders of our warrants exercise their warrants, we cannot guarantee that a registration statement will be effective, in which case holders of our warrants are entitled to a cashless exercise of their warrants.
Pursuant to the terms of certain of our warrants, we are obligated to have an effective registration statement covering the resale of the shares of common stock underlying such warrants. If no registration is effective at the time a warrant holder seeks to exercise their warrants, we would be obligated to issue shares to such warrant holder in a cashless exercise in exchange for such holders warrants, in which case we would not receive the cash that we would otherwise receive in an exercise of warrants for cash.
Risks Relating to this Offering
You will experience immediate dilution as a result of this offering and may experience additional dilution in the future.
The public offering price for shares of our common stock (or pre-funded warrants in lieu thereof) offered hereby will be substantially higher than the net tangible book value per share of our common stock immediately
64
Table of Contents
after this offering. If you purchase common stock (or pre-funded warrants in lieu thereof) in this offering, you will incur substantial and immediate dilution in the net tangible book value of your investment. Net tangible book value per share represents the amount of total tangible assets less total liabilities, divided by the number of shares of our common stock then outstanding. To the extent that options that are currently outstanding are exercised, there will be further dilution to your investment. Exercise of the pre-funded warrants will also dilute your investment. We may also issue additional common stock, options and other securities in the future that may result in further dilution of your shares of our common stock. See Dilution for a calculation of the extent to which your investment will be diluted.
There is no public market for the pre-funded warrants being offered by us in this offering.
There is no established public trading market for the pre-funded warrants being offered in this offering, and we do not expect a market to develop. In addition, we do not intend to apply to list the pre-funded warrants on any national securities exchange or other nationally recognized trading system, including The Nasdaq Capital Market. Without an active market, the liquidity of the pre-funded warrants will be limited.
Holders of the pre-funded warrants offered hereby will have no rights as common stockholders with respect to the common stock underlying the pre-funded warrants until such holders exercise their pre-funded warrants and acquire our common stock, except as otherwise provided in the pre-funded warrants.
Until holders of the pre-funded warrants acquire our common stock upon exercise thereof, such holders will have no rights with respect to the common stock underlying such pre-funded warrants, except to the extent that holders of such pre-funded warrants will have certain rights to participate in distributions or dividends paid on our common stock as set forth in the pre-funded warrants. Upon exercise of the pre-funded warrants, the holders will be entitled to exercise the rights of a common stockholder only as to matters for which the record date occurs after the exercise date.
Future sales of our common stock, or the perception that such sales may occur, could depress the trading price of our common stock.
After the completion of this offering, we expect to have shares ( shares if the underwriter exercises its option to purchase additional shares in full) of our common stock outstanding, which may be resold in the public market immediately after this offering. We and all of our directors and executive officers have signed lock-up agreements for a period of 90 days following the date of this prospectus, subject to specified exceptions. See Underwriting.
The underwriter may, in its sole discretion and without notice, release all or any portion of the shares of our common stock subject to lock-up agreements. As restrictions on resale end, the market price of our common stock could drop significantly if the holders of these shares of our common stock sell them or are perceived by the market as intending to sell them. These factors could also make it more difficult for us to raise additional funds through future offerings of our common stock or other securities.
We have broad discretion in the use of the net proceeds we receive from this offering and may not use them effectively.
Our management will have broad discretion in the application of the net proceeds we receive in this offering, including for any of the purposes described in the section entitled Use of Proceeds, and you will not have the opportunity as part of your investment decision to assess whether our management is using the net proceeds appropriately. Because of the number and variability of factors that will determine our use of our net proceeds from this offering, their ultimate use may vary substantially from their currently intended use. The failure by our management to apply these funds effectively could result in financial losses that could have a material adverse effect on our business and cause the price of our common stock to decline. Pending their use, we may invest our net proceeds from this offering in short-term, investment-grade, interest-bearing securities. These investments may not yield a favorable return to our stockholders.
65
Table of Contents
We estimate that the net proceeds from this offering will be approximately $ million, or $ million if the underwriter exercises its option to purchase additional shares of common stock in full, assuming a public offering price of $ per share, the last reported sale price per share of our common stock on , 2020 as reported on Nasdaq, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and assuming no sale of any pre-funded warrants in this offering. The public offering price per share will be determined between us, the underwriter and investors based on market conditions at the time of pricing and may be at a discount to the current market price of our common stock.
Each $ increase or decrease in the assumed public offering price of $ per share would increase or decrease the net proceeds from this offering by $ million, assuming the number of shares and pre-funded warrants offered by us, as set forth on the cover of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. We may also increase or decrease the number of shares and pre-funded warrants we are offering. An increase of decrease of in the number of shares (or the common stock underlying the pre-funded warrants) offered by us, as set forth on the cover of this prospectus, would increase or decrease the net proceeds from this offering by approximately $ million, assuming the assumed public offering price per share remains the same, and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. We do not expect that a change by these amounts in either the public offering price per share or in the number of shares offered by us would have a material effect on our use of the proceeds from this offering, although it may accelerate the time at which we will need to seek additional capital.
We intend to use the net proceeds from this financing for the continued advancement of development activities for our product pipeline, general corporate purposes and strategic growth opportunities.
The amount and timing of these expenditures will depend on a number of factors, including the progress of our research and development efforts, the progress of any partnering efforts, technological advances and the competitive environment for our product candidates. Accordingly, you will be relying on the judgment of our management with regard to the use of these net proceeds, and you will not have the opportunity, as part of your investment decision, to assess whether the proceeds are being used appropriately. It is possible that the proceeds will be used in a way that does not yield a favorable, or any, return for us. Pending application of the net proceeds as described above, we intend to invest the proceeds in investment grade interest bearing instruments, or will hold the proceeds in interest bearing or non-interest bearing bank accounts.
66
Table of Contents
The following table sets forth our cash and cash equivalents and capitalization, each as of March 31, 2020:
| | on an actual basis; and |
| | on a pro forma basis, as adjusted to give effect to the issuance of shares of common stock in the offering at an assumed public offering price of $ per share, the last reported sale price per share of our common stock on , 2020, as reported on Nasdaq, assuming no sale of any pre-funded warrants in this offering and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
You should consider this table in conjunction with our financial statements and the notes to those financial statements included in this prospectus. The pro forma as adjusted information set forth in the table below is illustrative only and will be adjusted based on the actual public offering price and other terms of this offering determined at pricing.
| As of March 31, 2020 | ||||||||
| (Unaudited) | ||||||||
| (in thousands) | Actual | Pro Forma, As Adjusted(1) |
||||||
| Cash and cash equivalents |
$ | 11,400 | $ | |||||
| Total debt obligations |
1,244 | |||||||
|
|
|
|
|
|||||
| Stockholders equity: |
||||||||
| Common stock, par value $0.0001 per share: |
||||||||
| 10,000,000 shares authorized; 1,070,957 actual; pro forma |
1 | |||||||
| Additional paid-in capital |
128,897 | |||||||
| Accumulated deficit |
(113,829 | ) | ||||||
|
|
|
|
|
|||||
| Accumulated other comprehensive loss |
(2,733 | ) | ||||||
|
|
|
|
|
|||||
| Total stockholders equity |
12,336 | |||||||
|
|
|
|
|
|||||
| Total capitalization |
$ | 13,580 | $ | |||||
| (1) | The number of shares in the table above is based on 1,070,957 shares of common stock outstanding as of March 31, 2020 and does not include: |
| | 72,384 shares of common stock issuable upon exercise of stock options outstanding as of March 31, 2020 at a weighted-average exercise price of $63.87 per share; and |
| | 252,516 shares of common stock issuable upon the exercise of warrants exercisable for shares of common stock outstanding as of March 31, 2020 at a weighted-average exercise price of $21.43 per share. |
67
Table of Contents
Dilution in net tangible book value per share to new investors is the amount by which the effective public offering price paid by the purchasers of the shares sold in this offering exceeds the as adjusted net tangible book value per share of common stock after giving effect to the offering. Net tangible book value per share is determined by dividing our total tangible assets (total assets less intangible assets), less total liabilities, by the number of shares of our common stock outstanding. Our net tangible book value as of March 31, 2020 was approximately $11.9 million, or approximately $11.11 per share of common stock.
After giving effect to the issuance of shares of common stock in this offering at an assumed public offering price of $ per share, the last reported sale price per share of our common stock on , 2020 as reported on Nasdaq, assuming no sale of any pre-funded warrants in this offering and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us, our as adjusted net tangible book value as of March 31, 2020 would have been approximately $ million, or approximately $ per share of common stock. This represents an immediate increase in our as adjusted net tangible book value of $ per share to our existing stockholders and an immediate dilution of $ per share to investors participating in this offering. The following table illustrates this dilution on a per share basis:
| Assumed public offering price per share |
$ | |||||||
| Net tangible book value per share as of March 31, 2020 |
$ | 11.9 | ||||||
| Increase in net tangible book value per share attributable to this offering |
||||||||
|
|
|
|||||||
| As adjusted net tangible book value per share after giving effect to this offering |
||||||||
|
|
|
|||||||
| Dilution per share to new investors |
$ | |||||||
|
|
|
Each $ increase or decrease in the assumed public offering price per share of $ , the last reported sale price of our common stock on the Nasdaq Capital Market on , 2020 would increase or decrease the net proceeds to us from this offering by $ million, increase or decrease our as adjusted net tangible book value per share after this offering by $ , and increase or decrease the dilution per share to new investors purchasing in this offering by $ , assuming that the number of shares of common stock offered by us, as set forth on the cover page of this prospectus, remains the same, assuming no sale of any pre-funded warrants and after deducting the estimated underwriter discounts and commissions and estimated offering expenses payable by us. We may also increase or decrease the number of shares of common stock and pre-funded warrants offered in this offering. Each increase or decrease of in the number of shares (or the common stock underlying the pre-funded warrants) offered by us would increase or decrease the net proceeds to us by approximately $ million, increase or decrease our as adjusted net tangible book value per share after this offering by $ , and increase or decrease the dilution per share to new investors purchasing this offering by $ , assuming the assumed public offering price per share of $ remains the same, assuming no sale of any pre-funded warrants, after deducting the underwriting discounts and commissions and estimated offering expenses payable by us.
If the underwriter exercises its option to purchase additional shares of common stock in full, our as adjusted net tangible book value would be approximately $ per share, an increase in adjusted net tangible book value of approximately $ per share to existing stockholders and a decrease of approximately $ per share in the dilution to new investors in this offering, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us.
The dilution information discussed above is illustrative only and will change based on the actual public offering price and other terms of this offering determined at pricing.
68
Table of Contents
The foregoing discussion and table are based on 1,070,957 shares of common stock outstanding at March 31, 2020 and excludes as of such date the following:
| | 72,384 shares of common stock issuable upon exercise of stock options outstanding at March 31, 2020 at a weighted-average exercise price of $63.87 per share; and |
| | 252,516 shares of common stock issuable upon exercise of warrants at a weighted-average exercise price of $21.43 per share. |
69
Table of Contents
MARKET PRICE OF OUR COMMON STOCK AND RELATED STOCKHOLDER MATTERS
Our common stock trades on Nasdaq under the symbol BNTC. Prior to the Re-domiciliation, the American Depositary Shares of Benitec Limited traded on Nasdaq under the same trading symbol and the ordinary shares of Benitec Limited traded on the Australian Stock Exchange (ASX). In connection with the Re-domiciliation, Benitec Limiteds ordinary shares were delisted from the ASX. On August 13, 2020, the closing sale price of our common stock as reported on the Nasdaq was $8.31 per share.
As of August 10, 2020, we had approximately 3,376 record holders of our common stock. The number of record holders is based on the actual number of holders registered on the books of our transfer agent and does not reflect holders of shares in street name or persons, partnerships, associations, corporations or other entities identified in security position listings maintained by depository trust companies.
We never have declared or paid any cash dividends on our capital stock. Currently, we anticipate that we will retain all available funds for use in the operation and expansion of our business and do not anticipate paying any cash dividends for the foreseeable future. Any future determination relating to dividend policy will be made at the discretion of our Board and will depend on our future earnings, capital requirements, financial condition, prospects, applicable Delaware law, which provides that dividends are only payable out of surplus or current net profits, and other factors that our Board deems relevant.
70
Table of Contents
MANAGEMENTS DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
You should read the following discussion and analysis of financial condition and operating results together with our consolidated financial statements and the related notes and other financial information included elsewhere in this prospectus. This discussion contains forward-looking statements that involve risks and uncertainties. As a result of many factors, such as those set forth in the section of the prospectus captioned Risk Factors and elsewhere in this prospectus, our actual results may differ materially from those anticipated in these forward-looking statements.
Overview
We endeavor to become the leader in discovery, development, and commercialization of therapeutic agents capable of addressing significant unmet medical need via the application of the silence and replace approach to the treatment of genetic disorders.
Benitec Biopharma, Inc. (Benitec or the Company or in the third person, we or our) is a development-stage biotechnology company focused on the advancement of novel genetic medicines with headquarters in Hayward, California. The proprietary platform, called DNA-directed RNA interference, or ddRNAi, combines RNA interference, or RNAi, with gene therapy to create medicines that facilitate sustained silencing of disease-causing genes following a single administration. The Company is developing ddRNAi-based therapeutics for chronic and life-threatening human conditions including Oculopharyngeal Muscular Dystrophy (OPMD), and Chronic Hepatitis B.
BB-301 is the most advanced ddRNAi-based genetic medicine currently under development by Benitec. BB-301 is an internally optimized, AAV-based gene therapy agent that is designed to both silence the expression of mutated, disease-causing genes (to slow, or halt, the underlying mechanism of disease progression) and replace the mutant genes with normal, wild type genes (to drive restoration of function in diseased cells). This fundamental approach to disease management is called silence and replace and this biological mechanism offers the potential to restore the underlying physiology of the treated tissues and, in the process, improve treatment outcomes for patients suffering from the chronic and, potentially, fatal effects of Oculopharyngeal Muscular Dystrophy (OPMD). BB-301 has been granted Orphan Drug Designation in the United States and the European Union.
Through the combination of the targeted gene silencing effects of RNAi and the durable transgene expression achievable via the use of modified viral vectors, the silence and replace approach has the potential to produce long-term silencing of disease-causing genes along with simultaneous replacement of wild type gene function following a single administration of the proprietary genetic medicine. We believe this novel attribute of the investigational agents under development by Benitec may facilitate the achievement of robust clinical activity while greatly reducing the dosing frequencies traditionally expected for medicines employed for the management of chronic diseases. Additionally, the establishment of chronic gene silencing and gene replacement may significantly reduce the risk of patient non-compliance during the course of medical management of potentially fatal clinical disorders.
Unless otherwise indicated, all dollar amounts in this section are provided in thousands.
Re-domiciliation
On April 15, 2020, or the Implementation Date, the Re-domiciliation of Benitec Limited, a public company incorporated under the laws of the State of Western Australia, or Benitec Limited, was completed in accordance with the Scheme Implementation Agreement, as amended and restated as of January 30, 2020, between Benitec Limited and us. As a result of the Re-domiciliation, the jurisdiction of incorporation was changed from Australia to Delaware, and Benitec Limited became our wholly owned subsidiary.
71
Table of Contents
The Re-domiciliation was effected pursuant to a statutory scheme of arrangement under Australian law, or the Scheme, whereby on the Implementation Date, all of the issued and outstanding ordinary shares of Benitec Limited were exchanged for newly issued shares of our common stock, on the basis of one share of our common stock, par value $0.0001 per share, for every 300 ordinary shares of Benitec Limited issued and outstanding. Holders of Benitec Limiteds American Depository Shares, or ADSs (each of which represented 200 ordinary shares), received two shares of our common stock for every three ADSs held.
COVID-19
In December 2019, an outbreak of a novel strain of coronavirus was identified in Wuhan, China. This virus continues to spread globally, has been declared a pandemic by the World Health Organization and has spread to nearly every country, including Australia and the United States. The impact of this pandemic has been and will likely continue to be extensive in many aspects of society, which has resulted in and will likely continue to result in significant disruptions to businesses and capital markets around the world. The extent to which the coronavirus impacts us will depend on future developments, which are highly uncertain and cannot be predicted, including new information which may emerge concerning the severity of the coronavirus and the actions to contain the coronavirus or treat its impact, among others.
Certain of our research and development efforts are conducted globally, including the ongoing development of our silence and replace therapeutic for the treatment of Oculopharyngeal Muscular Dystrophy (OPMD), and will be dependent upon our ability to initiate preclinical and clinical studies despite the ongoing COVID-19 pandemic. As we continue to actively advance our preclinical programs, including our ongoing tissue transduction studies for BB-301, we are in close contact with our principal investigators and preclinical trial sites, which are primarily located in the France, and are assessing the impact of COVID-19 on our studies and the expected development timelines and costs of all of our product candidates, on an ongoing basis. In light of recent developments relating to the COVID-19 global pandemic, the focus of healthcare providers and hospitals on fighting the virus, and consistent with the FDAs updated industry guidance for conducting clinical trials issued on March 18, 2020, we have experienced delays to the original timeline regarding the initiation and anticipated completion of the ongoing BB-301 IND-enabling development work. The initiation of the BB-301 tissue transduction study, which represents a key component of the IND-enabling work, was delayed by several months, however, the study has been recently initiated and the dosing of the initial preclinical cohorts has proceeded without incident. We will continue to evaluate the impact of the COVID-19 pandemic on our business and expect to reevaluate the timing of our anticipated preclinical and clinical milestones as we learn more and the impact of COVID-19 on our industry becomes more clear.
We had also implemented work-from-home measures for the majority of our employees between March 2020 and June 2020, resulting in a reduction of laboratory work and a halt of non-essential business travel. As we transition our employees back to our premises, there is a risk that COVID-19 infections occur at our offices or laboratory facilities and significantly affect our operations. Additionally, if any of our critical vendors are impacted, our business could be affected if we become unable to timely procure essential equipment, supplies or services in adequate quantities and at acceptable prices.
Axovant Termination
Benitecs License and Collaboration Agreement, dated July 9, 2018, with Axovant Sciences GmbH, or Axovant, was terminated as of September 3, 2019. As a result, all rights and licenses which Benitec had granted to Axovant to develop and commercialize BB-301 and related gene therapy product candidates terminated.
Prior to such termination, the Benitec team endeavored to conduct several additional exploratory nonclinical analyses in order to potentially improve the biological efficacy of BB-301 via further optimization of the route of administration employed to dose the target muscle tissues.
72
Table of Contents
Nonclinical data derived from in vivo evaluations of BB-301 in two distinct large animal species suggested the existence of an opportunity to further improve the biological efficacy of the compound via additional optimization of the proprietary delivery method employed to dose key target tissues that underlie the morbidity and mortality associated with the natural history of OPMD. The initial biological efficacy profile observed for BB-301 following in vivo testing in the A17 mouse model of OPMD, including full correction of the disease phenotype, remained unchanged. However, the Benitec management team desired to complete a series of exploratory analyses prior to the formal IND filing and the subsequent initiation of clinical testing.
Completion of the experimental work noted above would have delayed the initiation of the BB-301 clinical study beyond the timelines that were initially outlined by Axovant following the execution of the License and Collaboration Agreement between Benitec and Axovant. As such, Axovant elected to terminate the License and Collaboration Agreement between Benitec and Axovant, and all rights and licenses granted to Axovant terminated, including the rights to BB-301, which was in preclinical development for the treatment of OPMD, and all other early stage research collaboration programs that were governed by the agreement.
Nonclinical Programs
Nonclinical research efforts supporting the development of ddRNAi-based therapeutic agents and silence and replace-based therapeutic agents targeting the treatment of Chronic Hepatitis B Virus Infection (HBV) and Age-Related Macular Degeneration (AMD) have concluded and are no longer being continued by the Company.
Workforce Reduction
On July 31, 2019, Benitec announced the completion of a workforce reduction of approximately 50%. Through this streamlining of operations, the Company retained staff members who are key to the achievement of the core research and development goals. The rationalization of resources were deemed to support an extended financial runway for the Company while allowing Benitec to continue to advance the BB-301 program.
Royalties, milestone payments and other license fees
We are required to pay royalties, milestone payments and other license fees in connection with our licensing of intellectual property from third parties, including as discussed below.
In December 2016, we entered into an exclusive sublicense agreement with NantWorks, LLC, pursuant to which we agreed to make certain milestone and royalty payments, as well as periodic payments for so long as the agreement remained in effect. We terminated the exclusive sublicense agreement for convenience, with the termination effective as of June 2020.
We have collaborated with Biomics Biotechnologies Co., Ltd., or Biomics, pursuant to several collaboration agreements in relation to single-stranded RNA and shRNA sequences for treatment of hepatitis B. In July 2015, we entered into an earn-out agreement with Biomics which confirmed Benitecs ownership of certain patents resulting from the collaboration in exchange for an upfront payment and equity issuance to Biomics and a share of certain future licensing revenue received by Benitec.
Foreign Currency Translation and Other Comprehensive Income (Loss)
The Companys functional currency is the Australian dollar (AUD). For financial reporting purposes, the Australian dollar has been translated into United States dollar ($) and/or (USD) as the reporting currency. Assets and liabilities are translated at the exchange rate in effect at the balance sheet date. Revenues and expenses are translated at the average rate of exchange prevailing during the reporting period. Equity transactions are
73
Table of Contents
translated at each historical transaction date spot rate. Translation adjustments arising from the use of different exchange rates from period to period are included as a component of stockholders equity as Accumulated other comprehensive income (loss). Gains and losses resulting from foreign currency transactions are included in the statements of operations and comprehensive income (loss) as other comprehensive income (loss).
Other Comprehensive Income (Loss) for all periods presented includes only foreign currency translation gains (losses).
Results of Operations
Revenues
The Company currently generates revenue from our operations through two activities: revenue from customers and revenue from government research and development grants. The Company has not generated any revenues from the sales of products. Revenues from licensing fees and interest income are included in the revenue from customers line item on our statements of operations and comprehensive income (loss). The Research and Development Tax Incentive is recognized as Government research and development grants.
Our licensing fees have been generated through the licensing of our ddRNAi technology to biopharmaceutical companies, and in the fiscal year-ended June 30, 2019, revenue was generated through a Licensing and Collaboration Agreement with Axovant Sciences (the Axovant Agreement).
The following tables set forth a summary of our revenues for each of the periods set forth below:
| Year Ended June 30, | ||||||||
| 2019 | 2018 | |||||||
| (US$000) | ||||||||
| Revenues: |
||||||||
| Revenues from customers |
$ | 11,551 | $ | 292 | ||||
| Government research and development grants |
648 | 3,098 | ||||||
|
|
|
|
|
|||||
| Total revenues |
$ | 12,199 | $ | 3,390 | ||||
|
|
|
|
|
|||||
| Three Months Ended March 31, |
Nine Months Ended March 31, |
|||||||||||||||
| 2020 | 2019 | 2020 | 2019 | |||||||||||||
| (unaudited) | ||||||||||||||||
| (US$000) | ||||||||||||||||
| Revenues: |
||||||||||||||||
| Revenues from customers |
$ | 28 | $ | 449 | $ | 137 | $ | 10,955 | ||||||||
| Government research and development grants |
| 111 | | 601 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total revenues |
$ | 28 | $ | 560 | $ | 137 | $ | 11,556 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
Revenues from customers
On July 9, 2018, the Company entered into the Axovant Agreement. The Axovant Agreement granted Axovant Sciences an exclusive worldwide license to develop, manufacture, and commercialize products containing the Companys product known as BB-301, which was designed for the potential treatment of Oculopharyngeal Muscular Dystrophy. Service revenue consists of payments for services provided to Axovant Sciences pursuant to the Axovant Agreement. On June 6, 2019, the termination of the Axovant Agreement was announced. The termination of the Axovant Agreement was effective as of September 3, 2019. The termination discharges all future performance obligations at termination date under the contract.
74
Table of Contents
During the year ended June 30, 2019, the Company recognized $11,551 in customer revenues, as compared to $292 for the comparable year ended June 30, 2018. The increase in revenues from customers is due to the company entering into to the contract with Axovant Sciences.
During the three and nine months ended March 31, 2020, the Company recognized $28 and $137 in customer revenues, respectively, as compared to $449 and $10,955 for the comparable three and three and nine months ended March 31, 2019, respectively. The decrease in revenues from customers is due to the termination of the Axovant Sciences contract on June 6, 2019, effective as of September 3, 2019.
Government research and development grants
The Company has historically received, but is currently not receiving, grants through the Australian federal governments Research and Development Tax Incentive program, under which the government provides a cash refund for the 43.5% of eligible research and development expenditures by small Australian entities, which are defined as Australian entities with less than A$20 million in revenue, having a tax loss. The Research and Development Tax Incentive grant is made by the Australian federal government for eligible research and development purposes based on the filing of an annual application. Prior to the Re-domiciliation, this grant was available for our research and development activities in Australia, as well as activities in the United States to the extent such U.S.-based expenses relate to our activities in Australia, did not exceed half the expenses for the relevant activities and were approved by the Australian government. Grants are recorded when a reliable estimate can be made.
During the year ended June 30, 2019, we recognized $648 in government research and development grants, as compared $3,098 for the comparable year ended June 30, 2018. The decrease in grant revenue is due to excluding the OPMD program from the R&D claim of the grant from the Australian government. Further, the company no longer continued the nonclinical research efforts targeting the treatment of Chronic Hepatitis B Virus Infection (HBV) and Age-Related Macular Degeneration (AMD).
During the three and nine months ended March 31, 2020, we recognized $0 and $0 in government research and development grant revenue, as compared to $111 and $601 for the comparable three and nine month periods ended March 31, 2019, respectively. The decrease in grant revenue is a result of Benitec no longer claiming the grant from the Australian government due to the planned re-domiciliation of Benitec to the United States of America.
Research and Development Expenses
Research and development expenses relate primarily to the cost of conducting clinical and pre-clinical trials. Clinical development costs are a significant component of research and development expenses. Estimates have been used in determining the expense liability under certain clinical trial contracts where services have been performed but not yet invoiced. Generally, the costs, and therefore estimates, associated with clinical trial contracts are based on the number of patients, drug administration cycles, the type of treatment and the outcome being the length of time before actual amounts can be determined will vary depending on length of the patient cycles and the timing of the invoices by the clinical trial partners.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries, related benefits, travel, and equity-based compensation expense. General and administrative expenses also include facility expenses, professional fees for legal, consulting, accounting and audit services and other related costs.
We anticipate that our general and administrative expenses may increase as the Company focuses on the continued development of the pre-clinical OPMD program. The Company also anticipates an increase in
75
Table of Contents
expenses relating to accounting, legal and regulatory-related services associated with maintaining compliance with the exchange listing and the SEC requirements, director and officer insurance premiums and other costs associated with being a public company.
Expenses
The following tables set forth a summary of our expenses for each of the periods set forth below:
| Year Ended June 30, | ||||||||
| 2019 | 2018 | |||||||
| (US$000) | ||||||||
| Expenses: |
||||||||
| Royalties and license fees |
$ | 435 | $ | 349 | ||||
| Research and development |
4,567 | 7,704 | ||||||
| General and Administrative |
4,614 | 4,539 | ||||||
|
|
|
|
|
|||||
| Total expenses |
9,616 | 12,592 | ||||||
|
|
|
|
|
|||||
| Three Months Ended March 31, |
Nine Months Ended March 31, |
|||||||||||||||
| 2020 | 2019 | 2020 | 2019 | |||||||||||||
| (unaudited) | ||||||||||||||||
| (US$000) | ||||||||||||||||
| Expenses: |
||||||||||||||||
| Royalties and license fees |
$ | 47 | $ | 30 | $ | (233 | ) | $ | 405 | |||||||
| Research and development |
805 | 1,159 | 2,095 | 3,304 | ||||||||||||
| General and Administrative |
1,287 | 1,103 | 3,669 | 3,754 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total expenses |
2,139 | 2,292 | 5,531 | 7,463 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
During the year ended June 30, 2019, we incurred $435 in royalties and license fees, as compared $349 for the comparable year ended June 30, 2018. The increase in royalties and license fees during 2019 was due to an increase in the NantWorks license fee related to BB-401 which was offset by a decrease in the research license fee for our in connection with short hairpin RNA. However, in contrast, during 2018 the NantWorks fee decreased and our research license fees increased.
During the three and nine months ended March 31, 2020, we incurred $47 and $(233) in royalties and license fees, respectively, as compared $30 and $405 for the comparable three and nine months ended March 31, 2019, respectively. The decrease in royalties and license fees for the nine months is due to the Company determining that there was no longer a requirement to pay a previously anticipated milestone.
During the year ended June 30, 2019, we incurred $4,567 in research and development, as compared $7,704 for the comparable year ended June 30, 2018. The decrease in research and development is due to the company being reimbursed by Axovant Sciences for costs relating to the OPMD program and the termination of the AMD program.
During the three and nine months ended March 31, 2020, we incurred $805 and $2,095 in research and development expenses, respectively, as compared $1,159 and $3,304 for the comparable three and nine months ended March 31, 2019, respectively. The decrease in research and development expenses is due to the company being reimbursed by Axovant Sciences for costs relating to the OPMD program and the termination of the AMD program.
General and administrative expense was $4,614 and $4,539 for the years ended June 30, 2019 and 2018, respectively. The increase was due to decreases in payroll, travel, and consultant costs offset by increases in share-based compensation and corporate costs.
76
Table of Contents
General and administrative expense was $1,287 and $3,669 and $1,103 and $3,754 for the three and nine months ended March 31, 2020 and 2019, respectively. The increase in the three month period was due to increases in costs related to legal fees to re-domicile the company to the US. The decrease in the nine month period was due to decreases in payroll, travel, and consultant costs offset by increases in share-based compensation and corporate costs.
Other Income (Loss)
The following tables set forth a summary of our other income (loss) for each of the periods set forth below:
| Year Ended June 30, | ||||||||
| 2019 | 2018 | |||||||
| (US$000) | ||||||||
| Other Income (Loss): |
||||||||
| Foreign currency transaction loss |
(75 | ) | (30 | ) | ||||
| Finance income, net |
122 | 187 | ||||||
| Unrealized loss on investment |
(21 | ) | (32 | ) | ||||
|
|
|
|
|
|||||
| Total other income (expense) |
26 | 125 | ||||||
|
|
|
|
|
|||||
| Three Months Ended March 31, |
Nine Months Ended March 31, |
|||||||||||||||
| 2020 | 2019 | 2020 | 2019 | |||||||||||||
| (unaudited) | ||||||||||||||||
| (US$000) | ||||||||||||||||
| Other Income (Loss): |
||||||||||||||||
| Foreign currency transaction loss |
(7 | ) | 5 | 4 | (75 | ) | ||||||||||
| Finance income, net |
16 | 24 | 52 | 80 | ||||||||||||
| Unrealized loss on investment |
| (1 | ) | (1 | ) | (20 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total other income (expense) |
9 | 28 | 55 | (15 | ) | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
The other income (loss) during the year ended June 30, 2019 totaled $26 of income, which consists of foreign currency transaction loss, unrealized loss on investment, and finance income. During the year ended June 30, 2018, other income (loss) totaled $125. Foreign currency transaction loss has increased due to a change in foreign exchange rates. Finance income decreased due to fewer transactions with interest. Unrealized loss on investment decreased due to the change in fair market value of the investments.
The other income (loss) during the three and nine months ended March 31, 2020 totaled $9 and $55 of income, respectively, which consists of foreign currency transaction loss, unrealized loss on investment, and finance income. During the comparable three and nine months in 2019, other income (loss) totaled $28 and $(15), respectively. The change in foreign currency transaction loss is due to a change in foreign exchange rates. Finance income decreased due to fewer transactions with interest. Unrealized loss on investment decreased due to the change in fair market value of the investments.
Net Income (Loss)
The following tables set forth a summary of our net income (loss) for each of the periods set forth below:
| Year Ended June 30, | ||||||||
| 2019 | 2018 | |||||||
| (US$000) | ||||||||
| Net Income (Loss) |
$ | 2,609 | $ | (9,077 | ) | |||
|
|
|
|
|
|||||
77
Table of Contents
| Three Months Ended March 31, |
Nine Months Ended March 31, |
|||||||||||||||
| 2020 | 2019 | 2020 | 2019 | |||||||||||||
| (unaudited) | ||||||||||||||||
| (US$000) | ||||||||||||||||
| Net Income (Loss) |
$ | (2,102 | ) | $ | (1,704 | ) | $ | (5,339 | ) | $ | 4,078 | |||||
|
|
|
|
|
|
|
|
|
|||||||||
As a result of the changes in revenues and expenses noted above, our net income (loss) increased from approximately $(9,077) in the year ended June 30, 2018 to $2,609 in the year period ended June 30, 2019.
As a result of the changes in revenues and expenses noted above, our net income (loss) decreased from approximately $(1,704) and $4,078 in the three and nine month period ended March 31, 2019, respectively, to $(2,102) and $(5,339) in the three and nine month period ended March 31, 2020, respectively.
Basic and diluted loss attributable to common stockholders was $3.05 for the year ended June 30, 2019, compared to $(12.94) for the year ended June 30, 2018.
Basic and diluted loss attributable to common stockholders was $(1.96) and $(5.36) for the three and nine month period ended March 31, 2020, respectively, compared to $(1.99) and $4.76 for the three and nine month period ended March 31, 2019, respectively.
Off-Balance Sheet Arrangements
The Company had no material off-balance sheet arrangements as of June 30, 2019.
Liquidity and Capital Resources
The Company has incurred cumulative losses and negative cash flows from operations since our inception in 1995, except for the year ended June 30, 2019 where we had a profit of $2,609 and positive cash flows of $4,790. The Company had accumulated losses of $108,870 as of June 30, 2019. We expect that our research and development expenses may increase due to the continued development of the OPMD program. It is also likely that there will be an increase in the general and administrative expenses due to the obligations of being a public company in the United States.
We had no borrowings for the years ended June 30, 2019 and 2018 and the nine months ended March 31, 2020 and 2019 and do not currently have a credit facility.
As of June 30, 2019 and March 31, 2020, we had cash and cash equivalents of $15,718 and $11,400. Cash in excess of immediate requirements is invested in accordance with our investment policy, primarily with a view to liquidity and capital preservation. Currently, our cash and cash equivalents are held in bank accounts. Our short-term investments consist of term deposits with maturity within 180 days.
The following table sets forth a summary of the net cash flow activity for each of the periods set forth below:
| Year Ended June 30, | ||||||||
| 2019 | 2018 | |||||||
| (US$000) | ||||||||
| Net cash provided by (used in): |
||||||||
| Operating activities |
$ | 4,790 | $ | (7,463 | ) | |||
| Investing activities |
(400 | ) | (60 | ) | ||||
| Financing activities |
| 6,324 | ||||||
|
|
|
|
|
|||||
| Net increase (decrease) in cash |
$ | 4,390 | $ | (1,199 | ) | |||
|
|
|
|
|
|||||
78
Table of Contents
| Nine Months Ended March 31, |
||||||||
| 2020 | 2019 | |||||||
| (unaudited) | ||||||||
| (US$000) | ||||||||
| Net cash provided by (used in): |
||||||||
| Operating activities |
$ | (4,390 | ) | $ | 6,605 | |||
| Investing activities |
(81 | ) | (293 | ) | ||||
| Financing activities |
1,770 | | ||||||
|
|
|
|
|
|||||
| Net increase (decrease) in cash |
$ | (2,701 | ) | $ | 6,312 | |||
|
|
|
|
|
|||||
Operating activities
Net cash provided by operating activities for the year ended June 30, 2019 was $4,790. Net cash used in operating activities for the year ended June 30, 2018 was $7,463. The use of net cash in all periods is primarily related to payments to suppliers and employees.
Net cash used in operating activities for the nine months ended March 31, 2020 was $4,390. Net cash provided by operating activities for the nine months ended March 31, 2019 was $6,605. The use of net cash in all periods is primarily related to payments to suppliers and employees.
Investing activities
Net cash used in investing activities for the year ended June 30, 2019 and 2018 was $400 and $60, respectively, and primarily related to purchases of equipment in 2019 and, in 2018, a clinical trial deposit.
Net cash used in investing activities for the nine months ended March 31, 2020 and 2019 was $81 and $293, respectively, and primarily related to purchases of equipment in 2020 and 2019.
Financing activities
Net cash provided by financing activities was $0 and $6,324 for the years ended June 30, 2019 and 2018, respectively. Cash from financing activities related to the issuance of ordinary shares, including $6,556 in gross proceeds from a private placement and entitlement offer for the year ended June 30, 2018, partially offset by share issue transaction costs. There were no private placements for the year ended June 30, 2019.
Net cash provided by financing activities was $1,770 and $0 for the nine months ended March 31, 2020 and 2019, respectively. Cash from financing activities related to the issuance of ordinary shares, including $2,250 in gross proceeds from a private placement and entitlement offer for the nine months ended March 31, 2020, partially offset by share issue transaction costs. There were no private placements for the nine months ended March 31, 2019.
The future of the Company as an operating business will depend on its ability to generate revenues mostly from licensing, strategic alliances and collaboration arrangements with pharmaceutical companies. While we continue to progress discussions and advance opportunities to engage with pharmaceutical companies and continue to seek licensing partners for ddRNAi in disease areas that are not our focus, there can be no assurance as to whether we will enter into such arrangements or what the terms of any such arrangement could be.
While we have established some licensing arrangements, we do not have any products approved for sale and have not generated any revenue from product sales. We do not know when, or if, we will generate any revenue from product sales. We do not expect to generate significant revenue from product sales unless and until we obtain regulatory approval of and commercialize one of our current or future product candidates.
79
Table of Contents
Unless and until we establish significant revenues from licensing programs, strategic alliances or collaboration arrangements with pharmaceutical companies, or from product sales, we anticipate that we will continue to generate losses for the foreseeable future, and we expect the losses to increase as we continue the development of product candidates and begin to prepare to commercialize any product that receives regulatory approval. We are subject to the risks inherent in the development of new gene therapy products, and we may encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may adversely affect our business.
We expect that our cash and cash equivalents will be sufficient to support preclinical studies for the BB-301 program for the next 12 months.
We have based our projections of operating capital requirements on assumptions that may prove to be incorrect and we may use all of our available capital resources sooner than we expect. Because of the numerous risks and uncertainties associated with research, development and commercialization of pharmaceutical products, we are unable to estimate the exact amount of our operating capital requirements. Our future funding requirements will depend on many factors, including, but not limited to:
| | the timing and costs of our planned clinical trials for our ddRNAi and silence and replace product candidates; |
| | the timing and costs of our planned preclinical studies for our ddRNAi and silence and replace product candidates; |
| | the number and characteristics of product candidates that we pursue; |
| | the outcome, timing and costs of seeking regulatory approvals; |
| | revenue received from commercial sales of any of our product candidates that may receive regulatory approval; |
| | the terms and timing of any future collaborations, licensing, consulting or other arrangements that we may establish; |
| | the amount and timing of any payments we may be required to make, or that we may receive, in connection with the licensing, filing, prosecution, defense and enforcement of any patents or other intellectual property rights; |
| | the costs of preparing, filing and prosecuting patent applications, maintaining and protecting our intellectual property rights and defending against intellectual property related claims; and |
| | the extent to which we need to in-license or acquire other products and technologies. |
Contractual Obligations and Commercial Commitments:
On July 9, 2018, the Company entered into an Agreement with Axovant Sciences. The Agreement granted Axovant Sciences an exclusive worldwide license to develop, manufacture, and commercialize products containing the Companys product known as BB-301, which was designed for the potential treatment of Oculopharyngeal Muscular Dystrophy. On June 6, 2019, the termination of the Agreement with Axovant Sciences was announced. The termination of the Agreement was effective as of September 3, 2019. The termination discharges all future performance obligations at termination date under the contract.
Critical Accounting Principles
Our managements discussion and analysis of financial condition and results of operations is based on our financial statements, which have been prepared in accordance with generally accepted accounting principles in the U.S. (GAAP). The preparation of these financial statements requires us to make estimates and assumptions
80
Table of Contents
that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, as well as the reported expenses during the reporting periods. These items are monitored and analyzed by us for changes in facts and circumstances, and material changes in these estimates could occur in the future. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Changes in estimates are reflected in reported results for the period in which they become known. Actual results may differ materially from these estimates under different assumptions or conditions.
While our significant accounting policies are more fully described in the notes to our audited financial statements elsewhere in this prospectus, we believe that the policies related to Revenue Recognition and ShareBased compensation are most critical to understanding and evaluating our reported financial results.
Revenue Recognition
The Company adopted and implemented on July 1, 2018, ASC 606Revenue from Contracts with Customers (ASC 606). ASC 606 did not have a material impact on the consolidated financial statements.
Upon implementation of ASC 606, the Company recognizes revenue in accordance with that core principle by applying the following steps:
Step 1: Identify the contract(s) with a customer.
Step 2: Identify the performance obligations in the contract.
Step 3: Determine the transaction price.
Step 4: Allocate the transaction price to the performance obligations in the contract.
Step 5: Recognize revenue when (or as) the entity satisfies a performance obligation.
The Company applies judgement in determining whether contracts entered into fall within the scope of ASC 606. In doing so, management considers the commercial substance of the transaction and how risks and benefits of the contract accrue to the various parties to the contract. In determining the accounting treatment of the contract with Axovant, management assessed that the contract was within the scope of ASC 606.
Management has also made the judgement that the grant of the license and transfer of associated know-how and materials are accounted for as one performance obligation as they are not considered to be distinct; they are highly interrelated and could not provide benefits to the customer independently from each other. Judgements were made in relation to the transfer of the license and know-how and whether this should be recognized over time or a point in time. The point in time has been determined with regard to the point at which the transfer of know-how has substantially been completed and the customer has control of the asset and the ability to direct the use of and receive substantially all of the remaining benefits.
Licensing revenues
Revenue from licensees of the Companys intellectual property reflects the transfer of a right to use the intellectual property as it exists at the point in time in which the license is transferred to the customer. Consideration can be variable and is estimated using the most likely amount method. Subsequently, the estimate is constrained until it is highly probable that a significant revenue reversal will not occur when the uncertainty is resolved. Revenue is recognized as or when the performance obligations are satisfied.
The Company recognizes contract liabilities for consideration received in respect of unsatisfied performance obligations and reports these amounts as other liabilities in the statement of financial position. Similarly, if the Company satisfies a performance obligation before it receives the consideration, the Company recognizes either a contract asset or a receivable in its statement of financial position, depending on whether something other than the passage of time is required before the consideration is due.
81
Table of Contents
Royalties
Revenue from licensees of the Companys intellectual property reflect a right to use the intellectual property as it exists at the point in time in which the license is granted. Where consideration is based on sales of product by the licensee, revenue is recognized when the customers subsequent sales of product occurs.
Services revenue
Revenue is earned (constrained by variable considerations) from the provision of research and development services to customers. Services revenue is recognized when performance obligations are either satisfied over time or at a point in time. Generally, the provision of research and development services under a contract with a customer will represent satisfaction of a performance obligation over time where the Company retains the right to payment for services performed but not yet completed.
Share-Based Compensation
The Company records share-based compensation in accordance with ASC 718, Stock Compensation. ASC 718 requires the fair value of all stock-based employee compensation awarded to employees to be recorded as an expense over the shorter of the service period or the vesting period. The Company values employee and non-employee share-based compensation at fair value using the Black-Scholes Option Pricing Model.
The Company adopted ASU 2018-07 and accounts for non-employee share-based awards in accordance with the measurement and recognition criteria of ASC 718 and recognizes the fair value of such awards over the service period. The Company used the modified prospective method of adoption. There was no cumulative effect of adoption on July 1, 2019.
Recent Accounting Pronouncements
ASU 2016-02In February 2016, the FASB issued ASU No. 2016-02: Leases (Topic 842) whereby lessees will need to recognize most leases on their balance sheet as a right of use asset and a lease liability. This guidance is effective for interim and annual reporting periods beginning after December 15, 2018. The Company adopted this ASU effective July 1, 2019.
The determination of whether an arrangement is or contains a lease is based on the substance of the arrangement and requires an assessment of whether the fulfilment of the arrangement is dependent on the use of a specific asset or assets and the arrangement conveys a right to use the asset.
The Company has undertaken a detailed review and has concluded that it will have a material impact on its financial position on the transactions and balances recognized in the consolidated financial statements when it is first adopted for the year ending June 30, 2020 due to the material size of lease entered into by the Company. The Companys only lease is the lease on its research and development facilities.
ASU 2016-13In June 2016, the FASB issued ASU No. 2016-13: Financial InstrumentsCredit Losses (Topic 326). This ASU represents a significant change in the accounting for credit losses model by requiring immediate recognition of managements estimates of current expected credit losses (CECL). Under the prior model, losses were recognized only as they were incurred, which FASB has noted delayed recognition of expected losses that might not yet have met the threshold of being probable. This guidance is effective for interim and annual reporting periods beginning after December 15, 2019. The Company has determined that it has met the criteria of a smaller reporting company (SRC) as of November 15, 2019. As such, ASU 2019-10: Financial Instruments-Credit Losses, Derivatives and Hedging, and Leases: Effective Dates amended the effective date for the Company to be for reporting periods beginning after December 15, 2022. The Company will adopt this ASU effective July 1, 2023.
82
Table of Contents
ASU 2017-11In July 2017, the FASB issued ASU 2017-11 Earnings Per Share (Topic 260). The amendments in the update change the classification of certain equity-linked financial instruments (or embedded features) with down round features. The amendments also clarify existing disclosure requirements for equity-classified instruments. For freestanding equity-classified financial instruments, the amendments require entities that present earnings per share (EPS) in accordance with Topic 260, Earnings Per Share, to recognize the effect of the down round feature when it is triggered. That effect is treated as a dividend and as a reduction of income available to common shareholders in basic EPS. Convertible instruments with embedded conversion options that have down round features would be subject to the specialized guidance for contingent beneficial conversion features (in Subtopic 470-20, DebtDebt with Conversion and Other Options), including related EPS guidance (in Topic 260). For public business entities, the amendments in Part I of this update are effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2018. The Company adopted this ASU effective July 1, 2019.
ASU 2018-07In June 2018, the Financial Accounting Standards Board (the FASB) issued Accounting Standards Update (ASU) 2018-07 CompensationStock Compensation (Topic 718). This update is intended to reduce cost and complexity and to improve financial reporting for share-based payments issued to non-employees (for example, service providers, external legal counsel, suppliers, etc.). The ASU expands the scope of Topic 718, CompensationStock Compensation, which currently only includes share-based payments issued to employees, to also include share-based payments issued to non-employees for goods and services. Consequently, the accounting for share-based payments to non-employees and employees will be substantially aligned. This standard will be effective for financial statements issued by public companies for the annual and interim periods beginning after December 15, 2018. Early adoption of the standard is permitted. The standard will be applied in a retrospective approach for each period presented. The Company adopted this ASU, effective July 1, 2019.
83
Table of Contents
Company Overview
We endeavor to become the leader in discovery, development, and commercialization of therapeutic agents capable of addressing significant unmet medical need via the application of the silence and replace approach to the treatment of genetic disorders.
Benitec Biopharma, Inc. (Benitec or the Company or in the third person, we or our) is a development-stage biotechnology company focused on the advancement of novel genetic medicines with headquarters in Hayward, California. The proprietary platform, called DNA-directed RNA interference, or ddRNAi, combines RNA interference, or RNAi, with gene therapy to create medicines that facilitate sustained silencing of disease-causing genes following a single administration. The Company is developing ddRNAi-based therapeutics for chronic and life-threatening human conditions including Oculopharyngeal Muscular Dystrophy (OPMD), and Chronic Hepatitis B.
BB-301 is the most advanced ddRNAi-based genetic medicine currently under development by Benitec. BB-301 is an internally optimized, AAV-based gene therapy agent that is designed to both silence the expression of mutated, disease-causing genes (to slow, or halt, the underlying mechanism of disease progression) and replace the mutant genes with normal, wild type genes (to drive restoration of function in diseased cells). This fundamental approach to disease management is called silence and replace and this biological mechanism offers the potential to restore the underlying physiology of the treated tissues and, in the process, improve treatment outcomes for patients suffering from the chronic and, potentially, fatal effects of Oculopharyngeal Muscular Dystrophy (OPMD). BB-301 has been granted Orphan Drug Designation in the United States and the European Union.
Through the combination of the targeted gene silencing effects of RNAi and the durable transgene expression achievable via the use of modified viral vectors, the silence and replace approach has the potential to produce long-term silencing of disease-causing genes along with simultaneous replacement of wild type gene function following a single administration of the proprietary genetic medicine. We believe this novel attribute of the investigational agents under development by Benitec may facilitate the achievement of robust clinical activity while greatly reducing the dosing frequencies traditionally expected for medicines employed for the management of chronic diseases. Additionally, the establishment of chronic gene silencing and gene replacement may significantly reduce the risk of patient non-compliance during the course of medical management of potentially fatal clinical disorders.
We will require additional financing to progress our product candidates through to key inflection points.
Our proprietary technology platforms are designated as DNA-directed RNA interference, or ddRNAi, and silence and replace. ddRNAi is designed to produce long-term silencing of disease-causing genes, by combining RNA interference, or RNAi, with viral delivery agents typically associated with the field of gene therapy (i.e. viral vectors). Modified AAV vectors are employed to deliver genetic constructs which encode short hairpin RNAs that are, then, serially expressed and processed, to produce siRNA molecules within the transduced cell for the duration of the life of the target cell. These newly introduced siRNA molecules drive long-term, and potentially permanent, silencing of the expression of the disease-causing gene. The silence and replace approach further bolsters the biological benefits of long-term silencing of disease-causing genes by incorporating multifunctional genetic constructs within the modified AAV vectors to create an AAV-based gene therapy agent that is designed to both silence the expression of mutated, disease-causing genes (to slow, or halt, the underlying mechanism of disease progression) and, simultaneously, replace the mutant genes with normal, wild type genes (to drive restoration of function in diseased cells). This fundamentally distinct approach to disease management offers the potential to restore the underlying physiology of the treated tissues and, in the process, improve treatment outcomes for patients suffering from the chronic and, potentially, fatal effects of diseases like Oculopharyngeal Muscular Dystrophy (OPMD).
84
Table of Contents
Traditional gene therapy is defined by the introduction of an engineered transgene to correct the pathophysiological derangements derived from mutated or malfunctioning genes. Mutated genes can facilitate the intracellular production of disease-causing proteins or hamper the production of critical, life-sustaining, proteins, and the introduction of a new transgene can facilitate the restoration of production of normal proteins within the diseased cell, thus, restoring natural biological function. Critically, the implementation of this traditional method of gene therapy cannot eliminate the expression, or the potential deleterious effects of, the underlying mutant gene (as mutant proteins may be continually expressed and aggregate or drive the aggregation of other native proteins within the diseased cell). In this regard, the dual capabilities of the proprietary silence and replace approach to silence a disease-causing gene via ddRNAi and simultaneously replace the wildtype activity of a mutant gene via the delivery of an engineered transgene could facilitate the development of differentially efficacious treatments for a range of genetic disorders.
Overview of RNAi and the siRNA Approach
The mutation of a single gene can cause a chronic disease via the resulting intracellular production of a disease-causing protein (i.e. an abnormal form of the protein of interest), and many chronic and/or fatal disorders are known to result from the inappropriate expression of a single gene or multiple genes. In some cases, genetic disorders of this type can be treated exclusively by silencing the intracellular production of the disease-causing protein through well-validated biological approaches like RNA interference (RNAi). RNAi employs small nucleic acid molecules to activate an intracellular enzyme complex, and this biological pathway temporarily reduces the production of the disease-causing protein. In the absence of the disease-causing protein, normal cellular function is restored and the chronic disease that initially resulted from the presence of the mutant protein is partially or completely resolved. RNAi is potentially applicable to over 20,000 human genes and a large number of disease-causing microorganism-specific genes.
A small double stranded RNA, or dsRNA, molecule (A, Figure 1), comprising one strand known as the sense strand and another strand known as the antisense strand, which are complementary to each other, is synthesized in the laboratory. These small dsRNAs are called small interfering RNAs, or siRNAs. The sequence of the sense strand corresponds to a short region of the target gene mRNA. The siRNA is delivered to the target cell (B, Figure 1), where a group of enzymes, referred to as the RNA-Induced Silencing Complex, or RISC, process the siRNA (C, Figure 1), where one of the strands (usually the sense strand) is released (D, Figure 1). RISC uses the antisense strand to find the mRNA that has a complementary sequence (E, Figure 1) leading to the cleavage of the target mRNA (F, Figure 1). As a consequence, the output of the mRNA (protein production) does not occur (G, Figure 1). Several companies, including Alnylam, Arbutus, and Dicerna, utilize this approach in their RNAi product candidates.
Importantly, many genetic disorders are not amenable to the traditional gene silencing approach outlined in Figure 1, as the diseased cells may produce a mixture of the wildtype protein of interest and the disease-causing, mutant variant of the protein, and the underlying genetic mutation may be too small to allow for selective targeting of the disease-causing variant of the protein through the use of siRNA-based approaches exclusively. In these cases, it is extraordinarily difficult to selectively silence the disease-causing protein without simultaneously silencing the wildtype intracellular protein of interest whose presence is vital to the conduct of normal cellular functions.
Our proprietary silence and replace technology utilizes the unique specificity and robust gene silencing capabilities of RNAi while overcoming many of the key limitations of siRNA-based approaches to disease management.
85
Table of Contents
In the standard RNAi approach, double-stranded siRNA is produced synthetically and, subsequently, introduced into the target cell via chemical modification of the RNA or alternative methods of delivery. While efficacy has been demonstrated in several clinical indications through the use of this approach, siRNA-based approaches maintain a number of limitations, including:
| | Clinical management requires repeat administration of the siRNA-based therapeutic agent for multiple cycles to maintain efficacy; |
| | Long-term patient compliance challenges due to dosing frequencies and treatment durations; |
| | Therapeutic concentrations of siRNA are not stably maintained because the levels of synthetic siRNA in the target cells decrease over time; |
| | Novel chemical modifications or novel delivery materials are typically required to introduce the siRNA into the target cells, making it complicated to develop a broad range of therapeutics agents; |
| | Potential adverse immune responses, resulting in serious adverse effects; |
| | Requirement for specialized delivery formulations for genetic disorders caused by mutations of multiple genes; and |
| | siRNA acts only to silence genes, and cannot be used to replace defective genes with normally functioning genes. |
Our Approach to the Treatment of Genetic DiseasesddRNAi and Silence and Replace
Our proprietary silence and replace approach to the treatment of genetic diseases combines RNAi with wildtype gene replacement to drive sustained silencing of disease-causing genes and concomitant restoration of functional wildtype genes following a single administration of the therapeutic agent. Benitec employs ddRNAi in combination with classical gene therapy (i.e. transgene delivery via viral vectors) to overcome several of the fundamental limitations of RNAi.
The silence and replace approach to the treatment of genetic disorders employs adeno-associated viral vectors (AAVs) to deliver genetic constructs which may, after a single administration to the target tissues:
| | Chronically express RNAi molecules inside of the target, diseased, cells (to serially silence the intracellular production of mutant, disease-causing, protein and the wildtype protein of interest); |
| | Simultaneously drive the expression of a wildtype variant of the protein of interest (to restore native intracellular biological processes); and |
| | AAV vectors can accommodate the multi-functional DNA expression cassettes containing the engineered wildtype transgenes and the novel genes encoding short hairpinRNA/microRNA molecules (shRNA/miRNA) that are required to support the development of therapeutic agents capable of the achievement of the goals of the silence and replace approach to therapy. |
Our silence and replace technology utilizes proprietary DNA expression cassettes to foster continuous production of gene silencing shRNAs and wildtype proteins (via expression of the wildtype transgene). A range of viral and non-viral gene therapy vectors can be used to deliver the DNA construct into the nucleus of the target cell and, upon delivery, shRNA molecules are expressed and subsequently processed by intracellular enzymes into siRNA molecules that silence the expression of the mutant, disease causing protein (Figure 2).
In the silence and replace approach (Figure 2):
| | A DNA construct is delivered to the nucleus of the target cell by a gene therapy vector (A) such as an AAV; |
| | Once inside of the nucleus, the DNA construct drives the continuous production of shRNA molecules (B) which are processed by an enzyme called Dicer into siRNAs (C); |
86
Table of Contents
| | The processed siRNA is incorporated into RISC and silences the target gene using the same mechanism shown in Figure 1; and |
| | When the DNA expression cassette is also comprised of a wildtype transgene, upon entry of the DNA construct into the nucleus of the target cell via the use of the AAV vector, the DNA construct also drives the continuous production of wildtype protein (to restore native intracellular biological processes). |
Our strategy is to discover, develop and commercialize treatments that leverage the capabilities of ddRNAi and the silence and replace approach to disease management.
For selected product candidates, at the appropriate stage, we may collaborate with large biopharmaceutical companies to further co-develop and, if approved, commercialize our ddRNAi-based and silence and replace-based products to achieve broad clinical and commercial distribution. For specific clinical indications that we deem to be outside of our immediate areas of focus (e.g. HBV), we will continue to out-license, where appropriate, applications of our ddRNAi and silence and replace technology to facilitate the development of differentiated therapeutics, which could provide further validation of our proprietary technology and approach to disease management.
Our cash and cash equivalents will be deployed to advance our product candidate BB-301 for OPMD and, while we are not actively working on the development of BB-103 for the treatment of HBV due to financial constraints, we will seek a strategic partnership to support its development.
Oculopharyngeal Muscular DystrophyOPMD
OPMD is an insidious, autosomal-dominant, late-onset degenerative muscle disorder that typically presents in patients at 40-to-50 years of age. The disease is characterized by progressive swallowing difficulties (dysphagia) and eyelid drooping (ptosis). OPMD is caused by a specific mutation in the poly(A)-binding protein nuclear 1, or PABPN1, gene. OPMD is a rare disease; however, patients have been diagnosed with OPMD in at least 33 countries. Patients suffering from OPMD are well-identified, and significant geographical clustering has been noted for patients with this disorder, which could simplify clinical development and global commercialization efforts.
BB-301 is an internally optimized, AAV-based gene therapy agent that is designed to both silence the expression of mutated, disease-causing genes (to slow, or halt, the underlying mechanism of disease progression) and replace the mutant genes with normal, wild type genes (to drive restoration of function in diseased cells). This fundamental approach to disease management is called silence and replace and this biological mechanism offers the potential to restore the underlying physiology of the treated tissues and, in the process, improve treatment outcomes for patients suffering from the chronic and, potentially, fatal effects of Oculopharyngeal Muscular Dystrophy (OPMD). BB-301 has been granted Orphan Drug Designation in the United States and the European Union.
As of September 3, 2019, the License and Collaboration Agreement with Axovant was terminated. As a result, all rights and licenses which Benitec had granted to Axovant to develop and commercialize BB-301 and related gene therapy product candidates terminated. We are now solely responsible for the costs in connection with the development and commercialization of the BB-301 product candidates.
Prior to such termination, the Benitec team endeavored to conduct several additional exploratory nonclinical analyses in order to potentially improve the biological efficacy of BB-301 via further optimization of the route of administration employed to dose the target muscle tissues.
Hepatitis B
The Company is developing BB-103 for the treatment of HBV. Results of in vivo and in vitro studies, from December 2016, March 2016 and December 2015, demonstrated the potential utility of an approach that
87
Table of Contents
combines RNAi with gene therapy to treat HBV. In April 2017, the Company completed a pre-IND submission with the FDA in which the feedback provided by the agency included details regarding steps required to initiate a clinical trial for BB-103. As noted, due to financial constraints, the Company is seeking strategic partnerships to complete the IND enabling studies for BB-103.
Our Strengths
We believe that the combination of our proprietary ddRNAi technology and our deep expertise in the design and development of genetic medicines, and specifically ddRNAi-based therapeutics, will enable us to achieve and maintain a leading position in gene silencing and gene therapy for the treatment of human disease. Our key strengths include:
| | A first mover advantage for ddRNAi-based therapeutics; |
| | A proprietary ddRNAi-based silence and replace technology platform that may potentially enable the serial development of single-administration therapeutics capable of facilitating sustained, long-term silencing of disease-causing genes and concomitant replacement of wildtype gene function; |
| | A proprietary AAV vector technology which improves the endosomal escape capability of virus produced in insect cells using a baculovirus system. This technology has broad application in AAV-based gene therapies; |
| | The capabilities to drive the development of a pipeline of programs focused on chronic diseases with either large patient populations, including Chronic Hepatitis B virus infection, or rare diseases, which may potentially support the receipt of Orphan Drug Designation, including OPMD; and |
| | A growing portfolio of patents protecting improvements to our ddRNAi, and silence and replace, technology and product candidates through at least 2036, with additional patent life anticipated through at least 2040. |
Our Strategy
We endeavor to become the leader in discovery, development, and commercialization of therapeutic agents capable of addressing significant unmet medical need via the application of the silence and replace approach to the treatment of genetic disorders. We apply the following general strategy to drive the Company towards these goals:
| | Selectively develop proprietary and partnered programs; and |
| | Continue to explore and secure research and development partnerships with global biopharmaceutical companies supported by the differentiated nature of our scientific platform and intellectual property portfolio. |
Our senior leadership team will continue to explore partnership opportunities with global biopharmaceutical companies, as we expect that the unique attributes of the proprietary ddRNAi and silence and replace approaches, and the breadth of potential clinical indications amenable our proprietary methods, to support the formation of collaborations over a broad range of diseases with significant unmet medical need.
We seek to actively protect our intellectual property and proprietary technology. These efforts are central to the growth of our business and include:
| | Seeking and maintaining patents claiming our ddRNAi and silence and replace technologies and other inventions relating to our specific products in development or that are otherwise commercially and/or strategically important to the development of our business; |
| | Protecting and enforcing our intellectual property rights; and |
| | Strategically licensing intellectual property from third parties to advance development of our product candidates. |
88
Table of Contents
Our Pipeline
The following table sets forth our current product candidates and their development status:
Table 1. Pipeline: Oculopharyngeal Muscular Dystrophy and Chronic Hepatitis B Virus Infection
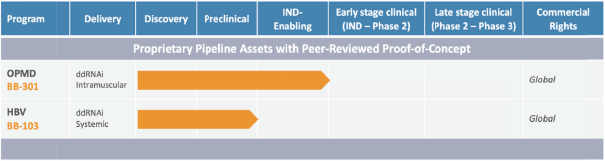
BB-301 is a late-stage nonclinical investigational agent currently in development for the treatment of Oculopharyngeal Muscular Dystrophy. BB-301 is the lead pipeline program for Benitec, and IND-enabling studies are currently being conducted. A summary of the BB-301 program is provided in Figure 3.
BB-103 has demonstrated robust nonclinical activity during the evaluation of this agent for the treatment of Chronic Hepatitis B Virus infection. Benitec is currently seeking strategic partners to advance BB-103 through IND-enabling studies.
In-House Programs
BB-301 for Treatment of Oculopharyngeal Muscular Dystrophy
OPMD is an insidious, autosomal-dominant, late-onset, degenerative muscle disorder that typically presents in patients at 40-to-50 years of age. The disease is characterized by progressive swallowing difficulties (dysphagia) and eyelid drooping (ptosis). OPMD is caused by a specific mutation in the poly(A)-binding protein nuclear 1, or PABPN1, gene. OPMD is a rare disease, however, patients have been diagnosed with OPMD in at least 33 countries. Patients suffering from OPMD are well-identified, and significant geographical clustering has been noted for patients with this disorder, which could simplify clinical development and global commercialization efforts.
PABPN1 is a ubiquitous factor that promotes interaction between the poly(A) polymerase and CPSF (cleavage and polyadenylation specificity factor) and, thus, controls the length of mRNA poly(A) tails, mRNA export from the nucleus, and alternative poly(A) site usage. The characteristic genetic mutation underlying OPMD results in trinucleotide repeat expansion(s) within exon 1 of PABPN1 and results in an expanded poly-alanine tract at the N-terminal end of PABPN1. The mutation generates a protein with an N-terminal expanded poly-alanine tract of up to 18 contiguous alanine residues prone to form aggregates called intranuclear inclusions (INIs). The INIs that sequester wildtype PABPN1 could also contribute to loss of the function phenotype associated with OPMD.
Current OPMD Treatments and Products in Development
No curative or disease-modifying therapies currently exist for OPMD patients. Surgical interventions can be undertaken for palliative purposes, including the use of cricopharyngeal myotomy.
Investigational therapies that have been explored, unsuccessfully, in the past include:
| | Intravenous administration of trehalose; and |
| | The use of autologous myoblast transplant. |
89
Table of Contents
Our Lead, Silence and Replace-Based, OPMD Therapeutic AgentBB-301
BB-301 is composed of a modified AAV serotype 9 (AAV9) capsid that expresses a bifunctional construct under the control of a single muscle specific Spc5-12 promoter to achieve co-expression of both the codon-optimized PABPN1 mRNA and two shmiR molecules directed against wild type and mutant PABPN1, and the agent is designed to correct the genetic defect underlying OPMD following a single localized administration.
BB-301Design and Mechanism of Action
BB-301 is designed to target two distinct regions of the PABPN1 mRNA via the concomitant expression of two distinct shmiRs from a single DNA construct and the simultaneous expression of a codon-optimized, siRNA-resistant, version of the wildtype PABPN1 gene (Figure 4).
Figure 4
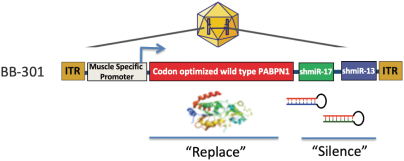
In collaboration with researchers at the Royal Holloway University of London and the Institut de Myologie in Paris, we developed a ddRNAi construct expressing three shRNAs against three distinct regions of PABPN1 mRNA and observed effective silencing of the PABPN1 gene in vitro using this ddRNAi construct. Furthermore, as part of this collaboration, we have generated a gene expression construct that produces a siRNA-resistant version of the wildtype PABPN1 gene.
90
Table of Contents
In subsequent studies undertaken exclusively by Benitec, a second set of target regions within PABPN1 were identified for therapeutic development and shmiRs designed against these regions. Additional shmiRs have also been designed for the original shRNA developed in collaboration with Royal Holloway University of London and the Institut de Myologie. The silence and replace construct, designated BB-301, incorporates the two best performing shmiRs, and the gene expression construct that produces a siRNA-resistant version of the wildtype PABPN1 gene, under the control of a muscle-specific promoter. The mechanism of action of BB-301 is shown in Figure 5.
Figure 5
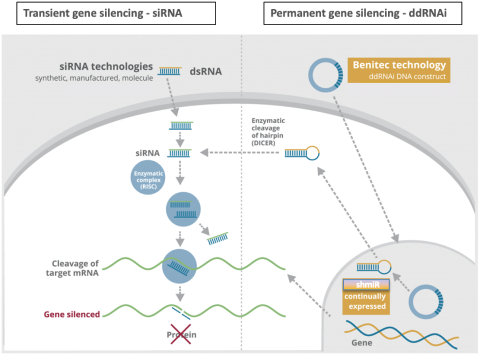
In initial in vivo studies evaluating the use of direct intramuscular injection of AAV-based constructs with the potential to drive the desired silence and replace approach in the A17 transgenic mouse model of OPMD at the Royal Holloway University of London and the Institut de Myologie, we observed decreases in muscle fibrosis, increases in cross sectional area of the treated muscles, decreases in intranuclear inclusions, and normalization of muscle strength. These nonclinical results were published in Nature Communications in April 2017.
In subsequent studies, Benitec demonstrated in a key non-clinical model (the A17 mouse model) that a single intramuscular injection of BB-301 results in robust intracellular silencing of PABPN1 protein production and concomitant expression of the normal, biologically functional PABPN1 protein. In the A17 mouse model, the treatment restores muscle strength and muscle weight to wild type levels and improves other physiological hallmarks of the disease (Figure 6a, Figure 6b, Figure 6c, Figure 6d):
| | Multiple A17 animal cohorts received single doses of BB-301 (over a range of doses spanning 4x108 vg/muscle-to-7.5x1011 vg/muscle) and, following BB-301 administration, each cohort was observed for 14- weeks |
| | BB-301 was injected into the Tibialis Anterior (TA) muscle of 10 week old-to-12 week old animals, and 14- weeks post administration each A17 cohort was anesthetized and the contractile properties of the injected TA muscles were analyzed via in-situ muscle electrophysiology |
91
Table of Contents
| | Intermediate doses of BB-301 resulted in 75% silencing of PABPN1 and 26% replacement of wild type PABPN1 activity, leading to full restoration of muscle strength, clearance of INIs, and a reduction of fibrosis |
| | An additional experiment conducted over the course of 20-weeks demonstrated that more modest doses of BB-301 (which supported only partial resolution of the disease phenotype at week-14) were, surprisingly, able to facilitate significant benefit at 20-weeks, as evidenced by full restoration of all parameters relating to muscle strength, weight and INI formation |
Figure 6a. Dose-Dependent shRNA Expression
Figure 6b. Dose-Dependent PABPN1 Inhibition
92
Table of Contents
Figure 6c. Dose-Dependent Decreases in Intranuclear Inclusions
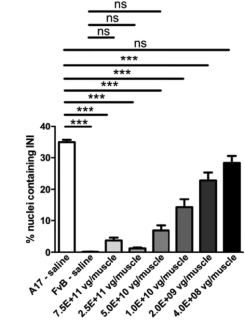
Figure 6d. Dose-Dependent Increases in Muscle Force
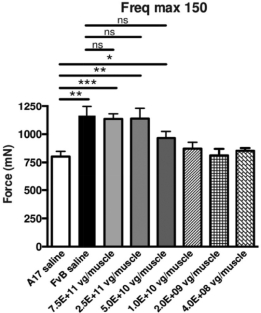
|
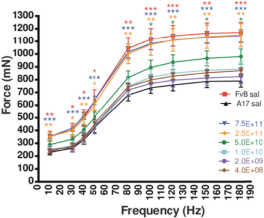
|
Ongoing Development Plans for BB-301
On July 8, 2020, Benitec announced the initiation of the BB-301 Tissue Transduction Study in large animal subjects.
The BB-301 Tissue Transduction Study is the first of three planned IND-enabling studies that will be conducted in large animals. These IND-enabling studies will be carried out under the guidance of the scientific
93
Table of Contents
team at Benitec, with key elements of the study design and execution conducted in close collaboration with a team of leading experts in both medicine and surgery that have been deeply engaged in the treatment of OPMD patients for several decades. The BB-301 Tissue Transduction study, along with the subsequent non-clinical studies, will be conducted in canine subjects and will support the validation and optimization of the newly designed method of BB-301 administration, confirm the efficiency of vector transduction in the key tissue compartments underlying the natural history of OPMD, confirm the optimal drug doses in advance of initiation of human clinical studies, and facilitate observation of key toxicological data-points.
The BB-301 Tissue Transduction Study is designed as an 8-week study in Beagle dogs to confirm the transduction efficiency of BB-301 upon administration via direct intramuscular injection into specific anatomical regions of the pharynx through the use of an open surgical procedure. This new route of BB-301 administration was developed in collaboration with key surgical experts in the field of Otolaryngology, and this novel method of BB-301 dosing will significantly enhance the ability of a treating physician to accurately administer the AAV-based investigational agent to the muscles that underlie the characteristic deficits associated with the progression of OPMD. It is important to note that prior non-clinical studies of BB-301 have reproducibly validated the robust biological activity achieved following direct intramuscular injection. As an example, direct injection of BB-301 into the tibialis anterior muscles of A17 mice facilitated robust transduction of the targeted skeletal muscle cells and supported complete remission of the OPMD disease phenotype in this animal model.
Interim data for the BB-301 Tissue Transduction Study is expected in late 2020 or early 2021.
BB-103 for the Treatment of Hepatitis B
We are seeking strategic partners to continue the development of BB-103 for the treatment of HBV. Results of in vivo and in vitro studies, from December 2016, March 2016 and December 2015, have, we believe, demonstrated the potential utility of an approach that combines RNAi with gene therapy to treat HBV. The Company is seeking strategic partnerships to complete the IND-enabling studies for BB-103.
The human hepatitis B virus is a small DNA virus that, according to the World Health Organization, infects up to 240 million people worldwide, resulting in up to 780,000 deaths per year. HBV infection can lead to differential outcomes, ranging from a silent, acute phase infection that can be resolved via the inherent action of the immune system, to a chronic infection requiring life-long therapy. In the case of a chronic HBV infection, the presence of viral proteins, particularly the hepatitis B surface antigen, causes hepatic inflammation, liver dysfunction, acute hepatic failure, cirrhosis and/or hepatocellular carcinoma.
Current Hepatitis B Treatments
HBV predominantly exists as eight genotypes, designated A through H, with distinct geographic distribution.
According to GlobalData, a market research firm, the global hepatitis B therapeutics market was worth $2.4 billion in 2014 and is expected to reach a total value of $3.0 billion by 2024 at a Compound Annual Growth Rate of 2.4%. The current standards of care for HBV consist of antivirals composed of nucleotide and nucleoside analogues, or NUCs, and, less commonly, interferon therapy.
Most of the currently employed anti-HBV therapies can provide long-term viral load suppression, however, these therapeutic agents have modest cure rates and possess the additional risk of driving the development of drug-resistant mutations. The long-term use of interferon, particularly in high doses, may also be associated with significant side effects, including nausea, vomiting, shortness of breath, dizziness and fatigue, adding to issues with patient compliance for the course of treatment.
94
Table of Contents
Our ddRNAi-based Hepatitis B Therapeutic-BB-103
BB-103 is a ddRNAi-based therapeutic designed to target the liver and inhibit viral replication and surface-antigen production. The initial in vivo data for BB-103 suggest that joint administration of BB-103 and a NUC could support reactivation of the host immune response which is believed to be essential for the achievement of cure.
BB-103-Design and Mechanism of Action
The design of the BB-103 DNA construct takes advantage of the structure of the HBV genome. The hepatitis B virus is a small DNA virus with four overlapping open reading frames, meaning several genes are produced from the same DNA sequence by shifting the starting point of the translation process (Figure 7B). These four genes are known as the core, surface, X and polymerase genes. The core gene encodes the core nucleocapsid protein, which is important in viral packaging and thought to help stabilize cccDNA, and the hepatitis B e-antigen. The surface gene encodes proteins, including s-antigen. The X gene encodes the X protein, which has properties that may be relevant to liver carcinogenesis. The polymerase gene encodes a large protein with functions critical for viral packaging and replication. Although HBV is a DNA virus, it replicates through an RNA intermediate. BB-103 targets the viral mRNA at three overlapping regions of the genome (Figure 7A and Figure 7B), simultaneously silencing the surface, X, core and polymerase genes. As a result, we believe that the long-term suppression of HBV viral replication, through silencing of the polymerase gene and the HBV RNA used for replication, the inhibition of HBV viral proteins production, including s-antigen production, through silencing of the surface gene, and the inhibition of the cccDNA, through silencing of the core protein gene, could lead to eradication of HBV infection in patients by a single administration of BB-103 when paired with a NUC.
In Vitro Development Highlights
Our bioinformatics analysis of the major HBV genotypes, A through H, has identified several well-conserved regions of the genome for targeting with ddRNAi therapeutics, and we have designed and evaluated numerous shRNAs to target these regions. The most advanced nonclinical construct is illustrated in Figure 7A.
Figure 7
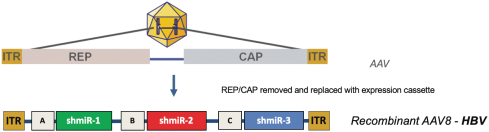
95
Table of Contents
Figure 8
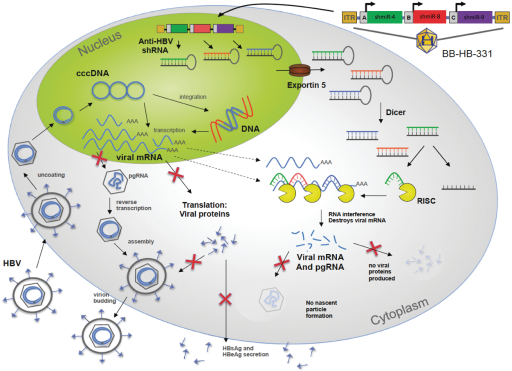
Regarding the mechanism of action of BB-103, the DNA construct is delivered to the nucleus of hepatocytes via an AAV8 vector. Upon reaching the nucleus the construct expresses three distinct shmiRs that are processed intracellularly to produce siRNAs that cleave the HBV mRNA and, thus, prevent the virus from replicating and producing viral proteins.
Intellectual Property
Benitec seeks to actively procure rights to and protect the intellectual property and proprietary technology that it believes is important to its business. Such intellectual property rights include patents claiming our ddRNAi and silence and replace technologies, as well as know-how and trade secrets related to our product candidates and proprietary technology.
ddRNAi-based treatment of Hepatitis B
The Benitec patent portfolio includes four patent families relevant to Benitecs ddRNAi-based candidate for treatment of hepatitis B virus (HBV) infection (BB-103). This includes three patent families directed to RNAi agents targeting HBV and Benitecs AAV patent family which covers the delivery system for BB-103.
The first patent family directed to RNAi agents targeting HBV, entitled HBV Treatment (HBV family #1), relates to single-stranded RNA and shRNA sequences to a range of target regions of the hepatitis B viral genome. Although drafted to explicitly cover shRNAs (as this was the RNAi format under development at the time), the claims encompass shRNAs comprised within a microRNA backbone (i.e., short hairpin microRNA (shmiR)) which is the format of RNAi currently in use within Benitecs HBV program.
A further patent family relating to shRNA sequences to a range of additional target regions of the hepatitis B viral genome was filed. This patent family is entitled Reagents for treatment of hepatitis B virus (HBV) infection and use thereof (HBV family #2) and, although drafted to cover shRNAs (as this was the RNAi format under development at the time), the claims encompass shRNA and shmiR.
96
Table of Contents
The third patent family relevant to HBV relates to the development of constructs. This patent family, entitled Reagents for treatment of hepatitis B virus (HBV) infection and use thereof (HBV family #3), was filed to cover single and triple shmiR constructs currently under development at Benitec.
ddRNAi-based treatment for OPMD
Benitecs patent portfolio for OPMD includes five patent families relating to shRNA and shmiRs targeting PABPN1 (the causative gene for OPMD), as well as silence and replace therapeutics and treatment strategies for OPMD. These five families cover the OPMD therapeutic candidate, BB-301, under development at Benitec, treatment strategies for OPMD that silence PABPN1 which is causative for OPMD and replace with functional PABPN1, and Benitecs AAV patent family which covers the delivery system for BB-301. BB-301 is a silence and replace construct encoding two shmiRs targeting the endogenous PABPN1 (including variants causative of OPMD) internally designated shmiR-13 and shmiR-17, as well as a codon-optimized PABPN1 replacement construct, the transcript of which is not targeted by shmiR-13 and shmiR-17. Both shmiRs and the codon-optimized PABPN1 replacement construct are under the control of a muscle-specific promoter.
The first patent family, entitled Reagents for treatment of oculopharyngeal muscular dystrophy (OPMD) and use thereof (OPMD family #1), arose out of a collaboration with Royal Holloway University of London (RHUL) and relates to three shRNA target regions within PABPN1. RHUL assigned its ownership interests in this patent family to Benitec, and the PCT application and the related U.S. priority document were filed solely in the name of Benitec. This patent family is directed to RNAi agents targeting specific regions within mutant PABPN1 variants causative of OPMD, as well as use of those RNAi agents in combination with PABPN1 replacement constructs to treat OPMD. More specifically, this family includes claims covering shmiR17 of BB-301 This patent family entered the national/regional phase in October/November 2018.
The second patent family, entitled Reagents for treatment of oculopharyngeal muscular dystrophy (OPMD) and use thereof (OPMD family #2) relates to a second set of target gene sequences within PABPN1 as well as silence and replace construct BB-301 under development at Benitec. The PCT application and the related U.S. priority document were filed solely in the name of Benitec, and this family entered the national/regional phase in June/July 2019. This patent family is directed to RNAi agents targeting specific regions within mutant PABPN1 variants causative of OPMD, as well as silence and replace constructs and use of same for treatment of OPMD. More specifically, this family includes claims covering shmiR13 and shmiR17 of BB-301 separately, as well as the full BB-301 knockdown and replacement construct.
A third patent family, entitled Methods for Treating Oculopharyngeal Muscular Dystrophy (OPMD) (OPMD family #3) has been filed to pursue claims which are broadly directed to the silence and replace treatment concept for OPMD, relying on RNAi agents to knockdown PABPN1 and replacement with functional PABPN1 which is not targeted by the RNAi agents. The claims in this application are not limited to BB-301. This patent family exists as a PCT application and was filed solely in the name of Benitec.
A fourth patent family, entitled Methods for Treating Oculopharyngeal Muscular Dystrophy (OPMD) (OPMD family #4) has been filed to specifically claim the OPMD therapeutic candidate developed by Benitec, BB-301 (described herein). This patent family exists as a PCT application and was filed solely in the name of Benitec.
AAV with modified phospholipase domain
The Benitec patent portfolio includes a single patent family, entitled Adeno-associated virus (AAV) with modified phospholipase domain, which relates to an AAV having a modified phospholipase (PLA2) domain in the capsid. The modified AAV will be used as the delivery system for the OPMD therapeutic candidate and the HBV therapeutic candidate.
97
Table of Contents
We are aware of a third party patent directed to AAV vectors that expires in 2026. In the event we receive regulatory marketing approval before the expiration date it may be necessary for us to obtain a license to the patent in order to commercialize. We cannot guarantee the availability of the license or that it can be obtained on commercially reasonable terms.
Know-How
In addition to patent protection of ddRNAi and other technology and our product candidates, we also rely on proprietary know-how that is not patentable or that we elect not to patent, as valuable intellectual property for our business. This know-how is related to the areas of, among others, identifying nucleic acid targets for ddRNAi technology and designing ddRNAi constructs for targeting preferred genes. We have implemented a number of security measures designed to safeguard our know-how including limiting access to our research facilities, databases and networks. We also seek to protect our know-how by way of confidentiality agreements when engaging with external providers for progressing our pipeline of therapeutic candidates.
Laws and Regulations Regarding Patent Terms
The term of individual patents depends upon the legal terms of the patents in the countries in which they are obtained. In most countries in which we file, the patent term is 20 years from the earliest date of filing a non-provisional patent application. In the United States, a patent term may be shortened if a patent is terminally disclaimed over another patent or as a result of delays in patent prosecution by the patentee. A patents term may be lengthened by a patent term adjustment, which compensates a patentee for administrative delays by the USPTO in granting a patent. The patent term of a European patent is 20 years from its filing date, which, unlike in the United States, is not subject to patent term adjustments.
The term of a patent that covers an FDA-approved biologic may also be eligible for patent term extension, which permits patent term restoration as compensation for the patent term lost during the FDA regulatory review process. The Drug Price Competition and Patent Term Restoration Act of 1984, or the Hatch-Waxman Act, permits a patent term extension of up to five years beyond the expiration of the patent. The length of the patent term extension is related to the length of time the biologic is under clinical testing regulatory review. Patent extension cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval and only one patent applicable to an approved biologic may be extended. Similar provisions are available in Europe and other jurisdictions to extend the term of a patent that covers an approved biologic although the eligibility requirements for any duration of such extension vary. In the future, if and when our products receive FDA approval, or approval from an equivalent regulatory body in another jurisdiction in which patent protection is sought or obtained, we expect to apply for patent term extensions on patents covering those products.
Trademarks
Our trademarks include registrations for company branding and product names for our pipeline in development. Several of the trademarks that we use in connection with our business are described below.
| Trade Mark (program) |
Number | Filing date | Jurisdiction | Status | ||||
| BENITEC® |
1103049 | 10 Mar 2006 | Australia | Registered | ||||
| BENITEC® |
86795296 | 21 Oct 2015 | United States | Registered | ||||
| BENITEC |
1728797 | 16 Oct 2015 | Australia | Registered | ||||
| BENITEC |
14680003 | 15 Oct 2015 | European Union | Registered | ||||
| BENITEC BIOPHARMA® |
1448046 | 13 Sep 2011 | Australia | Registered | ||||
| BENITEC BIOPHARMA® |
4636053 | 11 Feb 2014 | United States | Registered | ||||
| GIVING DISEASE THE SILENT TREATMENT® |
1851660 | 14 Jun 2017 | Australia | Registered | ||||
| GIVING DISEASE THE SILENT TREATMENT® |
1389399 | 15 Feb 2018 | European Union | Registered | ||||
| SILENCING GENES FOR LIFE® |
1448041 | 13 Sep 2011 | Australia | Registered | ||||
| SILENCING GENES FOR LIFE® |
4807242 | 22 Dec 2014 | United States | Registered |
98
Table of Contents
Manufacturing
The manufacture of the complex biological products required for gene therapy is complex and difficult. We do not currently own or operate manufacturing facilities for the production of preclinical, clinical or commercial quantities of any of our product candidates. We are exploring long- term manufacturing alliances with a number of potential partners to investigate manufacturing processes in order to produce materials at reasonable scale and cost of goods to support future commercialization efforts. We do not have a long-term agreement with any third-party manufacturer, but we plan to establish such a relationship with an appropriate manufacturer to serve our long-term needs.
Manufacturing is subject to extensive regulations that impose various procedural and documentation requirements, which govern record keeping, manufacturing processes and controls, personnel, quality control and quality assurance, among others. Our contract manufacturing organizations manufacture our product candidates under cGMP conditions. cGMP is a regulatory standard for the production of pharmaceuticals that will be used in humans.
Sales and Marketing
We have not yet established sales, marketing or product distribution operations because our product candidates are in preclinical or clinical development. If we receive marketing and commercialization approval for any of our product candidates, we intend to market the product through strategic alliances and distribution agreements with third parties. In certain cases, we may market an approved product directly worldwide or in selected geographical segments. The ultimate implementation of our strategy for realizing the financial value of our product candidates is dependent on the results of clinical trials for our product candidates, the availability of funds and the ability to negotiate acceptable commercial terms with third parties.
Competition
The biopharmaceutical industry is characterized by intense and dynamic competition to develop new technologies and proprietary therapies.
Any product candidates that we successfully develop and commercialize will have to compete with existing therapies and new therapies that may become available in the future. While we believe that our proprietary technology and scientific expertise in gene silencing using ddRNAi provide us with competitive advantages, we face potential competition from many different sources, including larger and better-funded pharmaceutical, specialty pharmaceutical and biotechnology companies, as well as from academic institutions and governmental agencies and public and private research institutions that may develop potentially competitive products or technologies. We are aware of several companies focused on developing gene therapy or gene silencing product candidates, including Alnylam, Arbutus and Arrowhead-developing siRNA-based therapeutics for hepatitis B.
We are not aware of any companies developing a gene therapy or gene silencing approach for OPMD. Our product candidates, if approved, would also compete with treatments that have already been approved and accepted by the medical community, patients and third-party payers.
Many of our competitors and potential competitors, alone or with their strategic partners, have substantially greater financial, technical and human resources than we do and significantly greater experience in the discovery and development of product candidates, obtaining FDA and other regulatory approvals of treatments and the commercialization of those treatments. Mergers and acquisitions in the biotechnology and pharmaceutical industries may result in even more resources being concentrated among a smaller number of our competitors. These competitors also compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical study sites and subject registration for clinical studies, as well as in acquiring technologies complementary to, or necessary for, our programs. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
99
Table of Contents
We anticipate that we will face intense and increasing competition as new products enter the market and advanced technologies become available. We expect any treatments that we develop and commercialize to compete on the basis of, among other things, efficacy, safety, convenience of administration and delivery, price, the level of competition and the availability of reimbursement from government and other third- party payers.
Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, have fewer or less severe side effects, are more convenient or are less expensive than any products that we may develop. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market. In addition, we expect that our therapeutic products, if approved, will be priced at a significant premium over competitive products and our ability to compete may be affected in many cases by insurers or other third-party payers seeking to encourage the use of competitive products including biosimilar or generic products.
This increasingly competitive landscape may compromise the development of our product candidates. For example, improvements in the efficacy, delivery and success rates of competitors product candidates, in conjunction with a reduction in the price and duration of their treatments, diminished partnering interest from pharmaceutical companies in our product candidate TT-034 for the treatment of HCV. This caused us to announce in February 2016 the discontinuation of our program to develop TT-034 before the conclusion of its clinical trial, despite the promising clinical results regarding the safety of that product candidate achieved to date.
Government Regulation
As a pharmaceutical and biological product company that wishes to conduct clinical trials and ultimately obtain marketing approval in the United States, we are subject to extensive regulation by the FDA, and other federal, state, and local regulatory agencies. The Federal Food, Drug, and Cosmetic Act, or the FDC Act, the Public Health Service Act, or PHS Act, and their implementing regulations set forth, among other things, requirements for the research, testing, development, manufacture, quality control, safety, effectiveness, approval, labeling, storage, record keeping, reporting, distribution, import, export, advertising and promotion of our products. A failure to comply explicitly with any requirements during the product development, approval, or post-approval periods, may lead to administrative or judicial sanctions. These sanctions could include the imposition by the FDA or an IRB, of a suspension on clinical trials, refusal to approve pending marketing applications or supplements, withdrawal of approval, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties or criminal prosecution.
Although the discussion below focuses on regulation in the United States, we anticipate seeking approval for the testing and marketing of our products in other countries. Generally, our activities in other countries will be subject to regulation that is similar in nature and scope as that imposed in the United States, although there can be important differences. Additionally, some significant aspects of regulation in the European Union are addressed in a centralized way through the EMA, but country-specific regulation remains essential in many respects.
Government regulation may delay or prevent testing or marketing of our products and impose costly procedures upon our activities. The testing and marketing approval process, and the subsequent compliance with appropriate statutes and regulations, requires substantial time, effort, and financial resources, and we cannot be certain that the FDA or any other regulatory agency will grant marketing approvals for our products or any future products on a timely basis, if at all. The FDAs or any other regulatory agencys policies may change and additional governmental regulations may be enacted that could prevent or delay regulatory approval of our products or any future products or approval of new indications or label changes. We cannot predict the likelihood, nature or extent of adverse governmental regulation that might arise from future legislative, judicial, or administrative action, either in the United States or abroad.
100
Table of Contents
Recent Developments in Regulation of Gene Therapy
The FDA has provided guidance for the development of gene therapy products. For example, the FDA has established the Office of Tissues and Advanced Therapies (formerly Office of Cellular, Tissue and Gene Therapies) within CBER, to consolidate the review of gene therapy and related products, and the Cellular, Tissue and Gene Therapies Advisory Committee to advise CBER on its reviews. In addition, the FDA has issued a growing body of clinical guidelines, chemical, manufacturing and control, or CMC, guidelines, regenerative medicine guidelines a and other guidelines, all of which are intended to facilitate industrys development of gene therapy products.
In 2016, Section 3033 of the 21st Century Cures Act created a new product category called regenerative medicine advanced therapy, or the RMAT designation. The RMAT designation gives the sponsor of a new investigational biologic access to increased meeting opportunities with the FDA, in a manner comparable to those offered to sponsors of therapies designated as breakthrough therapies by the FDA. Because the designated products meet the criteria for unmet medical need in the treatment of a serious condition, they may be eligible for priority review, in which the initial assessment of the BLA is reduced from 12 months to eight months, and accelerated approval, which bases approval on an effect on a predictive surrogate endpoint or an intermediate clinical endpoint. RMATs qualifying for such accelerated approval may be able to satisfy licensing requirements through commitment to post-approval clinical studies as well as real-world data such as patient registries and health record analysis. The eligibility of the RMAT-designated product for these expedited programs can be discussed with the FDA at specific development meetings, but we do not know whether any of our current or future product candidates will be eligible for RMAT designation. We believe the increased access to the FDA during early development is a benefit for sponsors, because the typical Type B development meetings are normally restricted to one each at the stages of pre-IND, end of Phase II/pre-Phase III and pre-BLA submission. In addition, the option to qualify for a fast- track program, also based on the potential to serve an unmet medical need in the treatment of a serious condition, allows for a so-called rolling review of parts of the BLA, which can be submitted for assessment following agreement of a review timetable with CBER.
The FDA plans to include certain gene therapy products that permanently alter tissue and produce a sustained therapeutic benefit as part of the products that will meet the definition of being eligible to come under the pathway enabled by RMAT designation. RMAT designation enables gene therapy products to access the FDAs existing expedited programs to help foster the development and approval of gene therapy products. Our product candidates may not be eligible for RMAT designation now or in the future.
In May 2016, the EMA approved a second gene therapy product called Strimvelis, the first approved ex vivo stem cell gene therapy, to treat patients with a very rare disease called ADA-SCID (Severe Combined Immunodeficiency due to Adenosine Deaminase deficiency).
In August 2017, the FDA approved the first gene therapy product in the United States. The FDA approved Kymriah (tisagenlecleucel) for the treatment of certain pediatric and young adult patients with a form of acute lymphoblastic leukemia (ALL). Kymriah is a genetically-modified autologous T-cell immunotherapy. Because of the risk of cytokine release syndrome and neurological events, Kymriah is being approved with a REMS. In December 2017, the FDA approved Luxturna (voretigene neparvovec-rzyl), a gene therapy to treat children and adult patients with an inherited form of vision loss that may result in blindness. Luxturna is the first directly administered gene therapy approved in the United States that targets a disease caused by mutations in a specific gene. To date, the FDA has approved a total of four gene therapy products.
Marketing Approval
In the United States, for premarket approval purposes, the FDA regulates gene therapy products as biologics under the FDC Act, the PHS Act and related regulations.
101
Table of Contents
The steps required before a new biologic may be marketed in the United States generally include:
| | nonclinical pharmacology and toxicology laboratory and animal tests according to good laboratory practices, or GLPs, and applicable requirements for the humane use of laboratory animals or other applicable regulations; |
| | submission of an IND application which must become effective before human clinical trials may begin; |
| | adequate and well-controlled human clinical trials according to GCPs and any additional requirements for the protection of human research subjects and their health information to establish the safety and efficacy of the investigational product for each targeted indication; |
| | submission of a biologics license application, or BLA, to the FDA; |
| | FDAs pre-approval inspection of manufacturing facilities to assess compliance with cGMPs and, if applicable, the FDAs good tissue practices, or GTPs, for the use of human cellular and tissue products to prevent the introduction, transmission, or spread of communicable diseases; |
| | FDAs audit of clinical trial sites that generated data in support of the BLA; and |
| | FDA approval of a BLA, which must occur before a product can be marketed or sold. |
Product Development Process
Before testing any biologic in humans, the product enters the nonclinical, or preclinical, testing stage. Nonclinical tests include laboratory evaluations of product chemistry, toxicity, and formulation, as well as animal studies to assess the potential safety and activity of the product. The conduct of nonclinical tests must comply with federal regulations and requirements including GLPs.
Where a gene therapy trial is conducted at, or sponsored by, institutions receiving NIH funding for recombinant DNA research, prior to the submission of an IND to the FDA, a protocol and related documentation is submitted to and the trial is registered with the NIH Office of Science Policy, or OSP.
The product sponsor then submits the results of the nonclinical testing, together with manufacturing information, analytical data, any available clinical data or literature, and a proposed clinical protocol, to the FDA in an IND, which is a request for authorization from the FDA to administer an investigational product to humans. Some nonclinical testing may continue even after the IND application is submitted. IND authorization is required before interstate shipping and administration of any new product to humans that is not the subject of an approved BLA. The IND automatically becomes effective 30 days after receipt by the FDA unless the FDA, within the 30-day time period, raises concerns or questions about the conduct of the clinical trial and places the clinical trial on a clinical hold. In such case, the IND sponsor must resolve any outstanding concerns with the FDA before the clinical trial may begin. Further, an IRB for each site proposing to conduct the clinical trial must review and approve the plan for any clinical trial before it commences at that site. If the site has an IBC, it may also have to review and approve the proposed clinical trial. Clinical trials involve the administration of the investigational product to patients under the supervision of qualified investigators following GCPs, requirements meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, investigators, and monitors. Clinical trials are conducted under protocols that detail, among other things, the objectives of the clinical trial, dosing procedures, subject selection and exclusion criteria, the parameters to be used in monitoring safety, including stopping rules that assure a clinical trial will be stopped if certain adverse events should occur, and the efficacy criteria to be evaluated. Each protocol involving testing on U.S. patients and subsequent protocol amendments must be submitted to the FDA as part of the IND. The informed written consent of each participating subject is required and the form and content of the informed consent must be approved by each IRB.
102
Table of Contents
The clinical investigation of an investigational product is generally divided into three phases. Although the phases are usually conducted sequentially, they may overlap or be combined in some cases. The three phases of an investigation are as follows:
| | Phase I includes the initial introduction of an investigational product into humans. Phase I clinical trials may be conducted in patients with the target disease or condition or on healthy volunteers. These studies are designed to evaluate the safety, metabolism, pharmacokinetics and pharmacologic actions of the investigational product in humans, the side effects associated with increasing doses, and if possible, to gain early evidence on effectiveness. During Phase I clinical trials, sufficient information about the investigational products pharmacokinetics and pharmacological effects may be obtained to permit the design of Phase II clinical trials. The total number of participants included in Phase I clinical trials varies, but is generally in the range of 20 to 80. |
| | Phase II includes the controlled clinical trials conducted to evaluate the effectiveness of the investigational product for a particular indication(s) in patients with the disease or condition under study, to determine dosage tolerance and optimal dosage, and to identify possible adverse side effects and safety risks associated with the product. Phase II clinical trials are typically well- controlled, closely monitored, and conducted in a limited patient population, usually involving no more than several hundred participants. Phase IIa trials provide information on the impact of dose ranging on safety, biomarkers and proof of concept, while Phase IIb trials are patient dose-ranging efficacy trials. |
| | Phase III clinical trials are controlled clinical trials conducted in an expanded patient population at geographically dispersed clinical trial sites. They are performed after preliminary evidence suggesting effectiveness of the investigational product has been obtained, and are intended to further evaluate dosage, clinical effectiveness and safety, to establish the overall benefit-risk relationship of the product, and to provide an adequate basis for product approval. Phase III clinical trials usually involve several hundred to several thousand participants. In most cases, the FDA requires two adequate and well controlled Phase III clinical trials to demonstrate the efficacy of the product. FDA may accept a single Phase III trial with other confirmatory evidence in rare instances where the trial is a large multicenter trial demonstrating internal consistency and a statistically very persuasive finding of a clinically meaningful effect on mortality, irreversible morbidity or prevention of a disease with a potentially serious outcome and confirmation of the result in a second trial would be practically or ethically impossible. |
Annual progress reports detailing the results of the clinical trials must be submitted to the FDA. Written IND safety reports must be promptly submitted to the FDA and the investigators for serious and unexpected adverse events; any findings from other studies, tests in laboratory animals or in vitro testing that suggest a significant risk for human subjects; or any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator brochure. The sponsor must submit an IND safety report within 15 calendar days after the sponsor determines that the information qualifies for reporting. The sponsor also must notify the FDA of any unexpected fatal or life-threatening suspected adverse reaction within seven calendar days after the sponsors initial receipt of the information. The FDA typically recommends that sponsors observe subjects for potential gene therapy-related delayed adverse events for a 15-year period, including a minimum of five years of annual examinations followed by 10 years of annual queries, either in person or by questionnaire, of trial subjects.
The decision to terminate a clinical trial of an investigational biologic may be made by the FDA or other regulatory authority, an IRB, an IBC, or institutional ethics committee, or by a company for various reasons. The FDA may place a clinical hold and order the temporary, or permanent, discontinuation of a clinical trial at any time, or impose other sanctions, if it believes that the clinical trial either is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. If the FDA imposes a clinical hold, trials may not recommence without FDA and IRB authorization and then only under terms authorized by the FDA and IRB. In some cases, clinical trials are overseen by an independent group of qualified
103
Table of Contents
experts organized by the trial sponsor, or the clinical monitoring board or DSMB. This group provides authorization for whether or not a trial may move forward at designated check points. These decisions are based on the limited access to data from the ongoing trial. The suspension or termination of a clinical trial can occur during any phase of clinical trials if it is determined that the participants or patients are being exposed to an unacceptable health risk. In addition, there are requirements for the registration of ongoing clinical trials of drugs and biologics on public registries and the disclosure of certain information pertaining to the trials as well as clinical trial results after completion.
Assuming successful completion of all required testing in accordance with all applicable regulatory requirements, detailed investigational product information is submitted to the FDA in the form of a BLA for a biologic to request marketing approval for the product in specified indications.
Human gene therapy products are a new category of therapeutics. Because this is a relatively new and expanding area of novel therapeutic interventions, there can be no assurance as to the length of the trial period, the number of patients the FDA will require to be enrolled in the trials in order to establish the safety, efficacy, purity and potency of human gene therapy products, or that the data generated in these trials will be acceptable to the FDA to support marketing approval. The NIH and the FDA have a publicly accessible database, the Genetic Modification Clinical Research Information System, which includes information on gene transfer trials and serves as an electronic tool to facilitate the reporting and analysis of adverse events on these trials. Over the last several years the FDA has issued helpful guidance on development of gene therapy products and shown a willingness to work closely with developers, especially with those working in orphan disease areas.
Biologics License Application Approval Process
In order to obtain approval to market a biologic in the United States, a BLA must be submitted to the FDA that provides data from nonclinical studies and clinical trials and manufacturing information establishing to the FDAs satisfaction the safety, purity, and potency or efficacy of the investigational product for the proposed indication. The BLA must be accompanied by a substantial user fee payment unless a waiver or exemption applies.
The FDA will initially review the BLA for completeness before it accepts it for filing. Under the FDAs procedures, the agency has 60 days from its receipt of a BLA to determine whether the application will be accepted for filing based on the agencys threshold determination that the application is sufficiently complete to permit substantive review. After the BLA submission is accepted for filing, the FDA reviews the BLA to determine, among other things, whether the proposed product is safe, pure and potent, which includes determining whether it is effective for its intended use, and whether the product is being manufactured in accordance with cGMP, to assure and preserve the products identity, safety, strength, quality, potency and purity, and in accordance with biological product standards. The FDA will inspect the facilities at which the product is manufactured to ensure the manufacturing processes and facilities are in compliance with cGMP requirements and are adequate to assure consistent production of the product within required specifications. For a human cellular or tissue product, the FDA also will not approve the product if the manufacturer is not in compliance with the GTPs. These are FDA regulations that govern the methods used in, and the facilities and controls used for, the manufacture of human cells, tissues, and cellular and tissue based products, which are human cells or tissue intended for implantation, transplant, infusion, or transfer into a human recipient. The primary intent of the GTP requirements is to ensure that cell and tissue based products are manufactured in a manner designed to prevent the introduction, transmission, and spread of communicable disease. Additionally, before approving a BLA, the FDA may inspect one or more clinical sites to assure compliance with GCP.
If the FDA determines the application, manufacturing process or manufacturing facilities are not acceptable, it typically will outline the deficiencies and often will request additional testing or information, or corrective action for a manufacturing facility. This may significantly delay further review of the application. If the FDA finds that a clinical site did not conduct the clinical trial in accordance with GCP, the FDA may determine the
104
Table of Contents
data generated by the clinical site should be excluded from the primary efficacy analyses provided in the BLA. Additionally, notwithstanding the submission of any requested additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval.
The FDA may refer applications for novel products or products that present difficult questions of safety or efficacy to an advisory committee. The FDA also may determine a REMS is necessary to assure the safe use of the biologic, in which case the BLA sponsor must submit a proposed REMS. The REMS may include, but is not limited to, a Medication Guide, a communications plan, and other elements to assure safe use, such as restrictions on distribution, prescribing, and dispensing.
After the FDA completes its initial review of a BLA, it will either license, or approve, the product, or issue a complete response letter to communicate that it will not approve the BLA in its current form and to inform the sponsor of changes that the sponsor must make or additional clinical, nonclinical or manufacturing data that must be received before the FDA can approve the application, with no implication regarding the ultimate approvability of the application. If a complete response letter is issued, the sponsor may either resubmit the BLA, addressing all of the deficiencies identified in the letter, or withdraw the application.
The testing and approval process for both a drug and biologic requires substantial time, effort and financial resources and this process may take several years to complete. Data obtained from clinical trials is not always conclusive and may be susceptible to varying interpretations, which could delay, limit or prevent regulatory approval. The FDA may not grant approval on a timely basis, or at all. We may encounter difficulties or unanticipated costs in our efforts to secure necessary governmental approvals, which could delay or preclude us from marketing our products.
Orphan Drug Designation
Under the Orphan Drug Act, the FDA may grant orphan designation to a drug or biological product candidate intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the United States, or more than 200,000 individuals in the United States and for which there is no reasonable expectation that the cost of developing and making a drug or biological product available in the United States for this type of disease or condition will be recovered from sales of the product candidate. Orphan product designation must be requested before submitting a BLA. After the FDA grants orphan product designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. Orphan product designation does not convey any advantage in or shorten the duration of the regulatory review and approval process.
If a product candidate that has orphan designation subsequently receives the first FDA approval for the disease or condition for which it has such designation, the product is entitled to orphan product exclusivity, which means that the FDA may not approve any other applications to market the same drug or biological product for the same indication for seven years, except in limited circumstances, such as a showing of clinical superiority to the product with orphan exclusivity. Competitors, however, may receive approval of different products for the indication for which the orphan product has exclusivity or obtain approval for the same product but for a different indication than the one for which the orphan product has exclusivity. Orphan product exclusivity also could block the approval of one of our products for seven years if a competitor obtains approval of the same biological product as defined by the FDA or if our product candidate is determined to be contained within the competitors product for the same indication or disease. If a drug or biological product designated as an orphan product receives marketing approval for an indication broader than what is designated, it may not be entitled to orphan product exclusivity. Orphan drug status in the European Union has similar, but not identical, benefits.
Expedited Development and Review Programs
The FDA has a fast track program that is intended to expedite or facilitate the process for reviewing new drugs and biological products that meet certain criteria. Specifically, new drugs and biological products are
105
Table of Contents
eligible for fast track designation if they are intended to treat a serious or life-threatening condition and demonstrate the potential to address unmet medical needs for the condition. Fast track designation applies to the combination of the product and the specific indication for which it is being studied. The sponsor of a new biologic or drug may request the FDA to designate the biologic or drug as a fast track product at any time during the clinical development of the product. Unique to a fast track product, the FDA may consider for review sections of the marketing application on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the application, the FDA agrees to accept sections of the application and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the application.
Any product submitted to the FDA for marketing, including under a fast track program, may be eligible for other types of FDA programs intended to expedite development and review, such as priority review and accelerated approval. Any product is eligible for priority review if it would, if approved, be a significant improvement in the safety, effectiveness, treatment, diagnosis or prevention of a disease compared to marketed products. The FDA will attempt to direct additional resources to the evaluation of an application for a new biological or drug product designated for priority review in an effort to reduce the review period from 12 to eight months. Additionally, a product may be eligible for accelerated approval. Biological or drug products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may receive accelerated approval, which means that they may be approved on the basis of adequate and well-controlled clinical trials establishing that the product has an effect on a surrogate endpoint that is reasonably likely to predict a clinical benefit, or on the basis of an effect on an intermediate clinical endpoint. As a condition of approval, the FDA may require that a sponsor of a biological or drug product receiving accelerated approval perform adequate and well-controlled post-marketing clinical trials. In addition, the FDA currently requires as a condition for accelerated approval pre-approval of promotional materials, which could adversely impact the timing of the commercial launch of the product. Lastly, under the provisions of the new Food and Drug Administration Safety and Innovation Act, enacted in 2012, a sponsor can request designation of a product candidate as a breakthrough therapy. A breakthrough therapy is defined as a drug or biological that is intended, alone or in combination with one or more other drugs or biological, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug or biological may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. Drugs and biologicals designated as breakthrough therapies receive the same benefits as drugs and biologicals with Fast Track designation. In addition, the FDA must take certain additional actions, such as intensive guidance on an efficient drug development program (beginning as early as Phase 1), and organizational commitment involving senior managers, intended to expedite the development and review of an application for approval of a breakthrough therapy.
Fast Track designation and breakthrough therapy designation may expedite the product development and approval process, and priority review may expedite the approval process. However, these three paths do not change the standards for approval. Accelerated approval designation changes the standards for product approval and thus may expedite the development and/or approval process.
FDA Additional Requirements
The FDA may require, or companies may pursue, additional clinical trials after a product is approved. These so-called Phase 4 clinical trials may be made a condition to be satisfied for continuing drug and biologic approval. The results of Phase 4 clinical trials can confirm the efficacy of a product candidate and can provide important safety information. In addition, the FDA has expressed statutory authority to require sponsors to conduct post-market studies to specifically address safety issues identified by the agency.
Even if a product candidate receives regulatory approval, the approval may be limited to specific disease states, patient populations and dosages, or might contain significant limitations on use in the form of warnings,
106
Table of Contents
precautions or contraindications, or in the form of an onerous REMS, restrictions on distribution, or post-marketing study requirements. Further, even after regulatory approval is obtained, later discovery of previously unknown problems with a product may result in restrictions on the product or even complete withdrawal of the product from the market. In addition, we cannot predict what adverse governmental regulations may arise from future U.S. or foreign governmental action.
Medical Device Requirements
Our contemplated diagnostics, for use with certain of our therapeutic products, are regulated by FDA as in vitro diagnostic, or IVD, medical devices. Such IVD devices must comply with applicable FDA IVD-specific regulations as well as FDA regulations applicable more broadly to medical devices. These FDA regulations include requirements for registering establishments with FDA; listing IVD devices with FDA; reporting certain adverse events related to IVD devices to FDA; complying with the Quality System Regulation (current good manufacturing practices for devices); labeling IVD devices; and obtaining premarket approval or clearance prior to marketing IVD devices (unless exempt). There are also regulations covering the requirements for investigational devices and the conduct of clinical investigations of devices. Like drugs and biologics, failure to comply with applicable device/IVD requirements can result in legal or administrative enforcement actions against an IVD device firm, its officers or employees, and/or its products.
FDA Post-Approval Requirements
Any products manufactured or distributed by us or on our behalf pursuant to FDA approvals are subject to continuing regulation by the FDA, including requirements for record-keeping, reporting of adverse experiences with the biologic or drug, and submitting biological product deviation reports to notify the FDA of unanticipated changes in distributed products. Manufacturers are required to register their facilities with the FDA and certain state agencies, and are subject to periodic announced or unannounced inspections by the FDA and certain state agencies for compliance with cGMP requirements, which impose certain quality processes, manufacturing controls and documentation requirements upon us and our third-party manufacturers in order to ensure that the product is safe, has the identity and strength, and meets the quality, purity and potency characteristics that it purports to have. In November 2013, the Drug Quality and Security Act, or DQSA, became law and establishes requirements to facilitate the tracing of prescription drug and biological products through the supply distribution chain. This law includes a number of new requirements that are being implemented over time and require us to devote additional resources to satisfy these requirements, including serializing the product and using new technology and data storage to electronically trace the product from manufacturer to dispenser. If our products are not covered by the serialization and tracing requirements of the DQSA, they may be subject to state pedigree and traceability requirements. We cannot be certain that we or our present or future suppliers will be able to comply with the cGMP and other FDA regulatory requirements. If our present or future suppliers are not able to comply with these requirements, the FDA may halt our clinical trials, refuse to approve any BLA, force us to recall a product from distribution, shut down manufacturing operations or withdraw approval of the applicable BLA. Noncompliance with cGMP or other requirements can result in issuance of warning or untitled letters, civil and criminal penalties, seizures, and injunctive action.
The FDA and other federal and state agencies closely regulate the labeling, marketing and promotion of drugs and biologics. Government regulators, including the Department of Justice and the Office of the Inspector General of the Department of Health and Human Services, as well as state authorities, recently have increased their scrutiny of the promotion and marketing of drugs and biologics. While doctors are free to prescribe any product approved by the FDA for any use, a company can only make claims relating to safety and efficacy of a product that are consistent with FDA approval, and the company is allowed to market a product only for the particular use and treatment approved by the FDA. In addition, any claims we make for our products in advertising or promotion must, among other things, be appropriately balanced with important safety information and otherwise be adequately substantiated. Failure to comply with these requirements can result in adverse publicity, warning or untitled letters, corrective advertising, injunctions, potential civil and criminal penalties,
107
Table of Contents
criminal prosecution, and agreements with governmental agencies that materially restrict the manner in which a company promotes or distributes products.
Pediatric Research Equity Act
Under the Pediatric Research Equity Act, or PREA, as amended, a BLA or supplement must contain data to assess the safety and efficacy of the product for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. Manufacturers must submit a pediatric study plan to the IND not later than 60 days after the end-of-phase 2 meeting with the FDA; if there is no such meeting, before the initiation of any phase 3 studies or a combined phase 2 and phase 3 study; or if no such study will be conducted, no later than 210 days before a marketing application or supplement is submitted. The intent of PREA is to compel sponsors whose products have pediatric applicability to study those products in pediatric populations, rather than ignoring pediatric indications for adult indications that could be more economically desirable. The FDA may grant deferrals for submission of data or full or partial waivers. By its terms, PREA does not apply to any product for an indication for which orphan designation has been granted, unless the FDA issues regulations stating otherwise. Because the FDA has not issued any such regulations, submission of a pediatric assessment is not required for an application to market a product for an orphan-designated indication. In a July 2018 guidance, the FDA announced that it does not expect to grant any additional orphan drug designations to drugs for pediatric subpopulations of common diseases (i.e., diseases or conditions with an overall prevalence of 200,000 or greater). Pediatric subpopulation orphan designations that have already been granted will not be affected by this change.
Patent Term Restoration and Marketing Exclusivity
Depending on the timing, duration and specifics of FDA marketing approval of our product candidates, some of our U.S. patents may be eligible for limited patent term extension under the Hatch-Waxman Amendments. The Hatch-Waxman Amendments permit a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the products approval date. The patent term restoration period is generally one-half the time between the effective date of an IND and the submission date of a BLA plus the time between the submission date of a BLA and the approval of that application. Only one patent applicable to an approved biological is eligible for the extension and the application for the extension must be submitted prior to the expiration of the patent and within sixty days of approval of the biological product. The USPTO, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration.
The Biologics Price Competition and Innovation Act of 2009, which was included within the Patient Protection and Affordable Care Act, created an abbreviated approval pathway for biological products shown to be similar to, or interchangeable with, an FDA-licensed reference biological product, and grants a reference biologic twelve years of exclusivity from the time of first licensure. Biosimilarity, which requires that there be no clinically meaningful differences between the biological product and the reference product in terms of safety, purity, and potency, can be shown through analytical studies, animal studies, and a clinical study or studies. Interchangeability requires that a product is biosimilar to the reference product and the product must demonstrate that it can be expected to produce the same clinical results as the reference product and, for products administered multiple times, the biologic and the reference biologic may be switched after one has been previously administered without increasing safety risks or risks of diminished efficacy relative to exclusive use of the reference biologic. However, complexities associated with the larger, and often more complex, structure of biological products, as well as the process by which such products are manufactured, pose significant hurdles to implementation that are still being worked out by the FDA.
Pediatric exclusivity is another type of exclusivity in the United States. Pediatric exclusivity, if granted, provides an additional six months of exclusivity to be attached to any existing marketing exclusivity, e.g., twelve
108
Table of Contents
year exclusivity, or patent protection for a drug. This six month exclusivity, which runs from the end of other exclusivity protection or patent delay, may be granted based on the voluntary completion of a pediatric trial in accordance with an FDA-issued Written Request for such a trial.
Government regulation outside the United States
In addition to regulations in the United States, we will be subject to a variety of regulations in other jurisdictions governing, among other things, clinical trials and any commercial sales and distribution of our products.
Whether or not we obtain FDA approval for a product, we must obtain the requisite approvals from regulatory authorities in foreign countries prior to the commencement of clinical trials or marketing of the product in those countries. Certain countries outside of the United States have a similar process that requires the submission of a clinical trial application much like the IND prior to the commencement of human clinical trials. In the European Union, for example, a request for a clinical trial authorization, or CTA, must be submitted to each countrys national health authority and an independent ethics committee, much like the FDA and the IRB, respectively. Once the CTA is approved in accordance with a countrys requirements, clinical trial development may proceed.
The requirements and process governing the conduct of clinical trials, product approval or licensing, pricing and reimbursement vary from country to country. In all cases, the clinical trials are conducted in accordance with GCP and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki.
To obtain regulatory approval of a biological product under European Union regulatory systems, we must submit a marketing authorization application. The application required in the European Union is similar to a BLA in the United States, with the exception of, among other things, country-specific document requirements. The European Union also provides opportunities for market exclusivity. For example, in the European Union, upon receiving marketing authorization, a new biological generally receives eight years of data exclusivity and an additional two years of market exclusivity. If granted, data exclusivity prevents regulatory authorities in the European Union from referencing the innovators data to assess a biosimilar application. During the additional two-year period of market exclusivity, a biosimilar marketing authorization can be submitted, and the innovators data may be referenced, but no biosimilar product can be marketed until the expiration of the market exclusivity. The innovator may obtain an additional one year of market exclusivity if the innovator obtains an additional authorization during the initial eight year period for one or more new indications that demonstrate significant clinical benefit over existing therapies. This data and market exclusivity regime in the European Union of a total of 10 or 11 years protects against generic competition, but does not protect against the launch of a competing product if the competitor, rather than referencing the clinical data of the originator, has conducted its own clinical trials to support its marketing authorization application.
Orphan drugs in the European Union are eligible for 10-year market exclusivity. This 10-year market exclusivity may be reduced to six years if, at the end of the fifth year, it is established that the product no longer meets the criteria for orphan designation, for example, if the product is sufficiently profitable not to justify maintenance of market exclusivity. Additionally, marketing authorization may be granted to a similar product for the same indication at any time if:
| | the second applicant can establish that its product, although similar, is safer, more effective or otherwise clinically superior; |
| | the applicant consents to a second orphan medicinal product application; or |
| | the applicant cannot supply enough orphan medicinal product. |
109
Table of Contents
If we fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Pharmaceutical Coverage, Pricing and Reimbursement
Sales of our products, when and if approved for marketing, will depend, in part, on the extent to which our products will be covered by third-party payers, such as federal, state, and foreign government healthcare programs, commercial insurance and managed healthcare organizations. These third-party payers are increasingly reducing reimbursements for medical products, biologicals, drugs and services. In addition, the U.S. government, state legislatures and foreign governments have continued implementing cost containment programs, including price controls, restrictions on coverage and reimbursement and requirements for substitution of interchangeable products. Adoption of price controls and cost containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit our net revenue and results. Decreases in third-party reimbursement for our product candidates or a decision by a third-party payer not to cover our product candidates could reduce physician usage of our products once approved and have a material adverse effect on our sales, results of operations and financial condition.
The containment of healthcare costs has become a priority of federal, state and foreign governments. Third-party payers are increasingly challenging the prices charged for drug products and medical services, examining the medical necessity and reviewing the cost effectiveness of drug products and medical services, in addition to questioning safety and efficacy. If these third-party payers do not consider our products to be cost-effective compared to other available therapies, they may not cover our products after FDA approval or, if they do, the level of payment may not be sufficient to allow us to sell our products at a profit.
In the United States and some foreign jurisdictions, there have been, and likely will continue to be, a number of legislative and regulatory changes and proposed changes regarding the healthcare system directed at broadening the availability of healthcare and containing or lowering the cost of healthcare. In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care Education Reconciliation Act, or the ACA, was enacted. The ACA includes measures that have significantly changed, and are expected to continue to significantly change, the way healthcare is financed by both governmental and private insurers. Other legislative changes have been proposed and adopted in the United States since the ACA was enacted. In August 2011, the Budget Control Act of 2011, among other things, created measures for spending reductions by Congress. A Joint Select Committee on Deficit Reduction, tasked with recommending a targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, was unable to reach required goals, thereby triggering the legislations automatic reduction to several government programs. This includes aggregate reductions of Medicare payments to providers up to 2% per fiscal year, which went into effect in April 2013 and will remain in effect through 2024 unless additional Congressional action is taken. In January 2013, former President Obama signed into law the American Taxpayer Relief Act of 2012, which, among other things, further reduced Medicare payments to several providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years.
Other Healthcare Laws
Although we currently do not have any products on the market, we may be subject to additional healthcare regulation and enforcement by the federal government and by authorities in the states and other countries in which we conduct our business. Such laws include, without limitation, state and federal anti-kickback, fraud and abuse, false claims, privacy and security and physician sunshine and open payment laws and regulations, many of which may become more applicable to us if our product candidates are approved and we begin commercialization. If our operations are found to be in violation of any of such laws or any other governmental regulations that apply to us, we may be subject to penalties, including, without limitation, administrative, civil and criminal penalties, damages, fines, disgorgement, the curtailment or restructuring of our operations,
110
Table of Contents
exclusion from participation in federal and state healthcare programs and imprisonment, any of which could adversely affect our ability to operate our business and our financial results.
Facilities
Our corporate headquarters and our research and development facility is located in Hayward, California, and consists of approximately 7,295 square feet of leased office space under a lease that expires in June 2022. We also have offices in Melbourne and Sydney, Australia.
Legal Proceedings
We are not currently a party to any material legal proceedings.
Employees
As of March 31, 2020, we had 15 full-time employees, 9 of whom have a Ph.D. or other post-graduate degrees. Of these full-time employees, 10 are engaged in research and development activities and 5 are engaged in finance, legal, human resources, facilities and general management. Our employees are located in Sydney and Melbourne, Australia and at our scientific laboratories located in Hayward, California. None of our employees is represented by any labor union. As of March 31, 2020, 12 of our employees were located in Hayward, California, and 3 of our employees were located in Sydney and Melbourne, Australia.
111
Table of Contents
Directors and Executive Officers
The following table sets forth information covering our current directors and executive officers.
| Name |
Age | Position |
||||
| Jerel Banks |
45 | Chief Executive Officer, Director | ||||
| Megan Boston |
48 | Executive Director, Director | ||||
| J. Kevin Buchi(1)(2)(3) |
64 | Director | ||||
| Peter Francis(1)(2)(3) |
64 | Director | ||||
| Edward Smith (1)(2)(3) |
49 | Director | ||||
| (1) | Member of the audit committee. |
| (2) | Member of the compensation committee. |
| (3) | Member of the nominating and corporate governance committee. |
Dr. Jerel Banks has been a Director since October 2016, Chairman of our Board since October 2017 and Chief Executive Officer since June 2018. Dr. Banks was formerly the Chief Investment Officer of Nant Capital, LLC. Prior to joining Nant Capital, LLC, Dr. Banks served as vice president, portfolio manager and research analyst for the Franklin Biotechnology Discovery Fund at Franklin Templeton Investments from 2012 to 2015. Prior to his tenure at Franklin Templeton Investments, he worked as a biotechnology senior equity research analyst at Sectoral Asset Management from 2011 to 2012. From 2008 to 2011, Dr. Banks worked as a biotechnology equity research analyst at Apothecary Capital, the healthcare investment management team for the family investment office of the Bass Family of Fort Worth, Texas. Dr. Banks began his career in investment management as a healthcare equity research associate at Capital Research Company where he was a member of the equity research team from 2006 to 2008. Dr. Banks earned an M.D. from the Brown University School of Medicine and a Ph.D. in Organic Chemistry from Brown University, and he holds an A.B. in Chemistry from Princeton University.
Dr. Banks experience in the healthcare industry and finance provides valuable experience and guidance to the Board.
Megan Boston has previously been Chief Executive Officer and Managing Director of several companies, including entities listed on the Australian Stock Exchange. With over 13 years of experience, Ms. Boston has been a director across a range of industries where she chaired company boards as well as board sub-committees particularly in the area of finance and risk management. Specifically, Ms. Boston has been a Director of Benitec since August 2016 and Executive Director since June 2018. From 2014 until joining Benitec, Ms. Boston was CEO of listed companies on the Australian Stock Exchange, Omni Market Tide Ltd and Rision Ltd. Previously, Ms. Boston held senior executive roles at various banking institutions in the area of risk and compliance, as well as working for PricewaterhouseCoopers. Ms. Boston holds a Bachelor of Commerce and is an Australian Chartered Accountant. Ms. Boston has also completed the company directors course diploma administered by the Australian Institute of Company Directors.
We believe Ms. Bostons operational and financial infrastructure experience gives her the necessary skills and qualifications to serve as a member of the Board.
J. Kevin Buchi has been a Director since April 2013. Mr. Buchi previously served as Chief Executive Officer of BioSpecifics Technologies Corp. and of TetraLogic Pharmaceuticals Corporation. Mr. Buchi served as Chief Executive Officer of Cephalon, Inc., or Cephalon, from December 2010 through its acquisition by Teva Pharmaceutical Industries Ltd in October 2011. After the acquisition Mr. Buchi served as Corporate Vice President, Global Branded Products of Teva Pharmaceuticals Industries Ltd. Mr. Buchi joined Cephalon in 1991 and held various positions, including Chief Operating Officer, from January 2010 to December 2010, Chief Financial Officer
112
Table of Contents
and Head of Business Development prior to being appointed Chief Executive Officer. Mr. Buchi is also on the board of directors of Amneal Pharmaceuticals, Inc. (NYSE: AMRX) and Dicerna Pharmaceuticals, Inc. (Nasdaq: DRNA. Mr. Buchi has a B.A. in chemistry from Cornell University and a Masters in Management from Kellogg Graduate School of Management at Northwestern University. He is also a Certified Public Accountant.
Based on his broad executive experience in the biotechnology industry, we believe Mr. Buchi has the appropriate skills and qualifications to serve on the Board.
Peter Francis has been a director since February 2006 and was previously Chairman of the Board until October 2017. Since 1993, Mr. Francis has been a partner at Francis Abourizk Lightowlers, a firm of commercial and technology lawyers with offices in Melbourne, Australia. He is a legal specialist in the areas of intellectual property and licensing and provides legal advice to corporations and research bodies. Mr. Francis completed his studies in law and jurisprudence at Monash University.
Based on his substantive intellectual property background and experience with our company, we believe Mr. Francis has the appropriate skills and qualifications to serve on the Board.
Edward Smith has been a director since April 2020. Edward brings more than 20 years of experience in executive finance and operations leadership in the biotechnology industry to our Board. Mr. Smith currently serves as the Chief Financial Officer of Marinus Pharmaceuticals, Inc. (Nasdaq: MRNS), a clinical-stage pharmaceutical company focused on developing and commercializing innovative therapeutics to treat patients suffering from rare seizure disorders. Prior to the start of his tenure at Marinus Pharmaceuticals in 2013, Mr. Smith served as the CFO of PolyMedix, Inc., and before his time at PolyMedix, Inc. he served as the executive director of finance at InKine Pharmaceutical Company, Inc.
Based on his extensive finance and operations background in the biotechnology industry, we believe Mr. Smith has the appropriate skill and qualifications to serve on the Board.
There are no family relationships among any of our directors or executive officers and no arrangements or understandings with major shareholders, customers, suppliers or others pursuant to which any of our directors or members of senior management was selected as such, except Dr. Banks was appointed by Nant Capital as a Director in October 2016 following the acquisition by Nant Capital, LLC of 19.9% of Benitecs then-outstanding ordinary shares.
The business addresses for each of our directors and executive officers is 3940 Trust Way, Hayward, California 9454.
Classified Board of Directors
The Companys business and affairs are managed under the direction of the Board. The Board currently consists of five directors, comprising the Companys Chief Executive Officer, Executive Director and three outside directors. The Board has determined that five directors, three of whom are independent, is the appropriate size for the Company. The number of directors is fixed from time to time by resolution of the Board pursuant to the Companys amended and restated certificate of incorporation (the Certificate).
Each of the Companys current directors will continue to serve as a director until the election and qualification of his or her successor, or until his or her earlier death, resignation, or removal.
The Companys Certificate provides that the Board is divided into three classes with staggered three-year terms. Only one class of directors is elected at each annual meeting of stockholders, with the other classes continuing for the remainder of their respective terms. The Board is designated as follows:
| | Mr. Smith is a Class I director, and his initial term will expire at the annual meeting of stockholders to be held in 2020; |
113
Table of Contents
| | Messrs. Buchi and Francis are Class II directors, and their initial terms will expire at the annual meeting of stockholders to be held in 2021; and |
| | Mr. Banks and Ms. Boston are Class III directors, and their terms will expire at the annual meeting of stockholders to be held in 2022. |
Any additional directorships resulting from an increase in the number of directors will be distributed among the three classes so that, as nearly as possible, each class will consist of one-third of the Companys directors.
Board Committees
The Board currently has, and appoints the members of, a standing Audit Committee, Compensation Committee, and Nominating and Corporate Governance Committee. Each of those committees has a written charter approved by the Board. The current charter for each standing Board committee is posted under Governance in the Investor Relations section of the Companys website, www.benitec.com.
The Board determined that each of Messrs. Buchi, Francis and Smith is independent under the Nasdaq listing standards, and Rule 10A-3 under the Exchange Act. Pursuant to Rule 10A-3(b)(1)(iv)(A)(2) under the Exchange Act, the Companys audit committee consists entirely of independent directors.
Audit Committee: Messrs. Buchi (Chairman), Francis and Smith. The purpose of the Audit Committee is to assist the board in fulfilling its oversight responsibilities with respect to (i) the integrity of the Companys financial statements, (ii) the Companys compliance with legal and regulatory requirements, and (iii) the independent auditors qualifications, independence and performance. The Audit Committee is also responsible for preparing a report to be included in the Companys annual proxy statement, advising and consulting the management and the Board regarding the Companys financial affairs, and appointing, overseeing and approving compensation for the work of the Companys independent auditors.
Our Board has determined that Messrs. Buchi, Francis and Smith each meet the independence requirements of Rule 10A-3 under the Exchange Act and the applicable Nasdaq rules. Our Board has determined that Mr. Smith is an audit committee financial expert as defined by applicable SEC rules and has the requisite financial sophistication as defined under the applicable Nasdaq rules.
Compensation Committee: Messrs. Francis (Chairman), Buchi and Smith. The Compensation Committee establishes and administers the Companys policies, programs and procedures for compensating and providing benefits to its executives and Board. The Committees responsibilities specifically include reviewing and approving the goals and objectives relevant to the chief executive officers and other executive officers compensation, evaluating the performance of the chief executive officer and other executive officers in light of those goals and objectives, and making recommendations to the Board with respect to non-employee director compensation. The Committee is also responsible for making recommendations to the Board with respect to incentive-compensation plans and equity-based plans.
Nominating and Corporate Governance Committee: Messrs. Francis (Chairman), Buchi and Smith. The Nominating and Corporate Governance Committee manages all aspects of the governance of the Companys Board. The Committees responsibilities include identifying individuals qualified to become members of the Board, recommending candidates to fill Board vacancies and newly created director positions, recommending whether incumbent directors should be nominated for re-election upon the expiration of their terms, recommending corporate governance guidelines applicable to the Board and to the Companys employees, overseeing the evaluation of the Board and its committees, and assessing and recommending Board members to the Board for committee membership.
Compensation Committee Interlocks and Insider Participation
No member of our compensation committee is or has been our current or former officer or employee. None of our executive officers served as a director or a member of a compensation committee (or other committee
114
Table of Contents
serving an equivalent function) of any other entity, one of whose executive officers served as a director or member of our compensation committee during the fiscal year ended June 30, 2020.
Code of Ethics and Business Conduct
We have established a Code of Ethics and Business Conduct as of April 12, 2020, which sets out the standards of behavior that apply to every aspect of our dealings and relationships, both within and outside the company. The following standards of behavior apply to all directors, executive officers and employees of the company:
| | comply with all laws that govern us and our operations; |
| | act honestly and with integrity and fairness in all dealings with others and each other; |
| | avoid or manage conflicts of interest; |
| | use our assets responsibly and in the best interests of the company; and |
| | be responsible and accountable for our actions. |
The Code of Ethics and Business Conduct is available on our website at www.benitec.com.
115
Table of Contents
As an emerging growth company and a smaller reporting company we have opted to comply with the scaled executive compensation disclosure rules applicable to smaller reporting companies.
Our named executive officers, or our NEOs, for the fiscal year ended June 30, 2020 were Dr. Jerel A. Banks, our Executive Chairman and Chief Executive Officer, and Megan Boston, our Executive Director.
The discussion of our compensation program prior to the completion of the Re-domiciliation reflects the compensation program of Benitec Limited, and of the Company for periods following the completion of the Re-domiciliation. This discussion may contain forward-looking statements that are based on our current plans, considerations, expectations and determinations regarding future compensation programs. Actual compensation programs that we adopt following the closing of this offering may differ materially from the currently planned programs summarized in this discussion.
Summary Compensation Table
The following table sets forth information regarding compensation earned by our NEOs during the fiscal year ended June 30, 2020 and the fiscal year ended June 30, 2019:
| Named Executive Officer and |
Fiscal Year |
Salary ($)(1) |
Bonus ($)(2) |
Option Awards ($) |
All Other Compensation ($) |
Total ($)(7) |
||||||||||||||||||
| Dr. Jerel A. Banks, M.D. Ph.D. |
2020 | 400,000 | | (2) | | 31,704 | (5) | 431,704 | ||||||||||||||||
| Executive Chairman and Chief Executive |
2019 | 400,000 | 200,000 | | 15,296 | (5) | 615,296 | |||||||||||||||||
| Megan Boston |
2020 | 221,430 | | (2) | | 16,775 | (6) | 238,205 | ||||||||||||||||
| Executive Director |
2019 | 185,543 | 117,975 | (4) | 74,737 | (3) | 16,463 | (6) | 394,718 | |||||||||||||||
| (1) | Ms. Bostons salary was paid in Australian dollars and has been converted to U.S. dollars using a conversion rate of A$1.00 to $0.671, and A$1.00 to $0.715, for the fiscal years ended June 30, 2020 and June 30, 2019, respectively. |
| (2) | Amounts reflect cash bonuses awarded to the NEOs after review by the Remuneration Committee of the board of directors of Benitec Limited (the Remuneration Committee), and individual performance for the prior fiscal year. The amount of cash bonuses earned in respect of the fiscal year ended June 30, 2020 is not calculable through the latest practicable date. The Compensation Committee expects to determine such bonuses in November 2020, and the amounts of these bonuses will be disclosed in a filing by the Company in a Current Report on Form 8-K under Item 5.02(f) once the amounts are determined. |
| (3) | Amount represents the aggregate grant date fair value of stock and option awards granted by the Company in the fiscal year ended June 30, 2019, computed in accordance with FASB ASC Topic 718. For further information on how we account for stock-based compensation, see Note 11 to the Companys consolidated financial statements for the year ended June 30, 2019 included in this prospectus. These amounts reflect the Companys accounting expense for these awards and do not correspond to the actual amounts, if any, that will be recognized by the NEOs. |
| (4) | Ms. Bostons performance-based cash bonus was paid in Australian dollars and has been converted to U.S. dollars using a conversion rate of A$1.00 to $0.715. |
| (5) | Amounts reflect company-paid health and life insurance premiums. |
| (6) | Amounts reflect the Companys compulsory contributions to Ms. Bostons superannuation account. The superannuation contributions were paid in Australian dollars and were converted to U.S. dollars using a conversion rate of A$1.00 to $0.671, and A$1.00 to $0.715, for the fiscal years ended June 30, 2020 and June 30, 2019, respectively. |
| (7) | The amounts reported in this table include compensation paid by Benitec Limited prior to the completion of the Re-domiciliation. |
116
Table of Contents
Narrative Disclosure to Summary Compensation Table
Annual Base Salary
We use base salaries to recognize the experience, skills, and responsibilities required of all of our employees, including our NEOs. Base salaries are reviewed annually by our Board. For the year ended June 30, 2020, the annual base salaries for each of Dr. Banks and Ms. Boston were $400,000 and A$330,000 ($221,430) (Ms. Bostons salary has been converted using a conversion rate of A$1.00 to $0.671), respectively.
Bonus Compensation
We have awarded our NEOs annual discretionary bonuses. Prior to the completion of the Re-domiciliation, the Remuneration Committee determined the amount of any annual discretionary bonus payment following the completion of the applicable fiscal year, based on Benitec Limiteds overall performance and the achievement of key strategic objectives. For fiscal year 2019, performance bonuses were based on achieving certain clinical, development and operational milestones. The actual annual cash bonuses awarded to our NEOs for fiscal year 2019 performance are set forth above in the Summary Compensation Table in the column titled Bonus. For the fiscal year ended June 30, 2020, the annual discretionary bonuses are not calculable through the latest practicable date. The Compensation Committee expects to determine such bonuses in November 2020, and the amounts of these bonuses will be disclosed in a filing by the Company in a Current Report on Form 8-K under Item 5.02(f) once the amounts are determined
Equity or Equity-Linked Incentive Awards
Although we do not have a formal policy with respect to the grant of equity incentive awards to our NEOs, we believe that equity grants provide our NEOs with a strong link to our long-term performance, create an ownership culture and help to align the interests of our NEOs and our shareholders. In addition, we believe that equity grants with a time-based vesting feature promote executive retention because this feature incentivizes our NEOs to remain in our employment during the vesting period. The equity awards held by our NEOs are described in more detail in the Outstanding Equity Awards at 2019 Fiscal Year-End table below.
Employment Agreements with our NEOs.
We are a party to employment agreements with each of our NEOs. These employment agreements provide for at will employment and may be terminated at any time.
Employment Agreement with Dr. Jerel A. Banks, M.D. Ph.D.
In September 2018, Tacere Therapeutics, Inc., a subsidiary of the Company, entered into an employment agreement with Dr. Banks setting forth the terms of his employment as Executive Chairman and Chief Executive Officer of Benitec Limited. In connection with the Re-domiciliation, Dr. Banks was appointed Executive Chairman and Chief Executive Officer of the Company. The agreement provides for Dr. Banks employment and sets forth his (i) annual base salary, (ii) discretionary annual bonus, (iii) eligibility to participate in employee benefit plans, (iv) eligibility for accrued paid vacation, (v) expense reimbursements in accordance with Company policy, (vi) eligibility to participate in the Companys Share Option Plan (as defined below), (vii) post-employment obligations to refrain from soliciting our employees for one year following the end of employment, and (viii) certain non-disparagement obligations. Dr. Banks employment agreement also provides for confidentiality of information and ownership of proprietary property restrictions.
Pursuant to the employment agreement, Dr. Banks employment is at will and can be terminated at any time. However, the Company must provide Dr. Banks with at least six months prior notice (or pay in lieu of notice) prior to any termination. Dr. Banks may terminate his employment on no fewer than six months prior written notice to the Company.
117
Table of Contents
Notwithstanding any provisions in the employment agreement, the Company may terminate Dr. Banks employment immediately without prior notice to Dr. Banks if he (a) commits any serious or persistent breach of any of the provisions of the employment agreement, (b) commits any act of willful or serious misconduct or negligence in the discharge of his duties, (c) becomes of unsound mind or under the control of any committee or officer under any law relating to mental health, (d) is convicted of a felony, which in our reasonable opinion affects Dr. Banks position, or (e) becomes permanently incapacitated by accident or illness from performing duties under the employment agreement for a period aggregating more than three months in any six-month period, or for any period beyond three consecutive months.
Employment Agreement with Megan Boston
In July 2018, Benitec Limited entered into an employment agreement with Megan Boston for the position of Executive Director. In connection with entering into the employment agreement, Ms. Boston ceased serving as a non-executive director of the Company. Ms. Boston remains on the Board as an executive director. The employment agreement provides for Ms. Bostons employment and sets forth her (i) annual base salary, (ii) discretionary annual bonus, (iii) superannuation contribution, (iv) eligibility for accrued paid vacation, (v) expense reimbursements in accordance with Company policy, and (vi) post-employment obligations to refrain from soliciting our employees for one year following the end of employment. Ms. Bostons employment agreement also provides for confidentiality of information and ownership of proprietary property restrictions.
Pursuant to the employment agreement, Ms. Bostons employment is at will and can be terminated at any time However, the Company must provide Ms. Boston with at least six months prior notice (or pay in lieu of notice) prior to any termination. Ms. Boston may terminate her employment on no fewer than six months prior written notice.
Notwithstanding any provisions in the employment agreement, the Company may terminate Ms. Bostons employment immediately without prior notice to Ms. Boston if she (a) commits any serious or persistent breach of any of the provisions of the employment agreement, (b) commits any act of willful or serious misconduct or negligence in the discharge of her duties, (c) becomes bankrupt or makes any arrangement or composition with her creditors, (d) becomes of unsound mind or under the control of any committee or officer under any law relating to mental health, (e) is convicted of any criminal offense other than an offense which in our reasonable opinion does not affect Ms. Bostons position, or (f) becomes permanently incapacitated by accident or illness from performing her duties under the employment agreement for a period aggregating more than three months in any six-month period, or any period beyond three consecutive months.
Outstanding Equity Awards at Fiscal Year-End
The following table sets forth information regarding outstanding equity or equity-linked awards for each of our NEOs as of June 30, 2020.
| Option Awards (1) | ||||||||||||||||||||
| Named Executive Officer |
Grant Date | Number of Securities Underlying Unexercised Options (#)Exercisable |
Number of Securities Underlying Unexercised Options (#)Unexercisable |
Option Exercise Price ($) |
Option Expiration Date |
|||||||||||||||
| Dr. Jerel A. Banks, M.D. Ph.D. |
6/26/2018 | (2) | 22,222 | 11,111 | 50.73 | 6/26/2023 | ||||||||||||||
| Executive Chairman and Chief Executive Officer |
||||||||||||||||||||
| Megan Boston |
3/12/2019 | (2) | 5,555 | 11,111 | 42.33 | 3/12/2024 | ||||||||||||||
| Executive Director |
||||||||||||||||||||
118
Table of Contents
| (1) | All of the awards in this table were granted under the Share Option Plan, the terms of which are described below under Equity Incentive Compensation PlansShare Option Plan. The share amounts and exercise prices of the awards shown in this table have been adjusted to reflect the terms of the Re-domiciliation. |
| (2) | The shares subject to each of the option awards vest in substantially equal installments on each of the first, second and third anniversaries of the grant date, generally subject to continued employment through the applicable vesting date. |
Other Elements of Compensation
Other Benefits and Perquisites
We offer participation in broad-based retirement, health and welfare plans to all of our colleagues, including our NEOs.
We maintain a tax-qualified defined contribution retirement plan that provides eligible U.S. employees (including Dr. Banks) with an opportunity to save for retirement on a tax-advantaged basis. Plan participants are eligible to defer eligible compensation subject to applicable annual Internal Revenue Code limits. The 401(k) plan is intended to be qualified under Section 401(a) of the Internal Revenue Code with the 401(k) plans related trust intended to be tax exempt under Section 501(a) of the Internal Revenue Code. As a tax-qualified retirement plan, contributions to the 401(k) plan and earnings on those contributions are not taxable to the employees until distributed from the 401(k) plan.
We contribute to the Australian superannuation scheme that provides eligible Australian employees (including Ms. Boston) with an opportunity to save for retirement on a tax-advantaged basis. We pay superannuation in accordance with legislative requirements and our minimum contribution is set by legislation. We offer flexibility for salary sacrifice to be added to the superannuation scheme and any actual increase in our contribution to the superannuation scheme is subject to legislative rules at the time.
Termination or Change in Control Benefits
The employment agreements with our NEOs provide for specified notice periods (or pay in lieu of notice) if the Company terminates the employment of our NEOs under certain circumstances, as described above in the Employment Agreements with our NEOs section. Our NEOs are not eligible to receive any additional payments or benefits in connection with their termination of employment or in connection with the Companys change in control.
Equity Incentive Compensation Plan
Benitec Officers and Employees Share Option Plan (Share Option Plan)
On the Implementation Date, the Re-domiciliation of Benitec Limited was completed, resulting in the Company becoming the ultimate parent company of the Benitec group of companies (the Scheme). Pursuant to the Scheme, the Company assumed Benitec Limiteds obligations with respect to the settlement of options (the Options) that were issued by Benitec Limited prior to the Implementation Date pursuant to the Share Option Plan. This includes the Companys assumption of the Share Option Plan and all award agreements pursuant to which each of the Options were granted. Accordingly, any exercise of the Options (subject to vesting conditions being met) entitles the holder to one share of the Companys Common Stock for every 300 ordinary shares of Benitec Limited the holder of the Option would have been entitled to receive upon exercise of such Option. The exercise price of the Options was also adjusted in connection with the Scheme. Other than as described above, the terms of the Share Option Plan and the Options remained unchanged following the Implementation Date.
Following the Implementation Date, no new options or other equity awards will be issued under the Share Option Plan. The Company intends to present a new equity incentive plan for approval by the Companys stockholders at the Companys next annual meeting of stockholders.
119
Table of Contents
The following summarizes the material terms of the Share Option Plan.
Administration. Our Board, or duly authorized persons, administers the Share Option Plan. Under the Share Option Plan, the Board has power to determine appropriate procedures for administration consistent with the Share Option Plan.
Eligibility. The Share Option Plan provides for an employee (including any director, a part-time employee or consultant) of the Company and its subsidiaries to be deemed an eligible employee by the Board (Eligible Employee).
Application for Options. The Board may from time to time, in its absolute discretion, issue written invitations to Eligible Employees to apply for up to a specified number of Options. The invitation will include: (i) the method of calculation of the exercise price, (ii) the period or periods during which all or any of the Options may be exercised, (iii) the number of shares over which the Option may be granted, (iv) the dates and times when the Options expire, (v) any Exercise Conditions (as defined in the Share Option Plan) that apply to the Options, and (vi) the date and time by which the application for Options must be received by the Company. Upon receipt of the invitation the Eligible Employee must send the person identified by the Company to receive the application, the Eligible Employees completed application. The application must be received from the Eligible Employee within a specified period listed in the invitation. The Eligible Employee may apply for the number of Options specified in the invitation or part thereof (but the application must be made in multiples of 1,000 Options).
Authorized Shares. The maximum number of shares that may be issued pursuant to awards under Share Option Plan, subject to certain adjustments is 5% of the current number the Companys ordinary shares.
Grant. Once the Company has received and accepted the Eligible Employees application for Options, the Company may: (i) grant the Options to the Eligible Employee or (ii) procure the grant of the Options by a third party. The grant will be based on the terms set out in the Share Option Plan and upon such additional terms and conditions as the Board determines. Options will have a maximum exercise period of seven years. Notwithstanding the above, following the Implementation Date, no new options or other equity awards will be issued under the Share Option Plan.
Transferability. The invitation to receive Options is not transferable and an Eligible Employee may only apply for the Options on his or her behalf and not on behalf of another person or entity unless that other person or entity is controlled by or otherwise associated with the Eligible Employee, agrees to be bound by the Share Option Plan and the terms of issue of the Options as if it were an Eligible Employee, and is approved by the Board. The Option may be transferred or encumbered: (i) by force of law upon death to the Participants legal personal representative, (ii) upon bankruptcy to the Participants trustee in bankruptcy, or (iii) with the prior written approval of the Board.
Termination of Employment. If a Participant (as defined in the Share Option Plan), who was an employee of the Company or one of its subsidiaries, dies prior to exercising the Option or the Participant terminates employment by reason of Retirement (as defined in the Share Option Plan), the Board may exercise discretion to allow the Options to be exercised by the Participant for a period of up to six months following the Participants termination due to death or retirement, even if certain Exercise Conditions had not been met. If a Participant, who was an employee of the Company or one of its subsidiaries, terminates employment for any other reason, the Board may exercise discretion to allow the Options to be exercised by the Participant for a period of up to three months following the Participants termination of employment, even if certain Exercise Conditions had not been met. If a Participant, who was an employee of the Company or one of its subsidiaries, dies prior to exercising the Option, the Participant terminates employment by reason of Retirement or terminates employment for any other reason, and the Exercise Conditions have been met, the Participant may exercise the Options for a period up to 12 months following termination, with such period to be determined by the Board.
120
Table of Contents
Notwithstanding any of the foregoing, if, in the opinion of the Board, a Participant acts fraudulently or dishonestly or is in breach of his or her obligations to the Company or any of its subsidiaries, then the Board of Directors may deem any unexercised Options held by the Participant to have lapsed.
Transactions. If a Takeover Bid (as defined in the Share Option Plan) is made or a Change in Control (as defined in the Share Option Plan) occurs, the Board may (unless, in the opinion of the Board, an intention to make an equivalent offer to the Participants to acquire all or substantial portion of their Options is given) give written notice to each Participant of the Takeover Bid or Change in Control. If a Takeover Bid is made or a Change in Control occurs after the Exercise Condition has been met, but before an Option has been exercised, the Option will be exercisable for a period of 30 days from the date of the notice. If a Takeover Bid is made or a Change in Control occurs before an Exercise Condition has been met, the Board may determine that all or part of those Options will be exercisable for a period of 30 days from the date of the notice. The Board may also exercise its discretion to permit the exercise of the Options upon certain other corporate transactions, including but not limited to the voluntary winding up of the Company, where an order is made for the compulsory winding up of the Company, or a person becoming bound or entitled to acquire shares in the Company under certain provisions of the Australian Corporations Act. If an Acquiring Company (as defined in the Share Option Plan) obtains control of the Company as a result of a Takeover Bid, a Change in Control or a proposed scheme or arrangement between the Company and its shareholders, and both the Company and the Acquiring Company agree, a Participant may upon the exercise of the Participants Options, the Participant may elect to acquire shares in the Acquiring Company or its parent, on substantially the same terms and subject to substantially the same conditions as the Participant may exercise Options to acquire shares, but with appropriate adjustments to the number and kind of shares subject to the Options, as well as to the exercise price.
If the Company issues shares pro rata to the Companys shareholders by way of bonus issue, Participants will be entitled to exercise Participants Options, to receive in addition to the shares in respect of which the Options are exercised and without the payment of any further consideration, an allotment of as many additional shares as would have been issues to a shareholder who, on the date for determining entitlements under the bonus issue, held shares equal in number to the shares in respect of which the Options are exercised.
In the event of certain other transactions, including if shares are offered pro rata for subscription by the Companys shareholders or upon the reorganization of the Company, the number of Options to which each Participant is entitled to or the exercise price of the Options or both as appropriate will be adjusted.
Plan Amendment. Our Board may amend the Share Option Plan at any time. However, no amendment to the Share Option Plan or the Options may be made which reduces the rights of Participants in respect of Options granted to them prior to the date of the amendment, other than in certain limited situations, including correction of error or as required by Australian law.
121
Table of Contents
The following table shows the total compensation paid to the Companys directors for the year ended June 30, 2020.
| Name | Fees Earned or Paid in Cash ($)(2) |
All Other Compensation ($)(3) |
Total ($) |
|||||||||
| J. Kevin Buchi |
51,432 | | 51,432 | |||||||||
| Peter Francis |
46,970 | 4,462 | 51,432 | |||||||||
| Edward F. Smith(4) |
10,417 | | 10,417 | |||||||||
| (1) | For information regarding the compensation of Dr. Banks and Ms. Boston, see Summary Compensation Table. |
| (2) | Fees paid to Mr. Buchi and Francis were paid in Australian dollars and has been converted to U.S. dollars using a conversion rate of A$1.00 to $0.671 for the fiscal year ended June 30, 2020. |
| (3) | Amounts reflect the Companys compulsory contributions to Mr. Franciss superannuation account. The superannuation contribution was paid in Australian dollars and has been converted to U.S. dollars using a conversion rate of A$1.00 to $0.671 for the fiscal year ended June 30, 2020. |
| (4) | Mr. Smith was appointed to the Board on April 14, 2020. Amounts reflect the total fees paid to Mr. Smith for his service in fiscal year 2020. |
For each director, the aggregate number of option awards outstanding at fiscal year-end for the year ended June 30, 2020 is set forth below:
| Name |
Option Awards (#) |
|||
| J. Kevin Buchi |
2,800 | |||
| Peter Francis |
4,667 | |||
Narrative Disclosure to Director Compensation Table
Each of our non-employee directors is paid an annual fee in respect of their service on the Board. Mr. Buchis annual fee is $51,432. Mr. Francis is paid a base fee of $46,970. In addition, the Company makes a superannuation contribution on Mr. Francis behalf in the amount of $4,462. Fees paid to Mr. Buchi and Francis were paid in Australian dollars and have been converted to U.S. dollars using a conversion rate of A$1.00 to $0.671. In accordance with Mr. Smiths Director Appointment Letter, he will be entitled to earn an annual fee of $50,000 in respect of his service on the Board. Upon their appointment as executive officers and employees, Dr. Banks and Ms. Boston no longer receive annual fees with respect to their service on the Board.
122
Table of Contents
CERTAIN RELATIONSHIPS AND RELATED-PARTY TRANSACTIONS
The following includes a summary of transactions since July 1, 2017 to which we have been a party in which the amount involved exceeded or will exceed $120,000, and in which any of our directors, executive officers or, to our knowledge, beneficial owners of more than 5% of our capital stock or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest, other than equity and other compensation, termination, change in control and other arrangements, which are described under Executive and Director Compensation. We also describe below certain other transactions with our directors, executive officers and stockholders.
Nant Capital, LLC
| | In October 2016, Benitec Limited entered into a strategic relationship with Nant Capital. As part of that strategic relationship, Benitec Limited initially issued Nant Capital ordinary shares which became an aggregate of 195,372 shares of common stock following the Re-domiciliation for a combined total consideration of $6.1 million. Jerel Banks, our Chairman and Chief Executive Officer, was initially appointed to the board of directors of Benitec Limited by Nant Capital. In connection with his separation from Nant Capital, Nant Capital agreed to pay Dr. Banks an annual separation payment (starting with calendar year 2018), in cash not later than March 15th of each year, equal to a fixed percentage of the amount by which the net increase in the fair market value of the 293,058 shares of Company common stock that Nant Capital owns (the Nant Shares) exceeds a 10% hurdle with respect to each calendar year. Dr. Banks does not own (beneficially or of record) any of the Nant Shares, nor does he have any right to vote, sell or dispose of the Nant Shares (or cause them to be voted, sold or disposed of), but for purposes of calculating his separation payment he can require Nant Capital to treat some or all of them as having effectively been sold prior to the end of a calendar year performance period. Dr. Banks has no obligation or responsibility to manage the Nant Shares. The arrangement is in effect and Dr. Banks has not accrued any payments thereunder. The Company is not a party to this arrangement. |
| | In December 2016, Benitec Limited entered into an exclusive sublicense agreement with NantWorks, LLC, an affiliate of Nant Capital, pursuant to which we obtained a sublicense to intellectual property relating to our product candidates for the treatment of HNSCC, including BB-401. We terminated the exclusive sublicense agreement for convenience, with the termination effective as of June 2020, because the BB-401 program has been terminated. Prior to termination of the agreement, we were required to make periodic payments to NantWorks, LLC for so long as the agreement remained in effect, as well as contingent milestone and royalty payments in connection with the development and sale of the licensed technology. In fiscal year 2017, we made license payments to NantWorks, LLC of $30,000, and reimbursed patent expense of $35,014. In fiscal year 2018, we paid (i) $4,125, as reimbursement in patent application extension, (ii) $30,000 in annual maintenance fee and (iii) $30,000 in annual maintenance fee. |
| | In January 2017, Benitec Limited entered into a research collaboration agreement with Nant Capital pursuant to which we agreed to advance the clinical development of BB-401, a recombinant DNA construct that produces an antisense RNA with specificity against EGFR for the treatment of HNSCC. The research collaboration agreement with Nant Capital allocates the $4.1 million received from Nant Capital in the March 2017 private placement first to the development of BB-401 and BB-501. Nant Capital will have a controlling vote on the joint steering committee that directs and oversees such development of BB-401 and BB-501 until those funds have been expended. The BB-401 and BB-501 programs have been terminated and, as a result, this collaboration is no longer active. |
123
Table of Contents
Other Related Parties
| | Legal services at normal commercial rates totaling A$726, or $509, for fiscal 2019, A$8,212, or $6,079, for fiscal 2018 and A$191,050, or $146,841 for fiscal 2017 were provided by Francis Abourizk Lightowlers, a law firm in which Mr. Peter Francis is a partner and has a beneficial interest. |
| | Annabel West, the wife of Greg West, our former Chief Executive Officer, was employed by us as a part-time clerical and administrative assistant. Annabel West was paid wages of A$42,278, or $31,298, and A$36,248, or $27,860, respectively, for fiscal 2018 and fiscal 2017. |
Review and approval of related party transactions
Our related parties include our directors, director nominees, executive officers, holders of more than five percent of the outstanding shares of our common stock and the foregoing persons immediate family members. We review relationships and transactions in which the Company and our related parties are participants to determine whether such related persons have a direct or indirect material interest. As required under SEC rules, transactions that are determined to be directly or indirectly material to a related party are disclosed in the appropriate SEC filing. In addition, the Audit Committee reviews and approves any related party transaction that is required to be disclosed. Set forth below is information concerning transactions with our related parties that is required to be disclosed under SEC rules.
Indemnification and Severance agreements
We have entered into indemnification agreements with our directors and executive officers which require us to indemnify such individuals to the fullest extent permitted by Delaware law. Our indemnification obligations under such agreements are not limited in amount or duration. Certain costs incurred in connection with such indemnities may be recovered under certain circumstances under various insurance policies. Given that the amount of any potential liabilities related to such indemnities cannot be determined until a lawsuit has been filed against a director or executive officer, we are unable to determine the maximum amount of losses that we could incur relating to such indemnities. Historically, any amounts payable pursuant to such director and officer indemnities have not had a material negative effect on our business, financial condition or results of operations.
We have also entered into severance and change in control agreements with certain of our executives. These agreements provide for the payment of specific compensation benefits to such executives upon the termination of their employment with us.
124
Table of Contents
The following table sets forth certain information regarding the beneficial ownership of the Companys common stock as of August 10, 2020 by (i) each person or group of persons known by us to beneficially own more than five percent of our common stock, (ii) each of our named executive officers, (iii) each of our directors and (iv) all of our directors and executive officers as a group.
The following table gives effect to the shares of common stock issuable within 60 days of August 10, 2020 upon the exercise of all options and other rights beneficially owned by the indicated stockholders on that date. Beneficial ownership is determined in accordance with Rule 13d-3 promulgated under Section 13 of the Securities Exchange Act and includes voting and investment power with respect to shares. Percentage of beneficial ownership is based on 1,108,374 shares of common stock outstanding at the close of business on August 10, 2020. Except as otherwise noted below, each person or entity named in the following table has sole voting and investment power with respect to all shares of our common stock that he, she or it beneficially owns.
Unless otherwise indicated below, the address for each beneficial owner listed is c/o 3940 Trust Way, Hayward, California 94545.
| Name of Beneficial Owner |
Number of Shares Beneficially Owned |
Percentage of Shares Beneficially Owned |
||||||||||
| Before Offering | After Offering | |||||||||||
| 5% or Greater Stockholders: |
||||||||||||
| Nant Capital, LLC(1) |
293,058 | 26.4 | % | |||||||||
| Directors and Named Executive Officers: |
||||||||||||
| Jerel A. Banks(2) |
22,222 | 2.0 | % | |||||||||
| Megan Boston(3) |
5,888 | * | ||||||||||
| J. Kevin Buchi(4) |
7,627 | * | ||||||||||
| Peter Francis(5) |
9,704 | * | ||||||||||
| Edward Smith |
| * | ||||||||||
| All Executive Officers and Directors As a Group |
45,441 | 4.1 | % | |||||||||
| * | Represents beneficial ownership of less than one percent of the companys outstanding common stock. |
| (1) | Based on the information included in the Form 3 filed by Nant Capital, LLC on April 24, 2020. Represents 195,372 shares held directly by Nant Capital, LLC and 97,686 shares of common stock held by Merrill Lynch (Australia) Nominees Pty Limited for the account of Nant Capital, LLC. The address of the principal business office of Nant Capital, LLC is 9922 Jefferson Blvd, Culver City, CA, 90232. |
| (2) | Represents stock options to acquire 22,222 that have vested or will vest within 60 days of August 10, 2020. |
| (3) | Includes 333 shares held by Boston Super Invest Pty A/C Boston Family Super that Megan Boston has sole voting power over and stock options to acquire 5,555 shares of common stock that have vested or will vest within 60 days of August 10, 2020. |
| (4) | Includes 4,827 shares of common stock and stock options to acquire 2,800 shares of common stock that have vested or will vest within 60 days of August 10, 2020. |
| (5) | Includes 4,738 shares of common stock held by the Francis Family Superannuation Fund, 300 shares held directly by Mr. Francis, and stock options to acquire 4,666 shares of common stock that have vested or will vest within 60 days of August 10, 2020. |
| (6) | Includes 9,898 shares of common stock and stock options to acquire 35,243 shares of common stock that have vested or will vest within 60 days of August 10, 2020. |
125
Table of Contents
The following description of our securities is intended as a summary only. We refer you to our amended and restated certificate of incorporation and restated bylaws, which are incorporated by reference into this prospectus, and to the applicable provisions of the Delaware General Corporation Law (DGCL). This description may not contain all of the information that is important to you and is subject to, and is qualified in its entirety by reference to, our certificate of incorporation, our by-laws and the applicable provisions of the DGCL. For information on how to obtain copies of our certificate of incorporation and by-laws, see Where You Can Find More Information.
General
Our authorized capital stock consists of 10,000,000 shares of our common stock, par value $0.0001 per share. During the Re-domiciliation, all of the issued and outstanding ordinary shares of Benitec Limited were exchanged for newly issued shares of common stock of the Company, on the basis of one share of the Companys common stock for every 300 ordinary shares issued and outstanding. As a result of the Re-domiciliation, Benitec Limited is a wholly-owned subsidiary of the Company.
Common Stock
Dividend Rights. Subject to preferences that may be applicable to any then outstanding preferred stock, holders of the Companys common stock are entitled to receive dividends, if any, as may be declared from time to time by the Companys Board out of legally available funds. Dividends may be paid in cash, in property or in shares of common stock, subject to the provisions of the Certificate and applicable law. Declaration and payment of any dividend will be subject to the discretion of the Board. The time and amount of dividends will be dependent upon the Companys financial condition, operations, cash requirements and availability, debt repayment obligations, capital expenditure needs, restrictions in the Companys debt instruments, industry trends, the provisions of Delaware law affecting the payment of distributions to stockholders and any other factors the Board may consider relevant.
Voting Rights. Each holder of common stock is entitled to one vote for each share on all matters submitted to a vote of the stockholders, including the election of directors. The Companys stockholders do not have cumulative voting rights in the election of directors.
Liquidation Rights. In the event of the Companys liquidation, dissolution or winding up, holders of the Companys common stock are entitled to share ratably in the net assets legally available for distribution to stockholders after the payment of all of the Companys debts and other liabilities and the satisfaction of any liquidation preference granted to the holders of any then outstanding shares of preferred stock.
Rights and Preferences. Holders of the Companys common stock have no pre-emptive, conversion, subscription or other rights, and there are no redemption or sinking fund provisions applicable to the Companys common stock. The rights, preferences and privileges of the holders of the Companys common stock are subject to and may be adversely affected by the rights of the holders of shares of any series of preferred stock that the Company may designate in the future.
Fully Paid and Nonassessable. All outstanding shares of the Companys common stock are fully paid and non-assessable.
Annual Stockholder Meetings. The Certificate and Bylaws provide that annual stockholder meetings will be held at a date, place (if any) and time, as exclusively selected by the Board. To the extent permitted under applicable law, the Company may but is not obligated to conduct meetings by remote communications, including by webcast.
126
Table of Contents
Pre-Funded Warrants to be Issued as Part of this Offering
The following summary of certain terms and provisions of the pre-funded warrants that are being offered hereby is not complete and is subject to, and qualified in its entirety by, the provisions of the pre-funded warrant, the form of which is filed as an exhibit to the registration statement of which this prospectus forms a part. Prospective investors should carefully review the terms and provisions of the form of pre-funded warrant for a complete description of the terms and conditions of the pre-funded warrants.
Exercise Price and Duration. Each pre-funded warrant offered hereby will have an initial exercise price per share equal to $0.01 and will be exercisable for share of our common stock. The pre-funded warrants will be immediately exercisable and may be exercised at any time until the pre-funded warrants are exercised in full. The exercise price and number of shares of common stock issuable upon exercise is subject to appropriate adjustment in the event of stock dividends, stock splits, reorganizations or similar events affecting our common stock and the exercise price.
Exercisability. The pre-funded warrants will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed exercise notice accompanied by payment in full for the number of shares of our common stock purchased upon such exercise (except in the case of a cashless exercise as discussed below). Purchasers of the pre-funded warrants in this offering may elect to deliver their exercise notice following the pricing of the offering and prior to the issuance of the pre-funded warrants at closing to have their pre-funded warrants exercised immediately upon issuance and receive shares of common stock underlying the pre-funded warrants upon closing of this offering. A holder (together with its affiliates) may not exercise any portion of the pre-funded warrant to the extent that the holder would own more than 4.99% (or, at the election of the purchaser, 9.99%) of the outstanding common stock immediately after exercise, except that upon at least 61 days prior notice from the holder to us, the holder may increase the amount of ownership of outstanding stock after exercising the holders pre-funded warrants. No fractional shares of common stock will be issued in connection with the exercise of a pre-funded warrant. In lieu of fractional shares, we will round down to the next whole share.
Cashless Exercise. If, at the time a holder exercises its pre-funded warrants, a registration statement registering the issuance of the shares of common stock underlying the pre-funded warrants under the Securities Act is not then effective or available, then in lieu of making the cash payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder may elect instead to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined according to a formula set forth in the pre-funded warrants.
Exercise Limitation. A holder (together with its affiliates) may not exercise any portion of the pre-funded warrant to the extent that the holder would own more than 4.99% of the outstanding common shares immediately after exercise, except that upon at least 61 days prior notice from the holder to us, the holder may increase the amount of beneficial ownership of outstanding stock after exercising the holders pre-funded warrants up to 9.99% of the number of our common shares outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the pre-funded warrants. Purchasers of pre-funded warrants in this offering may also elect prior to the issuance of the pre-funded warrants to have the initial exercise limitation set at 9.99% of our outstanding common shares.
Transferability. Subject to applicable laws, a pre-funded warrant may be transferred at the option of the holder upon surrender of the pre-funded warrant to us together with the appropriate instruments of transfer.
Fractional Shares. No fractional common shares will be issued upon the exercise of the pre-funded warrants. Rather, the number of common shares to be issued will be rounded up to the nearest whole number.
Trading Market. There is no established public trading market for the pre-funded warrants, and we do not expect a market to develop. In addition, we do not intend to apply to list the pre-funded warrants on any national securities exchange or other nationally recognized trading system. Without an active trading market, the liquidity of the pre-funded warrants will be limited.
127
Table of Contents
Right as a Stockholder. Except as otherwise provided in the pre-funded warrants or by virtue of such holders ownership of shares of our common stock, the holders of the pre-funded warrants do not have the rights or privileges of holders of our common stock, including any voting rights, until they exercise their pre-funded warrants.
Fundamental Transaction. In the event of a fundamental transaction, as described in the pre-funded warrants and generally including any reorganization, recapitalization or reclassification of our common stock, the sale, transfer or other disposition of all or substantially all of our properties or assets, our consolidation or merger with or into another person, the acquisition of more than 50% of our outstanding common stock, or any person or group becoming the beneficial owner of 50% of the voting power represented by our outstanding common stock, the holders of the pre-funded warrants will be entitled to receive upon exercise of the pre-funded warrants the kind and amount of securities, cash or other property that the holders would have received had they exercised the pre-funded warrants immediately prior to such fundamental transaction.
Amendment and Waiver. A pre-funded warrant may be modified or amended or the provisions thereof waived with the written consent of our company and the holder of the pre-funded warrant.
Warrants
In September of 2019, Benitec Limited issued warrants for the purchase of its ADSs. In connection with the Re-domiciliation, these warrants became obligations of the Company. Currently, there are warrants outstanding for the exercise of 22,516 shares of the Companys common stock at an exercise price of $21.43 per share. The exercise price is subject to appropriate adjustment in the event of certain share dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting our shares of common stock and also upon any distributions of assets, including cash, stock or other property to our stockholders. The warrants are exercisable, at the option of each holder, in whole or in part by delivering to us a duly executed exercise notice and, at any time a registration statement registering the issuance of the shares warrants under the Securities Act is effective and available for the issuance of such shares (or there is an effective registration statement for the resale of such shares), by payment in full in immediately available funds for the number of shares purchased upon such exercise. If such registration statement is currently effective at the time of an exercise, the warrants will be exercisable on a cashless basis. No fractional shares will be issued in connection with the exercise of a warrant. In lieu of fractional shares, we will round up to the next whole share or pay the holder an amount in cash equal to the fractional amount multiplied by the exercise price.
Subject to limited exceptions, holders of warrants do not have the right to exercise any portion of its warrants if the holder (together with such holders affiliates, and any persons acting as a group together with such holder or any of such holders affiliates) would beneficially own a number of ordinary shares in excess of 4.99% of the ordinary shares then outstanding after giving effect to such exercise (the Beneficial Ownership Limitation); provided, however, that, upon notice to the Company, the holder may increase or decrease the Beneficial Ownership Limitation, provided that in no event shall the Beneficial Ownership Limitation exceed 9.99% and any increase in the Beneficial Ownership Limitation will not be effective until 61 days following notice of such increase from the holder to us.
Except as otherwise provided in the warrants or by virtue of such holders ownership of shares of common stock, the holder of a warrant does not have the rights or privileges of a holder of our common stock, including any voting rights, until the holder exercises the warrant. The foregoing summary of the material terms and provisions the of the warrants is qualified in its entirety by the form of warrant, a copy of which is filed as an exhibit to the registration statement of which this prospectus is a part.
Anti-Takeover Effects of Provisions of the Certificate and Bylaws and DGCL
Some provisions of the DGCL, the Certificate and Bylaws could make the following transactions difficult: (i) acquisition of the Company by means of a tender offer; (ii) acquisition of the Company by means of a proxy
128
Table of Contents
contest or otherwise; or (iii) removal of incumbent officers and directors of the Company. It is possible that these provisions could make it more difficult to accomplish or could deter transactions that stockholders may otherwise consider to be in their best interest or in the best interests of the Company, including transactions that might result in a premium over the market price for the Companys common stock.
These provisions, summarized below, are expected to discourage coercive takeover practices and inadequate takeover bids. These provisions are also designed to encourage persons seeking to acquire control of the Company to first negotiate with the Board.
Delaware Anti-Takeover Statute. The Company is subject to Section 203 of the DGCL, which prohibits persons deemed interested stockholders from engaging in a business combination with a publicly-held Delaware corporation for three years following the date these persons become interested stockholders unless the business combination is, or the transaction in which the person became an interested stockholder was, approved in a prescribed manner or another prescribed exception applies. Generally, an interested stockholder is a person who, together with affiliates and associates, owns, or within three years prior to the determination of interested stockholder status did own, 15% or more of a corporations voting stock, and a business combination includes a merger, asset or stock sale, or other transaction resulting in a financial benefit to the interested stockholder. The existence of this provision may have an anti-takeover effect with respect to transactions not approved in advance by the Board, such as discouraging takeover attempts that might result in a premium over the market price of the Companys common stock.
Special Stockholder Meetings. The Bylaws provide that a special meeting of stockholders may be called by (i) the Chairman of the Board, if any, (ii) the President or Chief Executive Officer, or (iii) the Board pursuant to a resolution adopted by a majority of the total number of directors then in office.
Requirements for Advance Notification of Stockholder Nominations and Proposals. The Bylaws establish advance notice procedures with respect to stockholder proposals and the nomination of candidates for election as directors.
Composition of the Board of Directors; Election and Removal of Directors; Filling Vacancies
The Companys Board consists of five directors and the Board may, from time to time, fix the authorized number of directors by resolution of the Board. The Board is divided into three classes, designated Class I, Class II and Class III. As of the Implementation Date, there is one Class I director, two Class II directors and two Class III directors. Directors need not be stockholders of the Company.
Directors shall serve for a term ending on the date of the third annual meeting of stockholders following the annual meeting of stockholders at which such director was elected. However, each director initially assigned to Class I shall serve for a term expiring at the Companys first annual meeting of stockholders held after the time the initial classification of members of the Board becomes effective; each director initially assigned to Class II shall serve for a term expiring at the Companys second annual meeting of stockholders held following the time at which the initial classification of the members of the Board becomes effective; and each director initially assigned to Class III shall serve for a term expiring at the Corporations third annual meeting of stockholders held following the time at which the initial classification of the members of the Board becomes effective. The term of each director shall continue until the election and qualification of his or her successor and be subject to his or her earlier death, disqualification, resignation or removal. Except as otherwise provided by the DGCL, the Certificate or the Bylaws, directors shall be elected by a plurality of the votes of the shares present in person, by remote communication, if applicable, or represented by a duly authorized and executed proxy at the meeting and entitled to vote on the election of directors.
Subject to applicable law or by the Certificate, any director of the entire Board of the Company may be removed without cause by the affirmative vote of a majority of the holders of the Companys then-outstanding
129
Table of Contents
common stock entitled to vote generally at an election of directors. Furthermore, any vacancy on the Companys Board, however occurring, including a vacancy resulting from an increase in the size of the board, may be filled only by a majority vote of the Board then in office, even if less than a quorum, or by the sole remaining director.
Amendment of the Certificate and Bylaws. The Certificate may be amended in any manner permitted under the DGCL and the Bylaws may be amended by the vote or written consent of holders of a majority of the outstanding shares entitled to vote. The Board may also amend the Bylaws, other than a bylaw or amendment thereof specifying or changing a fixed number of directors or the maximum or minimum number or changing from a fixed to a variable board or vice versa.
Limitations of Liability and Indemnification Matters
Each of the Certificate and Bylaws provide that the Company is required to indemnify its directors and officers to the fullest extent not prohibited by Delaware law. The Bylaws also obligates the Company to advance expenses incurred by a director or officer in advance of the final disposition of any action or proceeding upon delivery to the Company of an undertaking, by or on behalf of such indemnitee, to repay all amounts so advanced if it shall ultimately be determined by final judicial decision, from which there is no further right to appeal, that such indemnitee is not entitled to be indemnified for such expenses.
To the fullest extent permitted by the DGCL, or any other applicable law, the Company, upon approval by the Board, may purchase insurance on behalf of any person required or permitted to be indemnified pursuant to the Bylaws.
Forum for Adjudication of Disputes
The Certificate provides that, unless the Company consents in writing to the selection of an alternative forum, the sole and exclusive forum for (i) derivative actions or proceedings brought on behalf of the Company, (ii) any action asserting a claim of breach of fiduciary duty owed by any director, officer or employee of the Company to the Company or the Companys stockholders, (iii) an action asserting a claim arising pursuant to any provision of the DGCL, or (iv) any action asserting a claim governed by the internal affairs doctrine shall be a state or federal court located within the state of Delaware. The Certificate further provides that the federal district courts of the United States of America shall, to the fullest extent permitted by law, be the exclusive forum for any complaint asserting a cause of action arising under the Securities Act.
Transfer Agent and Registrar
The transfer agent and registrar for the Companys common stock is Computershare Trust Company, N.A. The transfer agent and registrars address is 250 Royall St., Canton, Massachusetts 02021. Listing. Our common stock is listed on Nasdaq under the symbol BNTC.
130
Table of Contents
MATERIAL UNITED STATES FEDERAL INCOME TAX CONSIDERATIONS FOR HOLDERS OF OUR COMMON STOCK AND PRE-FUNDED WARRANTS
The following discussion is a summary of the material U.S. federal income tax considerations to U.S. Holders and Non-U.S. Holders (each as defined below and, together Holders) of the purchase, ownership and disposition of our common stock and pre-funded warrants that are issued pursuant to this offering. This does not purport to be a complete analysis of all potential tax effects to Holders of purchasing, owning or disposing our common stock or pre-funded warrants. The effects of other U.S. federal tax laws, such as estate and gift tax laws, and any applicable state, local or foreign tax laws are not included in this discussion, and Holders should consult their own tax advisors as to these matters. This discussion is based on the Internal Revenue Code of 1986, as amended (the Code), final, temporary and proposed Treasury Regulations promulgated thereunder, judicial decisions and administrative pronouncements of the IRS, in effect as of the date of this offering. These authorities may change or be subject to differing interpretations. Any such change may be applied retroactively in a manner that could adversely affect a Holder. We have not sought and will not seek any rulings from the IRS regarding the matters discussed below. There can be no assurance that the IRS will not take or that a court will not uphold a contrary position regarding the tax consequences of the purchase, ownership and disposition of our common stock and pre-funded warrants.
This discussion is limited to Holders that hold our common stock and pre-funded warrants as capital assets within the meaning of Section 1221 of the Code (generally, property held for investment). This discussion does not address all U.S. federal income tax consequences relevant to a Holders particular circumstances. In addition, it does not address consequences relevant to Holders subject to special rules, including, without limitation:
| | banks, insurance companies and other financial institutions; |
| | real estate investment trusts, regulated investment companies, and other entities treated as conduits for U.S. federal income tax purposes; |
| | brokers, dealers or traders in securities; |
| | Holders who are subject to the mark-to-market accounting rules under Section 475 of the Code; |
| | controlled foreign corporations, passive foreign investment companies and corporations that accumulate earnings to avoid U.S. federal income tax; |
| | partnerships or other entities or arrangements treated as partnerships for U.S. federal income tax purposes; |
| | tax-exempt organizations and governmental organizations or agencies; |
| | Holders who hold or receive our common stock or pre-funded warrants pursuant to the exercise of any employee stock option or otherwise as compensation; |
| | tax-qualified retirement plans; |
| | qualified foreign pension funds as defined in Section 897(l)(2) of the Code and entities all of the interests of which are held by qualified foreign pension funds; |
| | U.S. expatriates and certain former citizens or long-term residents of the United States; |
| | U.S. Holders whose functional currency is not the United States dollar; |
| | Holders deemed to sell the our common stock or pre-funded warrants under the constructive sale provisions of the Code; |
| | Holders for whom our common stock and pre-funded warrants constitute qualified small business stock within the meaning of Section 1202 of the Code; |
131
Table of Contents
| | Holders subject to special tax accounting rules as a result of any item of gross income with respect to the stock being taken into account in an applicable financial statement (as defined in the Code); |
| | Holders holding our common stock or pre-funded warrants as part of a hedge, straddle or other risk reduction strategy or as part of a conversion transaction or other integrated investment; and |
| | Holders subject to the alternative minimum tax. |
If a partnership (or other entity or arrangement treated as a partnership for U.S. federal income tax purposes) holds our common stock or pre-funded warrants, the tax treatment of a partner in the partnership will depend on the status of the partner, the activities of the partnership and certain determinations made at the partner level. Accordingly, partnerships holding our common stock or pre-funded warrants and the partners in such partnerships should consult their own tax advisors regarding the U.S. federal income tax consequences to them.
THIS DISCUSSION IS FOR INFORMATION PURPOSES ONLY AND IS NOT INTENDED AS TAX ADVICE. INVESTORS SHOULD CONSULT THEIR OWN TAX ADVISORS WITH RESPECT TO THE APPLICATION OF THE U.S. FEDERAL INCOME TAX LAWS TO THEIR PARTICULAR SITUATIONS AS WELL AS ANY TAX CONSEQUENCES OF THE PURCHASE, OWNERSHIP AND DISPOSITION OF OUR COMMON STOCK OR PRE-FUNDED WARRANTS ARISING UNDER THE U.S. FEDERAL ESTATE OR GIFT TAX LAWS OR UNDER THE LAWS OF ANY STATE, LOCAL OR NON-U.S. TAXING JURISDICTION OR UNDER ANY APPLICABLE INCOME TAX TREATY.
Characterization of the Pre-funded Warrants
Although the characterization of the pre-funded warrants for U.S. federal income tax purposes is not entirely clear, because the exercise price of the pre-funded warrants is a nominal amount, the Company expects to treat the pre-funded warrants as common stock of the Company for U.S. federal income tax purposes. Except where noted, the remainder of this discussion assumes that the pre-funded warrants will be so treated. Each Holder should consult its own tax advisor regarding the proper characterization of the pre-funded warrants for U.S. federal, state and local, and non-U.S. tax purposes, and the consequences to them of such treatment given their individual circumstances. Some portions of the below discussion make reference to potential consequences associated with the purchase, ownership and disposition of the pre-funded warrants independent of their potential characterization as common shares.
U.S. Holders
For purposes of this discussion, a U.S. Holder is any beneficial owner of Securities that is, for U.S. federal income tax purposes:
| | an individual who is a citizen or resident of the United States; |
| | a corporation (or other entity treated as a corporation for U.S. federal income tax purposes) created or organized under the laws of the United States, any state thereof, or the District of Columbia; |
| | an estate, the income of which is subject to U.S. federal income tax regardless of its source; or |
| | a trust that (i) is subject to the primary supervision of a U.S. court and the control of one or more U.S. persons (within the meaning of Section 7701(a)(30) of the Code), or (ii) has made a valid election under applicable Treasury Regulations to continue to be treated as a U.S. person. |
If you are not a U.S. Holder, this section does not apply to you. Please see the discussion under Non-U.S. Holders below.
Distributions on Common Stock
As described in the section captioned Dividend Policy, we do not anticipate declaring or paying distributions to Holders of our common stock in the foreseeable future. However, distributions, if any, on our
132
Table of Contents
common stock generally will constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles.
Distributions in excess of current and accumulated earnings and profits will constitute a return of capital that will be applied against and reduce (but not below zero) the U.S. Holders adjusted tax basis in our common stock. Any remaining excess will be treated as gain realized on the sale or other disposition of the common stock and will be treated as described under U.S. HoldersSale or Other Taxable Disposition of Common Stock, Warrants or Pre-funded Warrants below.
Dividends we pay to a U.S. Holder that is a taxable corporation generally will qualify for the dividends received deduction if the requisite holding period is satisfied. With certain exceptions (including, but not limited to, dividends treated as investment income for purposes of investment interest deduction limitations), and provided the common shares are held for more than 60 days during the 121-day period beginning 60 days before the ex- dividend date and certain other holding period requirements are met, dividends we pay to a non-corporate U.S. Holder generally will constitute qualified dividends that will be subject to tax at the maximum tax rate accorded to long-term capital gains. Dividends paid by us will generally be treated as income from U.S. sources. U.S. Holders should consult their own tax advisors regarding the holding period and other requirements that must be satisfied in order to qualify for the reduced maximum tax rate on dividends.
Sale or Other Taxable Disposition of Common Stock or Pre-funded Warrants
Upon a sale, exchange or other taxable disposition of common stock or pre-funded warrants, a U.S. Holder generally will recognize capital gain or loss in an amount equal to the difference between the amount realized and the U.S. Holders adjusted tax basis in the common stock or pre-funded warrant, as the case may be. A U.S. Holders adjusted tax basis in the common stock generally will equal the U.S. Holders acquisition cost of such security less, in the case of common stock (and potentially a pre-funded warrant) and as described further above, the amount of any prior distributions treated as a return of capital on such stock. If a U.S. Holder purchases or sells common stock and/or pre-funded warrants together in a single transaction in which the purchase price for each of the common stock and/or pre-funded warrants was not separately stated, the U.S. Holder generally would be required to allocate the purchase price among the subject securities so acquired or disposed of, as applicable, based on the relative fair market values of each (at the time of the acquisition or disposition, as applicable). U.S. Holders who purchase or sell common stock and/or pre-funded warrants in a single transaction should consult with their tax advisors regarding such allocation.
Any such capital gain or loss generally will be long-term capital gain or loss if the U.S. Holders holding period for the common stock or pre-funded warrants disposed of exceeds one year. Long-term capital gains recognized by non-corporate U.S. Holders will be eligible to be taxed at reduced rates. The deductibility of capital losses is subject to limitations.
Exercise of a Pre-funded Warrant
A U.S. Holder generally will not recognize taxable gain or loss on the acquisition of common stock upon exercise of a pre-funded warrant. The U.S. Holders aggregate tax basis in the share of our common stock received upon exercise of a pre-funded warrant generally will be an amount equal to the sum of the U.S. Holders tax basis in the pre-funded warrant prior to exercise and the warrants exercise price. Provided that a pre-funded warrant is treated as our common stock, a U.S. Holders holding period for the common stock received upon exercise of a warrant will include the holding period for the pre-funded warrant. On the other hand, if the pre-funded warrant is treated as an option to purchase our stock, a U.S. Holders holding period for the common stock received upon exercise of a warrant will begin on the date following the date of exercise of the warrant and will not include the period during which the U.S. Holder held the warrant.
133
Table of Contents
A U.S. Holder may be permitted to undertake a cashless exercise of pre-funded warrants into our common stock. The U.S. federal income tax treatment of a cashless exercise is unclear, and the tax consequences of a cashless exercise could differ from the consequences of an exercise described above. For example, the cashless exercise could be treated as a taxable disposition of a portion of the Warrants or the common shares into which they are exercisable. U.S. Holders should consult their own tax advisors regarding the U.S. federal income tax consequences of a cashless exercise.
Certain Adjustments to the Pre-funded Warrants and Payments in Respect of the Pre-funded Warrants
Under Section 305 of the Code, an adjustment to the number of shares of common stock that will be issued on the exercise of the pre-funded warrants, or an adjustment to the exercise price of the pre-funded warrants, may be treated as a constructive distribution to a U.S. Holder of the pre-funded warrants if, and to the extent that, such adjustment has the effect of increasing such U.S. Holders proportionate interest in our earnings and profits or assets, depending on the circumstances of such adjustment (for example, if such adjustment is to compensate for a distribution of cash or other property to our shareholders). Adjustments to the exercise price of pre-funded warrants made pursuant to a bona fide reasonable adjustment formula that has the effect of preventing dilution of the interest of the U.S. Holder of the pre-funded warrants generally should not be considered to result in a constructive distribution. Any constructive distribution would be treated as a dividend, return of capital or capital gain as described under the heading U.S. HolderDistributions on Common Stock above, and could be taxable whether or not there is an actual distribution of cash or other property. U.S. Holders should consult their tax advisors regarding the proper treatment of any adjustments to and payments in respect of pre-funded warrants.
Net investment income tax
An additional 3.8% tax is imposed on the net investment income of non-corporate U.S. Holders, and on the undistributed net investment income of certain estates and trusts. Among other items, net investment income generally includes dividends paid on our common stock and certain net gain from the sale or other taxable disposition of our common stock and pre-funded warrants, less certain deductions. U.S. Holders should consult their own tax advisors concerning the potential effect, if any, of this tax on holding our common stock and pre-funded warrants in such U.S. Holders particular circumstances.
Backup withholding and information reporting
For non-corporate U.S. Holders, information reporting requirements, on IRS Form 1099, generally will apply to:
| | dividend payments or other taxable distributions on our common stock and pre-funded warrants made to the non-corporate U.S. Holder within the United States or by a United States payor; and |
| | the payment of proceeds to the non-corporate U.S. Holder from the sale of a share of common stock or pre-funded warrants effected at a United States office of a broker or through certain U.S.-related financial intermediaries. |
Additionally, backup withholding may apply to such payments if the non-corporate U.S. Holder:
| | fails to provide an accurate taxpayer identification number; |
| | is notified by the IRS that it has failed to report all interest and dividends required to be shown on its U.S. federal income tax returns; or |
| | in certain circumstances, fails to comply with applicable certification requirements. |
134
Table of Contents
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be allowed as a refund or a credit against a non-corporate U.S. Holders U.S. federal income tax liability (if any), provided the required information is timely furnished to the IRS. U.S. Holders are urged to consult their own tax advisors regarding the application of backup withholding and the availability of and procedure for obtaining an exemption from backup withholding in their particular circumstances.
Non-U.S. Holders
For purposes of this discussion, a Non-U.S. Holder is a beneficial owner of our common stock or pre-funded warrants that, for U.S. federal income tax purposes, is neither a U.S. Holder (as defined above) nor a partnership or other pass-through entity. If you are not a Non-U.S. Holder, this section does not apply to you.
Distributions on Common Stock
As described in the section captioned Dividend Policy, we do not anticipate declaring or paying distributions to Holders of our common stock in the foreseeable future. However, distributions, if any, on our common stock in cash will generally constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. Amounts not treated as dividends for U.S. federal income tax purposes will constitute a return of capital and first be applied against and reduce a Non-U.S. Holders adjusted tax basis in its common stock, but not below zero. Any excess will be treated as capital gain and will be treated as described below under Non-U.S. HoldersSale or Other Taxable Disposition of Common Stock or Pre-Funded Warrants.
Subject to the discussion below regarding backup withholding and payments made to certain foreign accounts, dividends paid to a Non-U.S. Holder of our common stock (including constructive distributions treated as a dividend) that are not effectively connected with the Non-U.S. Holders conduct of a trade or business within the United States will be subject to U.S. federal withholding tax at a rate of 30% of the gross amount of the dividends (or such lower rate as may be specified by an applicable income tax treaty).
Non-U.S. Holders may be entitled to a reduction in or an exemption from withholding on dividends as a result of either (i) qualifying for the benefits of an applicable income tax treaty or (ii) the Non-U.S. Holder holding our common stock in connection with the conduct of a trade or business within the United States and dividends being paid in connection with that trade or business. To claim such a reduction in or exemption from withholding, the Non-U.S. Holder must provide the applicable withholding agent with a properly executed (i) IRS Form W-8BEN or W-8BEN-E (or applicable successor form) claiming an exemption from or reduction of the withholding tax under the benefit of an applicable income tax treaty, (ii) IRS Form W-8ECI (or applicable successor form) stating that the dividends are effectively connected with the conduct by the Non-U.S. Holder of a trade or business within the United States, or (iii) a suitable substitute form, as may be applicable. These certifications must be provided to the applicable withholding agent prior to the payment of dividends and must be updated periodically. Non-U.S. Holders that do not timely provide the applicable withholding agent with the required certification, but that qualify for a reduced rate under an applicable income tax treaty, may obtain a refund of any excess amounts withheld by timely filing an appropriate claim for refund with the IRS.
Subject to the discussion below regarding backup withholding and payments made to certain foreign accounts, if dividends paid to a Non-U.S. Holder are effectively connected with the Non-U.S. Holders conduct of a trade or business within the United States (or, if required by an applicable income tax treaty, the Non-U.S. Holder maintains a permanent establishment in the United States to which such dividends are attributable), then, although generally exempt from U.S. federal withholding tax (provided the Non-U.S. Holder provides appropriate certification, as described above), the Non-U.S. Holder will be subject to U.S. federal income tax on such dividends on a net income basis at the regular graduated U.S. federal income tax rates. In addition, a Non-U.S. Holder that is or is treated as a corporation for U.S. federal income tax purposes may be subject to an additional branch profits tax at a rate of 30% (or such lower rate specified by an applicable income tax treaty) on
135
Table of Contents
its effectively connected earnings and profits for the taxable year that are attributable to such dividends, as adjusted for certain items. Non-U.S. Holders should consult their own tax advisors regarding their entitlement to benefits under any applicable income tax treaty.
Sale or Other Taxable Disposition of Common Stock or Pre-funded Warrants
Subject to the discussion below regarding backup withholding and payments made to certain foreign accounts, a Non-U.S. Holder generally will not be subject to U.S. federal income tax on any gain recognized upon the sale or other taxable disposition of a share of our common stock or pre-funded warrants unless:
| | the gain is effectively connected with the Non-U.S. Holders conduct of a trade or business within the United States (or, if required by an applicable income tax treaty, the Non-U.S. Holder maintains a permanent establishment in the United States to which such gain is attributable); |
| | the Non-U.S. Holder is a non-resident alien individual present in the United States for 183 days or more during the taxable year of the disposition and certain other requirements are met; or |
| | we are or have been a U.S. real property holding corporation, or USRPHC, for U.S. federal income tax purposes at any time during the shorter of the five-year period ending on the date of disposition or the period that the Non-U.S. Holder held the common stock or pre-funded warrants. |
Gain described in the first bullet point above will generally be subject to U.S. federal income tax on a net income basis at the regular graduated U.S. federal income tax rates. A Non-U.S. Holder that is a foreign corporation also may be subject to an additional branch profits tax at a rate of 30% (or such lower rate specified by an applicable income tax treaty) on a portion of its effectively connected earnings and profits for the taxable year, as adjusted for certain items.
A Non-U.S. Holder described in the second bullet point above will be subject to U.S. federal income tax at a rate of 30% (or such lower rate as may be specified by an applicable income tax treaty) on any gain derived from the sale or other taxable disposition, which may be offset by certain U.S. source capital losses of the Non-U.S. Holder (even though the individual is not considered a resident of the United States) provided the Non-U.S. Holder timely files U.S. federal income tax returns with respect to such losses.
With respect to the third bullet point above, we believe that we are not, and do not anticipate that we will become, a USRPHC.
The method of determining the amount of gain by a Non-U.S. Holder on disposition of the common stock or pre-funded warrants generally will correspond to the method of determining the amount of gain (or loss) by a U.S. Holder on disposition of the common stock or pre-funded warrants, as described under U.S. HoldersSale or Other Taxable Disposition of Common Stock or Pre-funded Warrants above. Non-U.S. Holders should consult their own tax advisors regarding potentially applicable income tax treaties that may provide for different rules, and the potential application of other exceptions to these taxes.
Exercise of a Pre-funded Warrant
For certain Non-U.S. Holders engaged in the conduct of a trade or business in the United States, the U.S. federal income tax treatment of the exercise of a pre-funded warrant, generally will correspond to the U.S. federal income tax treatment of the exercise of a pre-funded warrant by a U.S. Holder, as described under U.S. Holders Exercise of a Pre-funded Warrant above. For all other Non-U.S. holders, the exercise of a pre-funded warrant generally will not be a U.S. taxable event.
136
Table of Contents
Certain Adjustments to the Pre-funded Warrants and Payments in Respect of the Pre-funded Warrants
Under Section 305 of the Code, an adjustment to the number of shares of common stock that will be issued on the exercise of the pre-funded warrants, or an adjustment to the exercise price of the pre-funded warrants, may be treated as a constructive distribution to a Non-U.S. Holder of the pre-funded warrants if, and to the extent that, such adjustment has the effect of increasing such Non-U.S. Holders proportionate interest in our earnings and profits or assets, depending on the circumstances of such adjustment (for example, if such adjustment is to compensate for a distribution of cash or other property to our shareholders). Adjustments to the exercise price of pre-funded warrants made pursuant to a bona fide reasonable adjustment formula that has the effect of preventing dilution of the interest of the Non-U.S. Holder of the pre-funded warrants generally should not be considered to result in a constructive distribution. Any constructive distribution would be treated as a dividend, return of capital or capital gain as described under the heading Non-U.S. HoldersDistributions on Common Stock above, and could be taxable whether or not there is an actual distribution of cash or other property.
In addition, regulations governing dividend equivalents under Section 871(m) of the Code may apply to the pre-funded warrants. Under those regulations, an implicit or explicit payment under the pre-funded warrants that references a dividend distribution on our common stock (including an adjustment to the amount due on the pre-funded warrant to take into account a dividend distribution on our common stock) would be taxable to a Non-U.S. Holder as described under the heading Non-U.S. HoldersDistributions on Common Stock above. Such dividend equivalent amount would be taxable and subject to withholding whether or not there is actual payment of cash or other property, and the Company may satisfy any withholding obligations it has in respect of the pre-funded warrants by withholding from other amounts due to the Holder. Non-U.S. Holders are encouraged to consult their own tax advisors regarding the application of Section 871(m) of the Code to the pre-funded warrants.
Information Reporting and Backup Withholding
Subject to the discussion below regarding payments made to certain foreign accounts, a Non-U.S. Holder generally will not be subject to backup withholding with respect to payments of dividends on our common stock we make to the Non-U.S. Holder, provided the applicable withholding agent does not have actual knowledge or reason to know such Holder is a U.S. person and the Holder certifies its non-U.S. status by providing a valid IRS Form W-8BEN, W-8BEN-E or W-8ECI, or other applicable certification (or applicable successor form), or otherwise establishes an exception. However, information returns will be filed with the IRS in connection with any dividends or other distributions on our common stock paid to the Non-U.S. Holder (including constructive distributions), regardless of whether any tax was actually withheld. Copies of these information returns may also be made available under the provisions of a specific treaty or agreement to the tax authorities of the country in which the Non-U.S. Holder resides or is established.
Information reporting and backup withholding may apply to the proceeds of a sale of a share of our common stock or pre-funded warrants within the United States, and information reporting may (although backup withholding will generally not) apply to the proceeds of the sale of a share of our common stock or pre-funded warrants outside the United States conducted through certain U.S.-related financial intermediaries, in each case, unless the beneficial owner certifies under penalty of perjury that it is a Non-U.S. person on IRS Form W-8BEN or other applicable form or successor form (and the payor does not have actual knowledge or reason to know that the beneficial owner is a U.S. person) or otherwise establishes an exemption.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be allowed as a refund or a credit against a Non-U.S. Holders U.S. federal income tax liability (if any), provided the required information is timely furnished to the IRS. Non-U.S. Holders are urged to consult their own tax advisors regarding the application of backup withholding and the availability of and procedure for obtaining an exemption from backup withholding in their particular circumstances.
137
Table of Contents
Additional Withholding Tax on Payments Made to Foreign Accounts
Withholding taxes may be imposed under the provisions of the law generally known as the Foreign Account Tax Compliance Act, or FATCA, on certain types of payments made to non-U.S. financial institutions and certain other non-U.S. entities. Specifically, a 30% withholding tax may be imposed on dividends (including constructive dividends) paid on our common stock or pre-funded warrants, or (subject to the proposed Treasury Regulations discussed below) gross proceeds from the sale or other disposition of our common stock or pre-funded warrants paid to a foreign financial institution or a non-financial foreign entity (each as defined in the Code), unless (i) the foreign financial institution undertakes certain diligence and reporting obligations, (ii) the non-financial foreign entity either certifies it does not have any substantial U.S. owners (as defined in the Code) or furnishes identifying information regarding each substantial U.S. owner, or (iii) the foreign financial institution or non-financial foreign entity otherwise qualifies for an exemption from these rules. If the payee is a foreign financial institution and is subject to the diligence and reporting requirements in (i) above, it must enter into an agreement with the U.S. Department of the Treasury requiring, among other things, that it undertake to identify accounts held by certain specified U.S. persons or U.S.-owned foreign entities (each as defined in the Code), annually report certain information about such accounts and withhold 30% on payments to non-compliant foreign financial institutions and certain other account holders. An intergovernmental agreement between the United States and an applicable foreign country, or future Treasury Regulations or other guidance, may modify these requirements.
Under the applicable Treasury Regulations and guidance from the IRS, withholding under FATCA generally applies to payments of dividends (including constructive dividends) on our common stock or pre-funded warrants. The FATCA withholding tax will apply to all withholdable payments without regard to whether the beneficial owner of the payment would otherwise be entitled to an exemption from imposition of withholding tax pursuant to an applicable tax treaty with the United States or U.S. domestic law. We will not pay additional amounts to Holders of our common stock or pre-funded warrants in respect of any amounts withheld. While withholding under FATCA would have applied also to payments of gross proceeds from the sale or other disposition of our common stock or pre-funded warrants on or after January 1, 2019, proposed Treasury Regulations eliminate FATCA withholding on payments of gross proceeds entirely. Taxpayers generally may rely on these proposed Treasury Regulations until final Treasury Regulations are issued.
Prospective investors should consult their own tax advisors regarding the potential application of withholding under FATCA to their investment in our common stock and pre-funded warrants.
138
Table of Contents
We have entered into an underwriting agreement with H.C. Wainwright & Co., LLC, as underwriter, with respect to the securities being offered hereby. Subject to the terms and conditions of the underwriting agreement, we have agreed to sell to the underwriter and the underwriter has agreed to purchase from us, at the public offering price less the underwriting discounts and commissions set forth on the cover page of this prospectus, shares of our common stock and pre-funded warrants.
Pursuant to the terms and subject to the conditions contained in the underwriting agreement, we have agreed to sell to the underwriter named below, and the underwriter has agreed to purchase from us, the respective number of shares of common stock and pre-funded warrants set forth opposite its name below:
| Underwriter |
Number of Shares of Common Stock |
Number of Pre- funded Warrants |
||||||
| H.C. Wainwright & Co., LLC |
||||||||
The underwriting agreement provides that the obligation of the underwriter to purchase the shares of common stock and/or pre-funded warrants offered by this prospectus is subject to certain conditions. The underwriter is obligated to purchase all of the shares of common stock and/or pre-funded warrants if any of the securities are purchased, other than those shares covered by the option to purchase additional securities described below. Delivery of the shares of common stock and any pre-funded warrants to purchasers is expected on or about , 2020, subject to certain customary closing conditions.
Option to Purchase Additional Securities
We have granted to the underwriter an option, exercisable not later than 30 days after the date of this prospectus, to purchase up to an additional shares of common stock at the public offering price, less the underwriting discounts and commissions, set forth on the cover page of this prospectus. If any additional shares of common stock are purchased pursuant to such option, the underwriter will offer these securities on the same terms as those on which the securities are being offered hereby.
Discounts, Commissions and Expenses
The underwriter proposes to offer the shares of common stock and/or pre-funded warrants pursuant to the underwriting agreement to the public at the public offering price set forth on the cover page of this prospectus and to certain dealers at that price less a concession not in excess of $ per share or per pre-funded warrant. After this offering, the public offering price and concession may be changed by the underwriter. No such change shall change the amount of proceeds to be received by us as set forth on the cover page of this prospectus.
In connection with the sale of the common stock and/or pre-funded warrants to be purchased by the underwriter, the underwriter will be deemed to have received compensation in the form of underwriting commissions and discounts. The underwriters commissions and discounts will be 8% of the gross proceeds of this offering, or $ per share of common stock or per pre-funded warrant.
The following table shows underwriting discounts and commissions payable to the underwriter by us in connection with this offering:
| Per Share | Per Pre-Funded Warrant |
Total without Option |
Total with Option |
|||||||||||||
| Public offering price |
$ | $ | $ | |||||||||||||
| Underwriting discounts and commissions |
$ | $ | $ | |||||||||||||
139
Table of Contents
We have also agreed to pay H.C. Wainwright & Co., LLC an expense allowance consisting of (a) a management fee equal to 1.0% of the gross proceeds raised in the offering, (b) $50,000 for non-accountable expenses, (c) up to $100,000 for fees and expenses of legal counsel and other out-of-pocket expenses and (d)$12,900 clearing fees. We estimate the total expenses payable by us for this offering will be approximately $ , which amount excludes underwriting discounts and commissions.
Right of First Refusal
We have also agreed, subject to certain conditions, to give the underwriter a twelve-month right of first refusal from the closing of the offering, to act (i) as our exclusive financial advisor if we decide to dispose of or acquire business units or acquire any of our outstanding securities out of the ordinary course or make any exchange or tender offer or enter into a merger, consolidation or other business combination or any recapitalization, reorganization, restructuring or other similar transaction and we decide to retain a financial advisor for such transaction; (ii) as sole book-runner, sole manager, sole placement agent or sole agent with respect to any financing or refinancing of any indebtedness using a manager or agent; or (iii) as sole book-running manager, sole underwriter or sole placement agent if we to raise funds by means of a public offering (including at-the-market facility) or a private placement or any other capital-raising financing of equity, equity-linked or debt securities using an underwriter or placement agent, in each case during such period.
Indemnification
Pursuant to the underwriting agreement, we have agreed to indemnify the underwriter against certain liabilities, including liabilities under the Securities Act, or to contribute to payments that the underwriter or such other indemnified parties may be required to make in respect of those liabilities.
Determination of Offering Price
The actual offering price of the securities we are offering will be negotiated between us and the underwriter based on the trading of our shares of common stock prior to the offering, among other things.
Lock-Up Agreements
We, and each of our directors and executive officers, have entered into lock-up agreements that prevent us or them from selling any shares of common stock or any securities convertible into or exercisable or exchangeable into shares of common stock, subject to certain exceptions, for a period of 90 days after the date of this prospectus supplement. The underwriter, in its sole discretion, may release shares of common stock and other securities subject to the lock-up agreements described above in whole or in part at any time. When determining whether or not to release the shares of common stock and other securities from lock-up agreements, the underwriter will consider, among other factors, the holders reasons for requesting the release and the number of shares of common stock or other securities for which the release is being requested.
We have also agreed, in the underwriting agreement, to a restriction on the issuance of any variable priced securities for 12 months following the closing of this offering, except that, after the 90th day following the date of the underwriting agreement we may enter into an at-the-market offering with the underwriter.
Price Stabilization, Short Positions and Penalty Bids
The underwriter may engage in syndicate covering transactions, stabilizing transactions and penalty bids or purchases for the purpose of pegging, fixing or maintaining the price of our shares of common stock:
| | Syndicate covering transactions involve purchases of securities in the open market after the distribution has been completed in order to cover syndicate short positions. Such a naked short position would be closed out by buying securities in the open market. A naked short position is more likely to be created if the |
140
Table of Contents
| underwriter is concerned that there could be downward pressure on the price of the securities in the open market after pricing that could adversely affect investors who purchase in the offering. |
| | Stabilizing transactions permit bids to purchase the underlying security so long as the stabilizing bids do not exceed a specific maximum. |
| | Penalty bids permit the underwriter to reclaim a selling concession from a syndicate member when the securities originally sold by the syndicate member are purchased in a stabilizing or syndicate covering transaction to cover syndicate short positions. |
These syndicate covering transactions, stabilizing transactions and penalty bids may have the effect of raising or maintaining the market prices of our securities or preventing or retarding a decline in the market prices of our securities. As a result, the price of our shares of common stock may be higher than the price that might otherwise exist in the open market. Neither we nor the underwriter make any representation or prediction as to the effect that the transactions described above may have on the price of our shares of common stock. These transactions may be effected on the Nasdaq Global Market, in the over-the-counter market or on any other trading market and, if commenced, may be discontinued at any time.
In connection with this offering, the underwriter also may engage in passive market making transactions in our shares of common stock in accordance with Regulation M during a period before the commencement of offers or sales of our shares of common stock in this offering and extending through the completion of the distribution. In general, a passive market maker must display its bid at a price not in excess of the highest independent bid for that security. However, if all independent bids are lowered below the passive market makers bid that bid must then be lowered when specific purchase limits are exceeded. Passive market making may stabilize the market price of the securities at a level above that which might otherwise prevail in the open market and, if commenced, may be discontinued at any time.
Neither we nor the underwriter make any representation or prediction as to the direction or magnitude of any effect that the transactions described above may have on the prices of our securities. In addition, neither we nor the underwriter make any representation that the underwriter will engage in these transactions or that any transactions, once commenced, will not be discontinued without notice.
Electronic Offer, Sale and Distribution of Securities
A prospectus in electronic format may be made available on the websites maintained by the underwriter, if any, participating in this offering and the underwriter may distribute prospectuses electronically. Other than the prospectus in electronic format, the information on these websites is not part of this prospectus supplement or the registration statement of which this prospectus supplement forms a part, has not been approved or endorsed by us or the underwriter, and should not be relied upon by investors.
Other Relationships
From time to time, the underwriter and its affiliates have provided, and may provide in the future, various advisory, investment and commercial banking and other services to us in the ordinary course of business, for which they have received and may continue to receive customary fees and commissions.
Selling Restrictions
European Economic Area
In relation to each member state of the European Economic Area (the EEA) that has implemented the Prospectus Regulation (as defined below) (each, a Relevant Member State), with effect from and including the date on which the Prospectus Regulation is implemented in that Relevant Member State, an offer of securities described in this prospectus may not be made to the public in that Relevant Member State other than:
| | to any legal entity which is a qualified investor as defined in the Prospectus Regulation; |
141
Table of Contents
| | by the placement agents to fewer than 150 natural or legal persons (other than qualified investors as defined in the Prospectus Regulation), as permitted under the Prospectus Regulation, subject to obtaining the prior consent of H.C. Wainwright & Co., LLC for any such offer; or |
| | in any other circumstances falling within Article 1(4) of the Prospectus Regulation; |
| | provided that no such offer of securities shall require us or any placement agent to publish a prospectus pursuant to Article 3 of the Prospectus Regulation or supplement a prospectus pursuant to Article 23 of the Prospectus Regulation. |
For purposes of this provision, the expression an offer of securities to the public in any Relevant Member State means the communication in any form and by any means of sufficient information on the terms of the offer and the securities to be offered so as to enable an investor to decide to purchase or subscribe for the securities, and the expression Prospectus Regulation means Regulation (EU) 2017/1129.
We have not authorized and do not authorize the making of any offer of securities through any financial intermediary on its behalf, other than offers made by the placement agents with a view to the final placement of the securities as contemplated in this prospectus. Accordingly, no purchaser of the securities is authorized to make any further offer of the securities on behalf of us or the placement agents.
The EEA selling restriction is in addition to any other selling restrictions set out in this prospectus.
United Kingdom
This prospectus is only being distributed to, and is only directed at, persons in the United Kingdom that are qualified investors within the meaning of Article 2(1)(e) of the Prospectus Directive that are also (i) investment professionals falling within Article 19(5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005 (the Order) or (ii) high net worth entities, and other persons to whom it may lawfully be communicated, falling within Article 49(2)(a) to (d) of the Order (each such person being referred to as a relevant person). This prospectus and its contents are confidential and should not be distributed, published or reproduced (in whole or in part) or disclosed by recipients to any other person in the United Kingdom. Any person in the United Kingdom that is not a relevant person should not act or rely on this document or any of its contents.
Canada
Our securities may be sold only to purchasers purchasing, or deemed to be purchasing, as principal that are accredited investors, as defined in National Instrument 45-106 Prospectus Exemptions or subsection 73.3(1) of the Securities Act (Ontario), and are permitted clients, as defined in National Instrument 31-103 Registration Requirements, Exemptions and Ongoing Registrant Obligations. Any resale of our securities must be made in accordance with an exemption from, or in a transaction not subject to, the prospectus requirements of applicable securities laws.
Securities legislation in certain provinces or territories of Canada may provide a purchaser with remedies for rescission or damages if the prospectus (including any amendment thereto) contains a misrepresentation, provided that the remedies for rescission or damages are exercised by the purchaser within the time limit prescribed by the securities legislation of the purchasers province or territory. The purchaser should refer to any applicable provisions of the securities legislation of the purchasers province or territory for particulars of these rights or consult with a legal advisor.
Pursuant to section 3A.3 (or, in the case of securities issued or guaranteed by the government of a non-Canadian jurisdiction, section 3A.4) of National Instrument 33-105 Underwriting Conflicts (NI 33-105), the underwriter is not required to comply with the disclosure requirements of NI 33-105 regarding underwriter conflicts of interest in connection with this offering.
142
Table of Contents
Israel
This document does not constitute a prospectus under the Israeli Securities Law, 5728-1968, or the Securities Law, and has not been filed with or approved by the Israel Securities Authority. In Israel, this prospectus is being distributed only to, and is directed only at, and any offer of the ordinary shares is directed only at (i) a limited number of persons in accordance with the Israeli Securities Law and (ii) investors listed in the first addendum (the Addendum) to the Israeli Securities Law, consisting primarily of joint investment in trust funds, provident funds, insurance companies, banks, portfolio managers, investment advisors, members of the Tel Aviv Stock Exchange, underwriters, venture capital funds, entities with equity in excess of NIS 50 million and qualified individuals, each as defined in the Addendum (as it may be amended from time to time), collectively referred to as qualified investors (in each case, purchasing for their own account or, where permitted under the Addendum, for the accounts of their clients who are investors listed in the Addendum). Qualified investors are required to submit written confirmation that they fall within the scope of the Addendum, are aware of the meaning of same and agree to it.
143
Table of Contents
The validity of the securities offered in this prospectus will be passed upon for us by Proskauer Rose LLP, Los Angeles, California. Lowenstein Sandler LLP, New York, New York, is acting as counsel for the underwriter in connection with this offering.
The audited financial statements included in this prospectus and elsewhere in the registration statement have been so included in reliance upon the report of Grant Thornton Audit Pty Ltd, independent registered public accountants, upon the authority of said firm as experts in accounting and auditing.
CHANGE IN AUDITOR
As previously disclosed, on June 5, 2020, the Audit Committee approved the engagement of Squar Milner LLP (Squar Milner) to serve as the Companys registered public accounting firm for the Companys fiscal year ending June 30, 2020. Grant Thornton Audit Pty Ltd (Grant Thornton) had been engaged as Benitec Limiteds independent registered public accounting firm for the periods ended June 30, 2018 and June 30, 2019. On August 14, 2020, Grant Thornton completed its audit of Benitec Limiteds financial statements for the fiscal years ended June 30, 2019 and 2018 prepared in accordance with U.S. GAAP. Upon the completion of Grant Thorntons audit for such periods on August 14, 2020, Grant Thorntons engagement was completed, and Squar Milner became, the Companys independent registered public accounting firm.
Prior to the Re-domiciliation, Grant Thornton acted as the independent registered public accounting firm of Benitec Limited. The financial statements for Benitec Limited for the fiscal years ended June 30, 2019 and 2018 were prepared in accordance with International Accounting Standards and International Financial Reporting Standards (IFRS), and interpretations issued by the International Accounting Standards Board. The audit reports of Grant Thornton on the financial statements of Benitec Limited prepared in accordance with IFRS, and on the financial statements of the Company prepared in accordance with U.S. GAAP, in each case as of and for the years ended June 30, 2019 and 2018, did not contain any adverse opinion or disclaimer of opinion, nor were they qualified or modified as to going concern, uncertainty, audit scope, or accounting principles.
During the fiscal years ended June 30, 2019 and 2018, and in the subsequent period through August 14, 2020, there were (i) no disagreements with Grant Thornton on any matter of accounting principles or practices, financial statement disclosure or auditing scope or procedures, which disagreements, if not resolved to the satisfaction of Grant Thornton, would have caused them to make reference to the subject matter of the disagreements in their audit reports, and (ii) no reportable events, as such term is defined in Item 304(a)(1)(v) of Regulation S-K.
The Company has provided Grant Thornton with a copy of the disclosure set forth in this section and requested that Grant Thornton furnish the Company with a letter addressed to the SEC stating whether or not it agrees with the statements made herein, each as required by applicable SEC rules. A copy of Grant Thorntons letter, dated August 14, 2020, is attached hereto as Exhibit 16.1.
144
Table of Contents
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the SEC a registration statement on Form S-1 under the Securities Act with respect to the securities offered hereby. This prospectus, which constitutes a part of the registration statement, does not contain all of the information set forth in the registration statement or the exhibits and schedules filed therewith. For further information with respect to us and the securities offered hereby, reference is made to the registration statement and the exhibits and schedules filed therewith. Statements contained in this prospectus regarding the contents of any contract or any other document that is filed as an exhibit to the registration statement are not necessarily complete, and as such we refer you to the full text of such contract or other document filed as an exhibit to the registration statement. A copy of the registration statement and the exhibits and schedules filed therewith may be inspected without charge at the public reference room maintained by the SEC, located at 100 F Street N.E., Washington, D.C. 20549, and copies of all or any part of the registration statement may be obtained from such offices upon the payment of the fees prescribed by the SEC. Please call the SEC at 1-800-SEC-0330 for further information about the public reference room. The SEC also maintains a website that contains reports, proxy and information statements and other information regarding registrants that file electronically with the SEC. The Internet address is www.sec.gov.
We are subject to the information and periodic reporting requirements of the Exchange Act, and we file periodic reports, proxy statements and other information with the SEC. These periodic reports, proxy statements and other information are available for inspection and copying at the public reference room and website of the SEC referred to above. We maintain a website at www.benitec.com. You may access our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act with the SEC free of charge at our website as soon as reasonably practicable after such material is electronically filed with, or furnished to, the SEC. Prior to the Re-domiciliation, Benitec Limited was a foreign private issuer. Information concerning Benitec Limited, including its annual reports on Form 20-F and current reports on Form 6-K, is also available free of charge on our website. The information contained in, or that can be accessed through, our website is not incorporated by reference in, and is not part of, this prospectus, and any references to such website or any other website are inactive textual references only. You may also request a copy of these filings, at no cost, by writing us at 3940 Trust Way, Hayward, California 94545 or info@benitec.com or telephoning us at (510) 780-0819.
145
Table of Contents
| Annual Financial Statements |
||||
| F-2 | ||||
| F-3 | ||||
| F-4 | ||||
| Consolidated Statements of Stockholders Equity for the Years Ended June 30, 2019 and 2018 |
F-5 | |||
| Consolidated Statements of Cash Flows for the Years Ended June 30, 2019 and 2018 |
F-6 | |||
| F-7 | ||||
| Interim Unaudited Financial Statements |
||||
| Unaudited Consolidated Balance Sheets as of March 31, 2020 and June 30, 2019 |
F-25 | |||
| F-26 | ||||
| F-27 | ||||
| Unaudited Consolidated Statements of Cash Flows for the Nine Months Ended March 31, 2020 and 2019 |
F-29 | |||
| F-30 | ||||
F-1
Table of Contents
Report of independent registered public accounting firm
Board of Directors and Shareholders
Benitec Biopharma Limited
Opinion on the financial statements
We have audited the accompanying consolidated balance sheets of Benitec Biopharma Limited and subsidiaries (the Company) as of June 30, 2019 and 2018, the related consolidated statements of comprehensive income, changes in shareholders equity, and cash flows for each of the two years in the period ended June 30, 2019, and the related notes (collectively referred to as the financial statements). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of June 30, 2019 and 2018, and the results of its operations and its cash flows for each of the two years in the period ended June 30, 2019, in conformity with accounting principles generally accepted in the United States of America.
Basis for opinion
These financial statements are the responsibility of the Companys management. Our responsibility is to express an opinion on the Companys financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Companys internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ GRANT THORNTON AUDIT PTY LTD
We have served as the Companys auditor since 2010.
Sydney, Australia
August 14, 2020
F-2
Table of Contents
CONSOLIDATED BALANCE SHEETS
June 30, 2019 and 2018
|
2019 (US$000) |
2018 (US$000) |
|||||||
| ASSETS |
|
|||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 15,718 | $ | 11,879 | ||||
| Trade and other receivables |
2,536 | 3,143 | ||||||
| Other assets |
502 | 410 | ||||||
|
|
|
|
|
|||||
| Total current assets |
18,756 | 15,432 | ||||||
| Property and equipment, net |
470 | 236 | ||||||
| Deposits |
9 | 92 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 19,235 | $ | 15,760 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS EQUITY |
|
|||||||
| Current liabilities: |
||||||||
| Trade and other payables |
$ | 2,494 | $ | 1,754 | ||||
| Other current liabilities |
147 | 126 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
2,641 | 1,880 | ||||||
| Other liabilities |
| 36 | ||||||
|
|
|
|
|
|||||
| Total Liabilities |
2,641 | 1,916 | ||||||
|
|
|
|
|
|||||
| Commitments and contingencies (Note 12) |
||||||||
| Stockholders Equity: |
||||||||
| Common stock, authorized 10,000,000 shares, par value $.0001, 856,765 and 856,765 shares issued and outstanding respectively |
1 | 1 | ||||||
| Additional paid In capital |
127,327 | 127,715 | ||||||
| Accumulated deficit |
(108,870 | ) | (112,539 | ) | ||||
| Accumulated other comprehensive loss |
(1,864 | ) | (1,333 | ) | ||||
|
|
|
|
|
|||||
| Total stockholders equity |
16,594 | 13,844 | ||||||
|
|
|
|
|
|||||
| Total liabilities and stockholders equity |
$ | 19,235 | $ | 15,760 | ||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these consolidated financial statements.
F-3
Table of Contents
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
For the Years Ended June 30, 2019 and 2018
|
2019 (US$000) |
2018 (US$000) |
|||||||
| Revenues: |
||||||||
| Revenues from customers |
$ | 11,551 | $ | 292 | ||||
| Government research and development grants |
648 | 3,098 | ||||||
|
|
|
|
|
|||||
| Total revenues |
12,199 | 3,390 | ||||||
|
|
|
|
|
|||||
| Expenses: |
||||||||
| Royalties and license fees |
435 | 349 | ||||||
| Research and development |
4,567 | 7,704 | ||||||
| General and administrative |
4,614 | 4,539 | ||||||
|
|
|
|
|
|||||
| Total expenses |
9,616 | 12,592 | ||||||
|
|
|
|
|
|||||
| Income (Loss) From Operations |
2,583 | (9,202 | ) | |||||
| Other Income (Loss): |
||||||||
| Foreign currency transaction loss |
(75 | ) | (30 | ) | ||||
| Finance income, net |
122 | 187 | ||||||
| Unrealized loss on investment |
(21 | ) | (32 | ) | ||||
|
|
|
|
|
|||||
| Total other income |
26 | 125 | ||||||
|
|
|
|
|
|||||
| Net Income (Loss) |
2,609 | (9,077 | ) | |||||
|
|
|
|
|
|||||
| Other Comprehensive Loss: |
||||||||
| Unrealized foreign currency translation loss |
(531 | ) | (219 | ) | ||||
|
|
|
|
|
|||||
| Total Other Comprehensive Loss |
(531 | ) | (219 | ) | ||||
|
|
|
|
|
|||||
| Total Comprehensive Income (Loss) |
$ | 2,078 | $ | (9,296 | ) | |||
|
|
|
|
|
|||||
| Net Income (Loss) available to Common Stockholders |
$ | 2,609 | $ | (9,077 | ) | |||
|
|
|
|
|
|||||
| Basic and Diluted Weighted Average Number of Shares Outstanding |
856,765 | 701,516 | ||||||
|
|
|
|
|
|||||
| Basic and Diluted Income (Loss) Per Share of Common Stock |
$ | 3.05 | $ | (12.94 | ) | |||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these consolidated financial statements.
F-4
Table of Contents
CONSOLIDATED STATEMENTS OF STOCKHOLDERS EQUITY
For the Years Ended June 30, 2019 and 2018
| Common Stock | ||||||||||||||||||||||||
| Shares |
Amount (US$000) |
Additional Paid in Capital (US$000) |
Accumulated Deficit (US$000) |
Accumulated Other Comprehensive Loss (US$000) |
Total (US$000) |
|||||||||||||||||||
| Balance at June 30, 2017 |
683,809 | $ | 1 | $ | 121,483 | $ | (103,890 | ) | $ | (1,114 | ) | $ | 16,480 | |||||||||||
| Common stock sold for cash, net of offering costs of $232 |
172,956 | | 6,324 | | | 6,324 | ||||||||||||||||||
| Share-based compensation |
| | 336 | | | 336 | ||||||||||||||||||
| Forfeitures of share-based payments |
| | (428 | ) | 428 | | | |||||||||||||||||
| Foreign currency translation loss |
| | | | (219 | ) | (219 | ) | ||||||||||||||||
| Net loss for the year ended June 30, 2018 |
| | | (9,077 | ) | | (9,077 | ) | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balance at June 30, 2018 |
856,765 | $ | 1 | $ | 127,715 | $ | (112,539 | ) | $ | (1,333 | ) | $ | 13,844 | |||||||||||
| Share-based compensation |
| | 672 | | | 672 | ||||||||||||||||||
| Forfeitures of share-based payments |
| | (1,060 | ) | 1,060 | | | |||||||||||||||||
| Foreign currency translation loss |
| | | | (531 | ) | (531 | ) | ||||||||||||||||
| Net income for the year ended June 30, 2019 |
| | | 2,609 | | 2,609 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balance at June 30, 2019 |
856,765 | $ | 1 | $ | 127,327 | $ | (108,870 | ) | $ | (1,864 | ) | $ | 16,594 | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-5
Table of Contents
CONSOLIDATED STATEMENTS OF CASH FLOWS
For the Years Ended June 30, 2019 and 2018
|
2019 (US$000) |
2018 (US$000) |
|||||||
| CASH FLOWS FROM OPERATING ACTIVITIES |
||||||||
| Net income (loss) |
$ | 2,609 | $ | (9,077 | ) | |||
| Adjustments to reconcile net income (loss) to net cash from operating activities |
||||||||
| Depreciation and amortization |
158 | 150 | ||||||
| Loss on disposal of fixed assets |
7 | 1 | ||||||
| Unrealized loss on investment |
21 | 32 | ||||||
| Share-based expense |
672 | 336 | ||||||
| Changes in operating assets and liabilities |
||||||||
| Other financial assets, other assets, and deposits |
(9 | ) | (165 | ) | ||||
| Trade and other receivables |
607 | 233 | ||||||
| Trade and other payables |
740 | 1,050 | ||||||
| Other liabilities |
(15 | ) | (23 | ) | ||||
|
|
|
|
|
|||||
| Net cash provided by (used in) operating activities |
4,790 | (7,463 | ) | |||||
|
|
|
|
|
|||||
| CASH FLOWS FROM INVESTING ACTIVITIES |
||||||||
| Purchases of property and equipment |
(404 | ) | (61 | ) | ||||
| Proceeds from disposal of property and equipment |
4 | 1 | ||||||
|
|
|
|
|
|||||
| Net cash used in investing activities |
(400 | ) | (60 | ) | ||||
|
|
|
|
|
|||||
| CASH FLOWS FROM FINANCING ACTIVITIES |
||||||||
| Proceeds from issues of shares |
| 6,556 | ||||||
| Share issue transaction costs |
| (232 | ) | |||||
|
|
|
|
|
|||||
| Net cash provided by financing activities |
| 6,324 | ||||||
|
|
|
|
|
|||||
| Net increase (decrease) in cash and cash equivalents |
4,390 | (1,199 | ) | |||||
| CASH AND CASH EQUIVALENTSbeginning of period |
11,879 | 13,314 | ||||||
|
|
|
|
|
|||||
| Effects of exchange rate changes on cash and cash equivalents |
(551 | ) | (236 | ) | ||||
|
|
|
|
|
|||||
| CASH AND CASH EQUIVALENTSend of period |
$ | 15,718 | $ | 11,879 | ||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these consolidated financial statements.
F-6
Table of Contents
BENITEC BIOPHARMA LIMITED
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1NATURE OF OPERATIONS AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Nature of Operations
Benitec Biopharma Limited (the Company, we, us or our) was incorporated under the laws of Australia in 1995 and was listed on the Australian Securities Exchange, or ASX, since 1997. Since then, the Company has focused on the development of novel genetic medicines. The proprietary platform, called DNA-directed RNA interference, or ddRNAi, combines RNA interference, or RNAi, with gene therapy to create medicines that facilitate sustained silencing of disease-causing genes.
In October 2012, the Company acquired Tacere Therapeutics, Inc. (Tacere), an RNA interference therapeutics company based in California with a development program focused on Hepatitis C and Age Related Macular Degeneration (AMD). As consideration for the acquisition, we issued a total of 4,092,854 ordinary shares (taking into account a 25:1 share consolidation that became effective in July 2013), representing 9.8% of our issued capital immediately after the transaction, having an aggregate value of $1.5m.
On July 9, 2018, the Company entered into a License and Collaboration Agreement (the Agreement) with Axovant Sciences GMBH (Axovant). The Agreement with the Company granted Axovant an exclusive worldwide license to develop, manufacture, and commercialize products containing the Companys product known as BB-301, which was designed for the potential treatment of Oculopharyngeal Muscular Dystrophy. On June 6, 2019, the termination of the Agreement with Axovant was announced. The termination of the Agreement is effective as of September 3, 2019. The termination discharges all future performance obligations at termination date under the contract.
On November 27, 2019, Benitec Biopharma Limited announced its intention to re-domicile from Australia to the United States of America. Benitec Biopharma Limited implemented a Scheme of Arrangement pursuant to which Benitec Biopharma Inc., a company newly incorporated for the purpose of effecting the re-domiciliation to acquire all Benitec Biopharma Limited shares. Benitec Biopharma Limited shareholders received 1 Benitec Biopharma Inc. share for every 300 Benitec Biopharma Limited shares. In accordance with the U.S. Securities and Exchange Commissions (SEC) Staff Accounting Bulletin Topic 4C, all issued and outstanding shares of the Companys common stock have been retroactively adjusted in these consolidated financial statements to reflect the 300:1 ratio and share consolidation as if it occurred on July 1, 2018.
Benitec Biopharma Limited announced that the transaction to re-domicile from Australia to the United States was fully implemented on April 15, 2020. The re-domiciliation was approved by Benitec Biopharma Limited shareholders at a Scheme Meeting held on March 26, 2020, and confirmed by the Supreme Court of Queensland on March 30, 2020.
The shares of Benitec Biopharma, Inc. common stock issued in connection with the re-domiciliation trade on The Nasdaq Capital Market LLC (Nasdaq) under the symbol BNTC, and Benitec Biopharma Inc. will continue to be subject to the reporting requirements of the SEC and applicable corporate governance rules of Nasdaq.
Benitec Biopharma Ltd. became a wholly owned subsidiary of Benitec Biopharma Inc. Benitec Biopharma Ltd. delisted from the ASX on April 15, 2020.
The consolidated financial statements of Benitec Biopharma Limited consist of Benitec Biopharma Limited and the entities it controlled at the end of, or during, the years ended June 30, 2019, and 2018. The consolidated financial statements are presented in United States dollars.
F-7
Table of Contents
Basis of Presentation
The Companys consolidated financial statements included in this report have been prepared in accordance with accounting principles generally accepted in the United States of America (US GAAP) and pursuant to the rules and regulations of the U.S. Securities and Exchange Commission (SEC). In the opinion of the Companys management, all adjustments (consisting of normal recurring adjustments and reclassifications and non-recurring adjustments) necessary to present fairly our financial position, results of operations, and cash flows for the years ended June 30, 2019 and 2018 have been made.
Reference is frequently made herein to the Financial Accounting Standards Board (the FASB) Accounting Standards Codification (ASC). This is the source of authoritative US GAAP recognized by the FASB to be applied to non-governmental entities.
Principles of Consolidation
The consolidated financial statements include the Companys accounts and the accounts of its wholly-owned subsidiaries. All intercompany transactions and balances have been eliminated.
Use of Estimates
The preparation of financial statements in conformity with US GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from these estimates. Significant estimates in the accompanying consolidated financial statements include the estimates of useful lives for depreciation, valuation of the operating lease liability and related right-of-use asset, allowance for uncollectable receivables, valuation of equity based instruments issued for other than cash, the valuation allowance on deferred tax assets and foreign currency translation due to certain average exchange rates applied in lieu of spot rates on transaction dates.
Foreign Currency Translation and Other Comprehensive Income (Loss)
The Companys functional currency is the Australian dollar (AUD). For financial reporting purposes, the Australian dollar has been translated into United States dollar ($) and/or (USD) as the reporting currency. Assets and liabilities are translated at the exchange rate in effect at the balance sheet date. Revenues and expenses are translated at the average rate of exchange prevailing during the reporting period. Equity transactions are translated at each historical transaction date spot rate. Translation adjustments arising from the use of different exchange rates from period to period are included as a component of stockholders equity as Accumulated other comprehensive income (loss). Gains and losses resulting from foreign currency transactions are included in the statements of operations and comprehensive income (loss) as other comprehensive income (loss).
Other Comprehensive Income (Loss) for all periods presented includes only foreign currency translation gains (losses).
As of June 30, 2019, and 2018, the exchange rates used to translate amounts in Australian dollars into USD for the purposes of preparing the consolidated financial statements were as follows:
| June 30, 2019 |
June 30, 2018 |
|||||||
| Exchange rate on balance sheet dates |
||||||||
| USD: AUD Exchange Rate |
0.70136 | 0.73852 | ||||||
| Average exchange rate for the period |
||||||||
| USD: AUD Exchange Rate |
0.71486 | 0.77432 | ||||||
The exchange rate used to translate amounts in AUD into USD for the year ended June 30, 2017 is: 0.76627 as of the balance sheet date.
F-8
Table of Contents
Fair Value of Financial Instruments and Fair Value Measurements
The Company measures its financial assets and liabilities in accordance with US GAAP using ASC 820, Fair Value Measurements. For certain financial instruments, including cash and cash equivalents, accounts receivable, accounts payable and accrued liabilities, the carrying amounts approximate fair value due to their short maturities.
The Company follows accounting guidance for financial assets and liabilities. ASC 820 defines fair value, provides guidance for measuring fair value and requires certain disclosures. This standard does not require any new fair value measurements, but rather applies to all other accounting pronouncements that require or permit fair value measurements. This guidance does not apply to measurements related to share-based payments. This guidance discusses valuation techniques, such as the market approach (comparable market prices), the income approach (present value of future income or cash flow), and the cost approach (cost to replace the service capacity of an asset or replacement cost).
The guidance utilizes a fair value hierarchy that prioritizes the inputs to valuation techniques used to measure fair value into three broad levels. The following is a brief description of those three levels:
Level 1: Observable inputs such as quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level 2: Inputs, other than quoted prices that are observable, either directly or indirectly. These include quoted prices for similar assets or liabilities in active markets and quoted prices for identical or similar assets or liabilities in markets that are not active.
Level 3: Unobservable inputs in which little or no market data exists, therefore developed using estimates and assumptions developed by us, which reflect those that a market participant would use.
Current classification
An asset is classified as current when: it is either expected to be realized or intended to be sold or consumed in normal operating cycle; it is held primarily for the purpose of trading; it is expected to be realized within 12 months after the reporting period; or the asset is cash or cash equivalent unless restricted from being exchanged or used to settle a liability for at least 12 months after the reporting period.
A liability is classified as current when: it is either expected to be settled in normal operating cycle; it is held primarily for the purpose of trading; it is due to be settled within 12 months after the reporting period; or there is no unconditional right to defer the settlement of the liability for at least 12 months after the reporting period.
Cash and Cash Equivalents
Cash and cash equivalents include cash on hand and at banks, short-term deposits with an original maturity of three months or less with financial institutions, and bank overdrafts. Bank overdrafts are reflected as a current liability on the balance sheets.
Trade and Other Receivables
As amounts become uncollectible, they will be charged to an allowance and operations in the period when a determination of uncollectability is made. Any estimates of potentially uncollectible customer accounts receivable will be made based on an analysis of individual customer and historical write-off experience. The Companys analysis includes the age of the receivable account, creditworthiness of the customer and general economic conditions.
F-9
Table of Contents
Property and Equipment
Property and equipment are stated at cost, net of accumulated depreciation and amortization. Expenditures for maintenance and repairs are expensed as incurred; additions, renewals, and improvements are capitalized. When property and equipment are retired or otherwise disposed of, the related cost and accumulated depreciation and amortization are removed from the respective accounts, and any gain or loss is included in operations. Depreciation and amortization of property and equipment is calculated using the straight-line basis over the following estimated useful lives:
| Leasehold improvements | shorter of the lease term or estimated useful lives | |
| Lab equipment | 3-7 years | |
| Furniture and fixtures | 3-7 years | |
| Computer hardware | 3-5 years | |
| Software | 3-4 years |
Impairment of Long-Lived Assets
Property and equipment are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability of long-lived assets to be held and used is measured by a comparison of the carrying amount of an asset to the estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated undiscounted future cash flows, an impairment charge is recognized by the amount by which the carrying amount of the asset exceeds the fair value of the assets. Fair value is generally determined using the assets expected future discounted cash flows or market value, if readily determinable.
Trade and other payables
These amounts represent liabilities for goods and services provided to the Company prior to the end of the fiscal year and which are unpaid. Due to their short-term nature, they are measured at amortized cost and are not discounted. The amounts are unsecured and are usually paid within 30 days of recognition.
Employee Benefit/Liability
Short-term employee benefits
Liabilities for wages and salaries and other employee benefits expected to be settled within 12 months of the reporting date are measured at the amounts expected to be paid when the liabilities are settled.
Other long-term employee benefits
Employee benefits not expected to be settled within 12 months of the reporting date are measured as the present value of expected future payments to be made in respect of services provided by employees up to the reporting date using the projected unit credit method. Consideration is given to expected future wage and salary levels, experience of employee departures and periods of service. Expected future payments are discounted using market yields at the reporting date on high quality corporate bonds with terms to maturity and currency that match, as closely as possible, the estimated future cash outflows.
Defined contribution superannuation expense
Contributions to defined contribution superannuation plans are expensed in the period in which they are incurred.
Australian Goods and Services Tax (GST) and other similar taxes
Revenues, expenses, and balance sheet items are recognized net of the amount of GST, except payable and receivable balances which are shown inclusive of GST. The GST incurred is payable on revenues to, and recoverable on purchases from, the Australian Taxation Office.
F-10
Table of Contents
Cash flows are presented in the statements of cash flow on a gross basis, except for the GST component of investing and financing activities, which are disclosed as operating cash flows.
Commitments and contingencies are disclosed net of the amount of GST recoverable from, or payable to, the tax authority.
Income Taxes
The Company is governed by Australia and United States income tax laws, which are administered by the Australian Taxation Office and the United States Internal Revenue Service, respectively. The Company follows ASC 740 Accounting for Income Taxes, when accounting for income taxes, which requires an asset and liability approach to financial accounting and reporting for income taxes. Deferred income tax assets and liabilities are computed annually for temporary differences between the financial statements and tax bases of assets and liabilities that will result in taxable or deductible amounts in the future based on enacted tax laws and rates applicable to the periods in which the differences are expected to affect taxable income. Valuation allowances are established when necessary to reduce deferred tax assets to the amount more likely than not to be realized. Income tax expense is the tax payable or refundable for the period plus or minus the change during the period in deferred tax assets and liabilities. As of the date of these consolidated financial statements, the tax years for 2015 to 2019 are subject to examination by the tax authorities.
For uncertain tax positions that meet a more likely than not threshold, the Company recognizes the benefit of uncertain tax positions in the consolidated financial statements. The Companys practice is to recognize interest and penalties, if any, related to uncertain tax positions in income tax expense in the consolidated statements of operations.
Share-Based Compensation
The Company records share-based compensation in accordance with ASC 718, Stock Compensation. ASC 718 requires the fair value of all stock-based employee compensation awarded to employees to be recorded as an expense over the shorter of the service period or the vesting period. The Company values employee and non-employee share-based compensation at fair value using the Black-Scholes Option Pricing Model.
The Company adopted ASU 2018-07 and accounts for non-employee share-based awards in accordance with the measurement and recognition criteria of ASC 718 and recognizes the fair value of such awards over the service period. The Company used the modified prospective method of adoption. There was no cumulative effect of adoption on July 1, 2019.
Basic and Diluted Net Income (Loss) Per Share
Basic net income (loss) per share is calculated by dividing net income (loss) by the weighted-average number of common shares outstanding during the period, Diluted net income (loss) per share is calculated by dividing net income (loss) by the weighted-average number of common shares outstanding plus potential common shares. Stock options, warrants and convertible instruments are considered potential common shares and are included in the calculation of diluted net income (loss) per share using the treasury stock method when their effect is dilutive. Potential common shares are excluded from the calculation of diluted net income (loss) per share when their effect is anti-dilutive. As of June 30, 2019 and 2018, there were 38,326 and 82,481 potential common shares, respectively, that were excluded from the calculation of diluted net income (loss) per share because their effect was anti-dilutive.
Revenue Recognition
The Company adopted and implemented on July 1, 2018, ASC 606Revenue from Contracts with Customers (ASC 606). ASC 606 did not have a material impact on the consolidated financial statements.
F-11
Table of Contents
Upon implementation of ASC 606, the Company recognizes revenue in accordance with that core principle by applying the following steps:
Step 1: Identify the contract(s) with a customer.
Step 2: Identify the performance obligations in the contract.
Step 3: Determine the transaction price.
Step 4: Allocate the transaction price to the performance obligations in the contract.
Step 5: Recognize revenue when (or as) the entity satisfies a performance obligation.
The Company applies judgement in determining whether contracts entered into fall within the scope of ASC 606. In doing so, management considers the commercial substance of the transaction and how risks and benefits of the contract accrue to the various parties to the contract. In determining the accounting treatment of the contract with Axovant, management assessed that the contract was within the scope of ASC 606.
Management has also made the judgement that the grant of the license and transfer of associated know-how and materials are accounted for as one performance obligation as they are not considered to be distinct; they are highly interrelated and could not provide benefits to the customer independently from each other. Judgements were made in relation to the transfer of the license and know-how and whether this should be recognized over time or a point in time. The point in time has been determined with regard to the point at which the transfer of know-how has substantially been completed and the customer has control of the asset and the ability to direct the use of and receive substantially all of the remaining benefits.
Licensing revenues
Revenue from licensees of the Companys intellectual property reflects the transfer of a right to use the intellectual property as it exists at the point in time in which the license is transferred to the customer. Consideration can be variable and is estimated using the most likely amount method. Subsequently, the estimate is constrained until it is highly probable that a significant revenue reversal will not occur when the uncertainty is resolved. Revenue is recognized as or when the performance obligations are satisfied.
The Company recognizes contract liabilities for consideration received in respect of unsatisfied performance obligations and reports these amounts as other liabilities in the consolidated balance sheet. Similarly, if the Company satisfies a performance obligation before it receives the consideration, the Company recognizes either a contract asset or a receivable in its consolidated balance sheet, depending on whether something other than the passage of time is required before the consideration is due.
Royalties
Revenue from licensees of the Companys intellectual property reflect a right to use the intellectual property as it exists at the point in time in which the license is granted. Where consideration is based on sales of product by the licensee, revenue is recognized when the customers subsequent sales of product occurs.
Services revenue
Revenue is earned (constrained by variable considerations) from the provision of research and development services to customers. Services revenue is recognized when performance obligations are either satisfied over time or at a point in time. Generally, the provision of research and development services under a contract with a customer will represent satisfaction of a performance obligation over time where the Company retains the right to payment for services performed but not yet completed.
Government Research and Development Grants
Government grants are recognized at fair value where there is reasonable assurance that the grant will be received and all grant conditions will be met. Grants relating to expense items are recognized as income over the periods necessary to match the grant costs they are compensating.
F-12
Table of Contents
Grant income is generated through the Australian federal governments Research and Development Tax Incentive program, under which the government provides a cash refund for the 43.5% (2018: 43.5%) of eligible research and development expenditures. This grant is available for our research and development activities in Australia, as well as activities in the United States to the extent such U.S.-based expenses relate to our activities in Australia, do not exceed half the expenses for the relevant activities and are approved by the Australian government. Grants are recorded when a reliable estimate can be made. In the twelve months ended June 30, 2019, the Company estimated the grant income that will be receivable following the lodgment of the 2019 tax return.
Research and development expenses
Research and development costs are expensed when incurred.
These costs have been recognized as an expense when incurred. Research and development expenses relate primarily to the cost of conducting clinical and pre-clinical trials. Clinical development costs are a significant component of research and development expenses. Estimates have been used in determining the expense liability under certain clinical trial contracts where services have been performed but not yet invoiced. Generally, the costs, and therefore estimates, associated with clinical trial contracts are based on the number of patients, drug administration cycles, the type of treatment and the outcome being the length of time before actual amounts can be determined will vary depending on length of the patient cycles and the timing of the invoices by the clinical trial partners.
Research and development refundable tax offsets
The Company accounts for the federal government research and development grant tax incentive when a reliable estimate of the amounts receivable can be made. In determining the estimate management reviews historical claims, Government overseas findings enabling the claim of overseas expenditure and the allocation of staff and overheads costs within approved projects. Judgement is also applied in determining the eligibility of the activities undertaken in Australia and overseas. This grant is available for our research and development activities in Australia, as well as activities in the United States to the extent such U.S.-based expenses relate to our activities in Australia, do not exceed half the expenses for the relevant activities and are approved by the Australian government. Grant Income for the years ended June 30, 2019, and 2018, includes an estimate of Research and Development grant receivable for June 30, 2019, and 2018, of $648k and $3,098k respectively (refer to Note 3).
Legal Expenses
All legal costs for litigation are charged to expense as incurred.
New Accounting Standards and Interpretations not yet mandatory or early adopted
Certain FASB Accounting Standard Updates (ASU) that are not effective until after June 30, 2019 are not expected to have a significant effect on the Companys financial position or results of operations.
Future pronouncements are as follows:
ASU 2016-02 - In February 2016, the FASB issued ASU No. 2016-02: Leases (Topic 842) whereby lessees will need to recognize most leases on their balance sheet as a right of use asset and a lease liability. This guidance is effective for interim and annual reporting periods beginning after December 15, 2018. The Company adopted this ASU effective July 1, 2019.
The determination of whether an arrangement is or contains a lease is based on the substance of the arrangement and requires an assessment of whether the fulfilment of the arrangement is dependent on the use of a specific asset or assets and the arrangement conveys a right to use the asset.
F-13
Table of Contents
ImpactThe Company has undertaken a detailed review and has concluded that it will have a material impact on its financial position on the transactions and balances recognized in the consolidated financial statements when it is first adopted for the year ending June 30, 2020 due to the material size of lease entered into by the Company. The Companys only lease is the lease on its research and development facilities. The Companys existing lease commitments are set out in Note 12.
ASU 2016-13 - In June 2016, the FASB issued ASU No. 2016-13: Financial InstrumentsCredit Losses (Topic 326). This ASU represents a significant change in the accounting for credit losses model by requiring immediate recognition of managements estimates of current expected credit losses (CECL). Under the prior model, losses were recognized only as they were incurred, which FASB has noted delayed recognition of expected losses that might not yet have met the threshold of being probable. This guidance is effective for interim and annual reporting periods beginning after December 15, 2019. The Company has determined that it has met the criteria of a smaller reporting company (SRC) as of November 15, 2019. As such, ASU 2019-10: Financial Instruments-Credit Losses, Derivatives and Hedging, and Leases: Effective Dates amended the effective date for the Company to be for reporting periods beginning after December 15, 2022. The Company will adopt this ASU effective July 1, 2023.
ASU 2017-11 - In July 2017, the FASB issued ASU 2017-11 Earnings Per Share (Topic 260). The amendments in the update change the classification of certain equity-linked financial instruments (or embedded features) with down round features. The amendments also clarify existing disclosure requirements for equity-classified instruments. For freestanding equity-classified financial instruments, the amendments require entities that present earnings per share (EPS) in accordance with Topic 260, Earnings Per Share, to recognize the effect of the down round feature when it is triggered. That effect is treated as a dividend and as a reduction of income available to common shareholders in basic EPS. Convertible instruments with embedded conversion options that have down round features would be subject to the specialized guidance for contingent beneficial conversion features (in Subtopic 470-20, DebtDebt with Conversion and Other Options), including related EPS guidance (in Topic 260). For public business entities, the amendments in Part I of this update are effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2018. The Company adopted this ASU effective July 1, 2019.
ASU 2018-07 - In June 2018, the Financial Accounting Standards Board (the FASB) issued Accounting Standards Update (ASU) 2018-07 CompensationStock Compensation (Topic 718). This update is intended to reduce cost and complexity and to improve financial reporting for share-based payments issued to non-employees (for example, service providers, external legal counsel, suppliers, etc.). The ASU expands the scope of Topic 718, CompensationStock Compensation, which currently only includes share-based payments issued to employees, to also include share-based payments issued to non-employees for goods and services. Consequently, the accounting for share-based payments to non-employees and employees will be substantially aligned. This standard will be effective for financial statements issued by public companies for the annual and interim periods beginning after December 15, 2018. Early adoption of the standard is permitted. The standard will be applied in a retrospective approach for each period presented. The Company adopted this ASU, effective July 1, 2019.
NOTE 2GOING CONCERN (US000)
The accompanying consolidated financial statements have been prepared in conformity with US GAAP, which contemplate continuation of the Company as a going concern. For the fiscal year ended June 30, 2019, and 2018, the Company had a net income (loss) of $2,609 and ($9,077), respectively and had net cash provided by (used in) operations of $4,790 and ($7,463), respectively. The Company has recognized the capital raisings in the last three years, performed a review of the cash flow forecasts, considered the cash flow needs of the Company, and believe that there will be sufficient cash to maintain the going concern status of the Company for a period of at least twelve months from the date of this report.
F-14
Table of Contents
While the Company has established some licensing arrangements, the main source of funding subsequent to June 30, 2019, was generated from a public offering which closed on September 30, 2019.
The Company entered into a securities purchase agreement with certain sophisticated and professional investors in the United States to issue 2,800,000 (before the reverse split) American Depositary Shares (ADSs), with each ADS representing 20 fully paid ordinary shares, at a purchase price of US$0.70 per ADS, in a registered direct offering.
The investors were also issued warrants to purchase up to 412,863 ADSs in aggregate, at a purchase price per warrant equal to US$0.6999 per ADS to be issued on exercise of the warrant (Pre-Funded Warrants). The Pre-Funded Warrants may be exercised at any time from issue, in whole or in part, at an exercise price of US$0.0001 per ADS issued on exercise.
The issue of the ADSs and Pre-Funded Warrants raised gross proceeds of approximately US$2.25m exclusive of costs. In a concurrent private placement, the Company agreed to issue additional warrants to the Investors to purchase up to a further 3,212,864 ADSs in aggregate (Purchase Warrants). The issue of the Purchase Warrants was approved by shareholders at the November 2019 Annual General Meeting. The Purchase Warrants were issued for nil consideration, have an exercise price of US$0.70 per ADS and will expire in five years from the date of issuance.
Consistent with the capital raise noted above, historically, the Company has funded its operations primarily from private placements of ordinary shares, including $4.1m in March 2017 and $1.9m in October 2016, a U.S. initial public offering in August 2015 of $13.8m and $28.3m in February 2014. In May 2018, we undertook a placement of 15,444,020 fully paid ordinary shares representing 772,201 ADSs, resulting in $1.96m in gross proceeds. In June 2018, we issued to Nant Capital, 29,305,819 shares in a 1-for-2 entitlement offer resulting in gross proceeds of $4.7m.
The Company has taken to account cumulative research and development grants from the Australian federal government since inception, totaling A$22.935m. Since the Nasdaq listing in July 2015, the Company has earned licensing revenue from licensing our ddRNAi technology to biopharmaceutical companies, totaling A$15.129m.
NOTE 3REVENUES
| Revenues from customers |
2019 (US$000) |
2018 (US$000) |
||||||
| Licensing revenue* |
10,273 | 182 | ||||||
| Royalty revenue* |
183 | 110 | ||||||
| Service revenue* |
1,095 | | ||||||
|
|
|
|
|
|||||
| 11,551 | 292 | |||||||
|
|
|
|
|
|||||
| * | On July 9, 2018, the Company entered into an Agreement with Axovant. The Agreement granted Axovant an exclusive worldwide license to develop, manufacture, and commercialize products containing the Companys product known as BB-301, which was designed for the potential treatment of Oculopharyngeal Muscular Dystrophy. Licensing revenue and royalties increased by $10.2 million, from $0.3 million in fiscal 2018 to $10.5 million in fiscal 2019, primarily due to the license agreement with Axovant, which resulted in an upfront non-refundable license payment of $10 million. Service revenue consists of payments for services provided to Axovant during the license agreement signed in July 2018. On June 6, 2019, the termination of the Agreement with Axovant was announced. The termination of the Agreement is effective as of September 3, 2019. The termination discharges all future performance obligations at termination date under the contract. As such, there are no contract liabilities recognized at June 30, 2019. The Company will retain all financial payments that |
F-15
Table of Contents
| were received during the term of the Axovant License and Collaboration Agreement prior to the termination of the Agreement. Additionally, the Company has a contract asset of approximately $1.7M as of June 30, 2019, related to the contract with Axovant which is included in Trade and other receivables on the consolidated balance sheet. |
| Other revenues |
2019 (US$000) |
2018 (US$000) |
||||||
| Government research and development grants |
648 | 3,098 | ||||||
|
|
|
|
|
|||||
| 648 | 3,098 | |||||||
|
|
|
|
|
|||||
| Disaggregated revenue |
Twelve months ended June 30, 2019 (US$000) | |||||||||||||||
| Licensing | Royalties | Development activities |
Total | |||||||||||||
| Services transferred at a point in time |
10,136 | | | 10,136 | ||||||||||||
| Services transferred over time |
137 | 183 | 1,095 | 1,415 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| 10,273 | 183 | 1,095 | 11,551 | |||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Twelve months ended June 30, 2018 (US$000) | ||||||||||||||||
| Licensing | Royalties | Development activities |
Total | |||||||||||||
| Services transferred at a point in time |
| | | | ||||||||||||
| Services transferred over time |
182 | 110 | | 292 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| 182 | 110 | | 292 | |||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
NOTE 4OTHER INCOME
| Other income |
2019 (US$000) |
2018 (US$000) |
||||||
| Foreign currency transaction loss |
(75 | ) | (30 | ) | ||||
| Finance income |
122 | 187 | ||||||
| Unrealized loss on investment |
(21 | ) | (32 | ) | ||||
|
|
|
|
|
|||||
| 26 | 125 | |||||||
|
|
|
|
|
|||||
F-16
Table of Contents
NOTE 5INCOME TAX BENEFIT
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Current tax |
| | ||||||
| Aggregate income tax benefit |
| | ||||||
|
|
|
|
|
|||||
| Reconciliation of income tax benefit and tax at statutory rate: |
||||||||
| Income (Loss) before income tax benefit |
2,609 | (9,077 | ) | |||||
|
|
|
|
|
|||||
| Tax at the statutory rate of 27.5% (27.5%) |
717 | (2,496 | ) | |||||
| Tax effect amounts which are not deductible/(taxable) in calculating taxable income: |
||||||||
| R&D expenses |
410 | 2,017 | ||||||
| R&D incentive income |
(179 | ) | (870 | ) | ||||
| Legal expenses |
161 | 54 | ||||||
| Share-based payments |
184 | 92 | ||||||
| Timing differences utilized not previously recognized |
(190 | ) | (152 | ) | ||||
| Impact of foreign exchange rate differences |
| | ||||||
|
|
|
|
|
|||||
| 1,103 | (1,355 | ) | ||||||
|
|
|
|
|
|||||
| (Utilization of carried forward losses) tax losses not brought to account |
(1,103 | ) | 1,355 | |||||
|
|
|
|
|
|||||
| Income tax benefit |
| | ||||||
|
|
|
|
|
|||||
Tax losses are recognized only if the consolidated entity considers it is probable that future taxable amounts will be available to utilize those temporary differences and losses.
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Tax losses for which no tax has been recognizedAustralia |
||||||||
| -Tax losses not recognized |
38,662 | 47,598 | ||||||
| -Capital losses not recognized |
909 | 985 | ||||||
| -Other deferred tax assets recognized |
701 | 486 | ||||||
|
|
|
|
|
|||||
| 40,272 | 49,069 | |||||||
|
|
|
|
|
|||||
| Potential tax benefit of tax assets not recognized at 27.5% (27.5%) |
11,075 | 13,494 | ||||||
| Tax losses for which no deferred tax asset has been recognized(US) Tacere |
||||||||
| -Tax losses not recognized |
551 | 655 | ||||||
|
|
|
|
|
|||||
| Potential tax benefit of tax assets not recognized at 21% US |
116 | 138 | ||||||
|
|
|
|
|
|||||
The above potential tax benefit, which excludes tax losses, for deductible temporary differences has not been recognized in the balance sheet as the recovery of this benefit is uncertain.
NOTE 6CASH AND CASH EQUIVALENTS
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Cash at Bank |
10,779 | 7,071 | ||||||
| Term Deposit |
4,939 | 4,808 | ||||||
|
|
|
|
|
|||||
| 15,718 | 11,879 | |||||||
|
|
|
|
|
|||||
F-17
Table of Contents
NOTE 7OTHER ASSETS
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Prepaid expenses |
375 | 314 | ||||||
| Security deposit |
103 | 74 | ||||||
| Other deposit |
23 | | ||||||
| Market value of listed shares |
1 | 22 | ||||||
|
|
|
|
|
|||||
| 502 | 410 | |||||||
|
|
|
|
|
|||||
NOTE 8TRADE AND OTHER RECEIVABLES
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Research & development grants |
636 | 3,043 | ||||||
| Other receivables* |
1,900 | 100 | ||||||
|
|
|
|
|
|||||
| 2,536 | 3,143 | |||||||
|
|
|
|
|
|||||
| * | The Company has a contract asset of approximately $1.7M as of June 30, 2019, related to the contract with Axovant which is included in other receivables on the consolidated balance sheet. On June 6, 2019, the termination of the Agreement with Axovant was announced. The termination of the Agreement is effective as of September 3, 2019. |
NOTE 9PROPERTY AND EQUIPMENT, NET
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Leasehold improvements |
77 | 58 | ||||||
| Lab equipment |
1,014 | 635 | ||||||
| Furniture and fixtures |
6 | 31 | ||||||
| Computer hardware |
32 | 43 | ||||||
| Software |
11 | 11 | ||||||
|
|
|
|
|
|||||
| Total property and equipment, gross |
1,140 | 778 | ||||||
| Accumulated depreciation and amortization |
(670 | ) | (542 | ) | ||||
|
|
|
|
|
|||||
| Total property and equipment, net |
470 | 236 | ||||||
|
|
|
|
|
|||||
Depreciation expense for the years ended June 30, 2019 and 2018 were $158k and $150k, respectively.
NOTE 10TRADE AND OTHER PAYABLES, OTHER CURRENT LIABILITIES
Trade and other payables
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Trade payable |
1,474 | 428 | ||||||
| Accrued license fees |
385 | 176 | ||||||
| Accrued research and development fees |
498 | 731 | ||||||
| Accrued professional fees |
61 | 134 | ||||||
| Other payables |
76 | 285 | ||||||
|
|
|
|
|
|||||
| 2,494 | 1,754 | |||||||
|
|
|
|
|
|||||
F-18
Table of Contents
Other current liabilities
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Employee benefits |
140 | 108 | ||||||
| Other |
7 | 18 | ||||||
|
|
|
|
|
|||||
| 147 | 126 | |||||||
NOTE 11STOCKHOLDERS EQUITY
Common Stock
Common stock entitles the holder to participate in dividends and the proceeds on the winding up of the Company in proportion to the number of and amounts paid on the shares held. The fully paid shares have a par value of $0.0001 and the Company has authorized shares of 10 million.
In accordance with SEC Staff Accounting Bulletin Topic 4C, all issued and outstanding shares of the Companys common stock have been retroactively adjusted in these consolidated financial statements to reflect the 300:1 ratio and share consolidation as if it occurred on July 1, 2018.
Equity Incentive Plan
Employee Share Option Plan
Upon the re-domiciliation, the Company assumed Benitec Biopharma Limiteds obligations with respect to the settlement of option that were issued by Benitec Biopharma Limited prior to the re-domiciliation pursuant to the Benitec Officers and Employees Share Option Plan (the Plan). This includes the Companys assumptions of the Plan and all award agreements pursuant to which each of the options were granted. Prior to the re-domiciliation, Benitec Biopharma Limited granted options under the Plan but after the re-domiciliation, no new options will be issued under the Plan. Each option when exercised entitles the option holder to one share in Benitec Biopharma Inc. Options are exercisable on or before an expiry date, do not carry any voting or dividend rights and are not transferable except on death of the option holder or in certain other limited circumstances. Employee options vest one third on each anniversary of the applicable grant date for three years. If an employee dies, retirees or otherwise leaves the organization and certain exercise conditions have been satisfied, generally, the employee has 12 months to exercise their options or the options are cancelled.
F-19
Table of Contents
Equity Awards
The activity related to equity awards, which comprised of stock options during the years ended June 30, 2019, and 2018, is summarized as follows:
| Stock Options |
Weighted-average Exercise Price |
Weighted-average Remaining Contractual Term |
Aggregate Intrinsic Value |
|||||||||||||
| Outstanding at July 1, 2017 |
32,413 | $ | 191.40 | |||||||||||||
| Granted |
66,500 | $ | 48.30 | |||||||||||||
| Exercised |
| | ||||||||||||||
| Forfeited |
(17,322 | ) | $ | 94.50 | ||||||||||||
|
|
|
|||||||||||||||
| Outstanding at June 30, 2018 |
81,591 | $ | 92.10 | 3.83 years | | |||||||||||
|
|
|
|||||||||||||||
| Exercisable at June 30, 2018 |
21,758 | $ | 92.10 | |||||||||||||
|
|
|
|||||||||||||||
| Granted |
27,333 | $ | 43.20 | |||||||||||||
| Exercised |
| | ||||||||||||||
| Forfeited |
(22,457 | ) | $ | 125.40 | ||||||||||||
|
|
|
|||||||||||||||
| Outstanding at June 30, 2019 |
86,467 | $ | 64.20 | 3.58 years | | |||||||||||
|
|
|
|||||||||||||||
| Exercisable at June 30, 2019 |
27,022 | $ | 64.20 | |||||||||||||
Share-Based Compensation Expense
The classification of share-based compensation expense for the years ended:
| June 30, 2019 (US$000) |
June 30, 2018 (US$000) |
|||||||
| Research and development |
87 | 172 | ||||||
| General and administrative |
585 | 164 | ||||||
|
|
|
|
|
|||||
| Total share-based compensation expense |
672 | 336 | ||||||
|
|
|
|
|
|||||
As of June 30, 2019, and 2018, there was $907k and $306k of unrecognized share-based compensation expense related to shares of common stock issued under the Plan.
Capital risk management
The Companys objectives when managing capital is to safeguard its ability to continue as a going concern, so that it can provide returns for shareholders and benefits for other stakeholders and to maintain an optimum capital structure to reduce the cost of capital.
The capital structure of the Company consists of cash and cash equivalents and equity attributable to equity holders. Operating globally, the Company develops specialty pharmaceutical products. The overall strategy of the Company is to continue its drug development programs, which depends on selling assets and raising additional equity to fund the activities.
The capital risk management policy remains unchanged from the prior year.
NOTE 12COMMITMENTS AND CONTINGENCIES
Operating Leases of Premises
In 2016, the Company entered into an agreement to lease office space for its corporate headquarters in Australia for a period of 3 years commencing on October 1, 2016. The lease contains customary default provisions,
F-20
Table of Contents
representations, warranties, and covenants. The lease is classified as an operating lease. Additionally, the Company entered an agreement to lease office space for its wholly-owned subsidiary Tacere in California. The lease originally expired on April 30, 2018 and was extended through June 14, 2018. The Company agreed to extend the lease for the period of June 15, 2018 to June 14, 2021. An option to extend the lease from June 15, 2021 to June 14, 2022, has been exercised by the Company. The lease contains customary default provisions, representations, warranties, and covenants. This lease is classified as an operating lease.
Future minimum lease payments under the Lease as of June 30, 2019 are as follows:
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Committed at the reporting date but not recognized as a liability: |
||||||||
| Within one year |
203 | 162 | ||||||
| One to five years |
443 | 216 | ||||||
|
|
|
|
|
|||||
| 646 | 378 | |||||||
|
|
|
|
|
|||||
Rent expense for the years ended June 30, 2019, and 2018 was $221k and $176k, respectively.
Contingent Liabilities
There are no contingent liabilities as of June 30, 2019.
NOTE 13RELATED PARTY TRANSACTIONS
Parent entity
Benitec Biopharma Limited is the parent entity.
Subsidiaries
Interests in subsidiaries are set out in note 14.
Compensation
The aggregate compensation made to directors and other members of key management personnel of the Company is set out below:
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Short-term employee benefits |
814 | 1,361 | ||||||
| Post-employment benefits |
26 | 63 | ||||||
| Long-term employee benefits |
| | ||||||
| Share-based payments |
506 | 190 | ||||||
|
|
|
|
|
|||||
| Total key management compensation |
1,346 | 1,614 | ||||||
|
|
|
|
|
|||||
| The following transactions occurred with related parties: |
||||||||
| Payment for other expenses: |
||||||||
| Legal services paid/payable to Francis Abourizk Lightowlers, a law firm in which Mr. Peter Francis is a partner and has a beneficial interest. |
1 | 6 | ||||||
|
|
|
|
|
|||||
| Annabel West, the wife of Greg West, our former Chief Executive Officer, was employed as a part-time clerical administrative assistant. |
| 31 | ||||||
|
|
|
|
|
|||||
F-21
Table of Contents
Receivable from and payable to related parties
There were no trade receivables from or trade payables to related parties at the current and previous reporting date.
Loans to/from related parties
There were no loans to or from related parties at the current and previous reporting date.
Terms and conditions
All transactions were made on normal commercial terms and conditions and at market rates.
NOTE 14INTERESTS IN SUBSIDIAIRIES
| Principal place of business/country of incorporation |
2019 | 2018 | ||||||||
| Benitec Australia Limited |
Australia | 100 | % | 100 | % | |||||
| Benitec Limited |
United Kingdom | 100 | % | 100 | % | |||||
| Benitec, Inc. |
USA | 100 | % | 100 | % | |||||
| Benitec LLC |
USA | 100 | % | 100 | % | |||||
| RNAi Therapeutics, Inc. |
USA | 100 | % | 100 | % | |||||
| Tacere Therapeutics, Inc.* |
USA | 100 | % | 100 | % | |||||
| * | Note Tacere year-end is December 31 which was the year-end date when the Company was acquired. |
NOTE 15EARNINGS PER SHARE
| 2019 (US$000) |
2018 (US$000) |
|||||||
| Profit/(Loss) after income tax attributable to the owners of Benitec Biopharma Limited |
2,609 | (9,077 | ) | |||||
| Weighted number of shares used in calculating basic and diluted earnings per share |
856,765 | 701,516 | ||||||
|
|
|
|
|
|||||
| Basic and diluted earnings per share |
$ | 3.05 | ($ | 12.94 | ) | |||
|
|
|
|
|
|||||
Outstanding options to acquire ordinary shares are not considered dilutive for the years ended June 30, 2019 and June 30, 2018, because they are anti-dilutive, as the strike price was higher than the share price.
NOTE 16SUBSEQUENT EVENTS
On June 6, 2019, the termination of the Agreement with Axovant was announced. The termination of the Agreement is effective as of September 3, 2019. The termination discharges all future performance obligations at termination date under the contract. As such, there are no contract liabilities recognized at June 30, 2019.
On September 30, 2019, Benitec Biopharma Limited entered into a securities purchase agreement (SPA) with certain sophisticated and professional investors in the United States to issue 2,800,000 American Depositary Shares (ADSs), with each ADS representing 20 fully paid ordinary shares, at a purchase price of US$0.70 per ADS, in a registered direct offering. The Investors were also issued warrants to purchase up to 412,863 ADSs in aggregate, at a purchase price per warrant equal to US$0.6999 per ADS to be issued on exercise of the warrant (Pre-Funded Warrants). The Pre-Funded Warrants may be exercised at any time from issue, in whole or in part, at an exercise price of US$0.0001 per ADS issued on exercise (subject to certain adjustments), provided that the beneficial ownership of the relevant Investor in the total number of ADSs on issue may not exceed 9.99%. The Pre-Funded Warrants do not expire.
F-22
Table of Contents
The issue of the ADSs and Pre-Funded Warrants raised gross proceeds of approximately US$2.25 million exclusive of costs. The ADSs (and the underlying ordinary shares) and Pre-Funded Warrants were issued without shareholder approval under the Companys existing placement capacity under ASX Listing Rules 7.1 and 7.1A.
In a concurrent private placement, the Company has also agreed to issue additional warrants to the Investors to purchase up to a further 3,212,864 ADSs in aggregate (Purchase Warrants). The issue of the Purchase Warrants has been approved by shareholders and the Purchase Warrants were issued for nil consideration, will have an exercise price of US$0.70 per ADS and will expire in five years from the date of issuance.
On November 27, 2019, Benitec Biopharma Limited announced its intention to re-domicile from Australia to the United States of America. Benitec Biopharma Limited implemented a Scheme of Arrangement pursuant to which Benitec Biopharma Inc., a company newly incorporated for the purpose of effecting the re-domiciliation to acquire all Benitec Biopharma Limited shares. Benitec Biopharma Limited shareholders received 1 Benitec Biopharma Inc. share for every 300 Benitec Biopharma Limited shares.
Benitec Biopharma Limited announced that the transaction to re-domicile from Australia to the United States was fully implemented on April 15, 2020. The re-domiciliation was approved by Benitec shareholders at a Scheme Meeting held on March 26, 2020, and confirmed by the Supreme Court of Queensland on March 30, 2020.
The shares of Benitec Biopharma, Inc. common stock issued in connection with the re-domiciliation trade on The Nasdaq Capital Market LLC (Nasdaq) under the symbol BNTC, and Benitec Biopharma Inc. will continue to be subject to the reporting requirements of the U.S. Securities and Exchange Commission (SEC) and applicable corporate governance rules of Nasdaq.
Benitec Biopharma Limited became a wholly owned subsidiary of Benitec Biopharma Inc. Benitec Biopharma Limited delisted from the ASX on April 15, 2020.
The Company is closely monitoring the impact of the pandemic of the novel strain of coronavirus COVID-19 (COVID-19) on all aspects of its business and geographies, including how it will impact its team members, suppliers, vendors and business partners. While the Company did experience some disruption from COVID-19 through the delay of the initiation of the pre-clinical trials through the date of issuance, it is unable to predict the impact that COVID-19 will have on its financial position and operating results due to numerous uncertainties.
F-23
Table of Contents
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| Unaudited Consolidated Balance Sheets as of March 31, 2020 and June 30, 2019 |
F-25 | |||
| F-26 | ||||
| F-27 | ||||
| Unaudited Consolidated Statements of Cash Flows for the Nine Months Ended March 31, 2020 and 2019 |
F-29 | |||
| F-30 | ||||
F-24
Table of Contents
CONSOLIDATED BALANCE SHEETS
|
March 2020 (US$000) (Unaudited) |
June 2019 (US$000) |
|||||||
| ASSETS |
|
|||||||
| Current Assets: |
||||||||
| Cash and cash equivalents |
$ | 11,400 | $ | 15,718 | ||||
| Trade and other receivables |
818 | 2,536 | ||||||
| Other assets |
510 | 502 | ||||||
|
|
|
|
|
|||||
| Total Current Assets |
12,728 | 18,756 | ||||||
| Property and equipment, net |
402 | 470 | ||||||
| Deposits |
9 | 9 | ||||||
| Right of use assets |
441 | | ||||||
|
|
|
|
|
|||||
| Total Assets |
$ | 13,580 | $ | 19,235 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS EQUITY |
|
|||||||
| Current Liabilities: |
||||||||
| Trade and other payables |
$ | 613 | $ | 2,494 | ||||
| Accrued employee benefits |
181 | 147 | ||||||
| Lease liabilities, current portion |
187 | | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
981 | 2,641 | ||||||
| Lease liabilities, less current portion |
263 | | ||||||
|
|
|
|
|
|||||
| Total Liabilities |
1,244 | 2,641 | ||||||
|
|
|
|
|
|||||
| Commitments and contingencies (Note 12) |
||||||||
| Stockholders Equity: |
||||||||
| Common stock , authorized 10,000,000 shares, par value $.0001, 1,070,957 and 856,765 shares issued and outstanding respectively |
1 | 1 | ||||||
| Additional paid In capital |
128,897 | 127,327 | ||||||
| Accumulated deficit |
(113,829 | ) | (108,870 | ) | ||||
| Accumulated other comprehensive loss |
(2,733 | ) | (1,864 | ) | ||||
|
|
|
|
|
|||||
| Total Stockholders Equity |
12,336 | 16,594 | ||||||
|
|
|
|
|
|||||
| Total Liabilities and Stockholders Equity |
$ | 13,580 | $ | 19,235 | ||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these consolidated financial statements.
F-25
Table of Contents
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
(Unaudited)
| Three Months Ended | Nine Months Ended | |||||||||||||||
|
March 2020 (US$000) |
March 2019 (US$000) |
March 2020 (US$000) |
March 2019 (US$000) |
|||||||||||||
| Revenues: |
||||||||||||||||
| Revenues from customers |
$ | 28 | $ | 499 | $ | 137 | $ | 10,955 | ||||||||
| Government research and development grants |
| 111 | | 601 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total revenues |
28 | 560 | 137 | 11,556 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Expenses: |
||||||||||||||||
| Royalties and license fees |
47 | 30 | (233 | ) | 405 | |||||||||||
| Research and development |
805 | 1,159 | 2,095 | 3,304 | ||||||||||||
| General and administrative |
1,287 | 1,103 | 3,669 | 3,754 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total expenses |
2,139 | 2,292 | 5,531 | 7,463 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Income (Loss) from Operations |
(2,111 | ) | (1,732 | ) | (5,394 | ) | 4,093 | |||||||||
| Other Income (Loss): |
||||||||||||||||
| Foreign currency transaction gain (loss) |
(7 | ) | 5 | 4 | (75 | ) | ||||||||||
| Finance income, net |
16 | 24 | 52 | 80 | ||||||||||||
| Unrealized loss on investment |
| (1 | ) | (1 | ) | (20 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total other income (loss) |
9 | 28 | 55 | (15 | ) | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net Income (Loss) |
(2,102 | ) | (1,704 | ) | (5,339 | ) | 4,078 | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Other Comprehensive Income (Loss): |
||||||||||||||||
| Unrealized foreign currency translation loss |
(901 | ) | 45 | (869 | ) | (36 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total Other Comprehensive Loss |
(901 | ) | 45 | (869 | ) | (36 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total Comprehensive Income (Loss) |
$ | (3,303 | ) | $ | (1,659 | ) | $ | (6,208 | ) | $ | 4,042 | |||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net Income (Loss) available to Common Stockholders |
$ | (2,102 | ) | $ | (1,704 | ) | $ | (5,339 | ) | $ | 4,078 | |||||
|
|
|
|
|
|
|
|
|
|||||||||
| Basic and Diluted Weighted Average Number of Shares Outstanding |
1,070,957 | 856,765 | 995,246 | 856,765 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Basic and Diluted Income (Loss) Per Share of Common Stock |
$ | (1.96 | ) | $ | (1.99 | ) | $ | (5.36 | ) | $ | 4.76 | |||||
|
|
|
|
|
|
|
|
|
|||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-26
Table of Contents
CONSOLIDATED STATEMENTS OF STOCKHOLDERS EQUITY
(Unaudited)
| Common Stock | ||||||||||||||||||||||||
| Shares |
Amount (US$000) |
Additional Paid in Capital (US$000) |
Accumulated Deficit (US$000) |
Accumulated Other Comprehensive Loss (US$000) |
Total (US$000) |
|||||||||||||||||||
| Balance at June 30, 2018 |
856,765 | $ | 1 | $ | 127,715 | $ | (112,539 | ) | $ | (1,333 | ) | $ | 13,844 | |||||||||||
| Share-based compensation |
| | 35 | | | 35 | ||||||||||||||||||
| Forfeiture of share-based payments |
| | (485 | ) | 485 | | | |||||||||||||||||
| Foreign currency translation loss |
| | | | (128 | ) | (128 | ) | ||||||||||||||||
| Net income for the period ended September 30, 2018 |
| | | 8,342 | | 8,342 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balance at September 30, 2018 |
856,765 | $ | 1 | $ | 127,265 | $ | (103,712 | ) | $ | (1,461 | ) | $ | 22,093 | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Share-based compensation |
| | 250 | | | 250 | ||||||||||||||||||
| Forfeiture of share-based payments |
| | 20 | (20 | ) | | | |||||||||||||||||
| Foreign currency translation gain |
| | | | 47 | 47 | ||||||||||||||||||
| Net loss for the period ended December 31, 2018 |
| | | (2,560 | ) | | (2,560 | ) | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balance at December 31, 2018 |
856,765 | $ | 1 | $ | 127,535 | $ | (106,292 | ) | $ | (1,414 | ) | $ | 19,830 | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Share-based compensation |
| | 282 | | | 282 | ||||||||||||||||||
| Foreign currency translation gain |
| | | | 45 | 45 | ||||||||||||||||||
| Net loss for the period ended March 31, 2019 |
| | | (1,704 | ) | | (1,704 | ) | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balance at March 31, 2019 |
856,765 | $ | 1 | $ | 127,817 | $ | (107,996 | ) | $ | (1,369 | ) | $ | 18,453 | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-27
Table of Contents
BENITEC BIOPHARMA LIMITED
CONSOLIDATED STATEMENTS OF STOCKHOLDERS EQUITY
(Unaudited)
| Common Stock | ||||||||||||||||||||||||
| Shares |
Amount (US$000) |
Additional Paid in Capital (US$000) |
Accumulated Deficit (US$000) |
Accumulated Other Comprehensive Loss (US$000) |
Total (US$000) |
|||||||||||||||||||
| Balance at June 30, 2019 |
856,765 | $ | 1 | $ | 127,327 | $ | (108,870 | ) | $ | (1,864 | ) | $ | 16,594 | |||||||||||
| Common stock sold for cash, net of offering costs of $240 |
186,666 | | 1,720 | | | 1,720 | ||||||||||||||||||
| Issuance of pre-purchased warrants, net of transaction costs of $240 |
| | 50 | | | 50 | ||||||||||||||||||
| Share-based compensation |
| | 55 | | | 55 | ||||||||||||||||||
| Forfeiture of share-based payments |
| | (61 | ) | 61 | | | |||||||||||||||||
| Foreign currency translation loss |
| | | | (304 | ) | (304 | ) | ||||||||||||||||
| Net loss for the period ended September 30, 2019 |
| | | (1,147 | ) | | (1,147 | ) | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balance at September 30, 2019 |
1,043,431 | $ | 1 | $ | 129.091 | $ | (109,956 | ) | $ | (2,168 | ) | $ | 16,968 | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Exercise of pre-purchased warrants |
27,526 | | | | | | ||||||||||||||||||
| Share-based compensation |
| | 37 | | | 37 | ||||||||||||||||||
| Forfeiture of share-based payments |
| | (319 | ) | 319 | | | |||||||||||||||||
| Foreign currency translation gain |
| | | | 336 | 336 | ||||||||||||||||||
| Net loss for the period ended December 31, 2019 |
| | | (2,090 | ) | | (2,090 | ) | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balance at December 31, 2019 |
1,070,957 | $ | 1 | $ | 128,809 | $ | (111,727 | ) | $ | (1,832 | ) | $ | 15,251 | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Share-based compensation |
| | 88 | | | 88 | ||||||||||||||||||
| Foreign currency translation loss |
| | | | (901 | ) | (901 | ) | ||||||||||||||||
| Net loss for the period ended March 31, 2020 |
| | | (2,102 | ) | | (2,102 | ) | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Balance at March 31, 2020 |
1,070,957 | $ | 1 | $ | 128,897 | $ | (113,829 | ) | $ | (2,733 | ) | $ | 12,336 | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-28
Table of Contents
BENITEC BIOPHARMA LIMITED
CONSOLIDATED STATEMENTS OF CASH FLOWS
| Nine Months Ended | ||||||||
|
March 2020 (US$000) |
March 2019 (US$000) |
|||||||
| CASH FLOWS FROM OPERATING ACTIVITIES |
||||||||
| Net income (loss) |
$ | (5,339 | ) | $ | 4,078 | |||
| Adjustments to reconcile net income (loss) to net cash from operating |
||||||||
| Depreciation and amortization |
288 | 107 | ||||||
| Loss on disposal of fixed assets |
1 | 4 | ||||||
| Unrealized loss on investment |
1 | 20 | ||||||
| Share based expense |
180 | 567 | ||||||
| Changes in operating assets and liabilities |
||||||||
| Trade and other receivables |
2,494 | 2,090 | ||||||
| Other assets |
(69 | ) | (178 | ) | ||||
| Trade and other payables |
(1,197 | ) | 193 | |||||
| Other liabilities |
(749 | ) | (276 | ) | ||||
|
|
|
|
|
|||||
| Net cash provided by (used in) operating activities |
(4,390 | ) | 6,605 | |||||
|
|
|
|
|
|||||
| CASH FLOWS FROM INVESTING ACTIVITIES |
||||||||
| Purchases of property and equipment |
(82 | ) | (293 | ) | ||||
| Proceeds from disposal of property and equipment |
1 | | ||||||
|
|
|
|
|
|||||
| Net cash used in investing activities |
(81 | ) | (293 | ) | ||||
|
|
|
|
|
|||||
| CASH FLOWS FROM FINANCING ACTIVITIES |
||||||||
| Proceeds from issues of shares |
2,250 | | ||||||
| Share issue transaction costs |
(480 | ) | | |||||
|
|
|
|
|
|||||
| Net cash provided by financing activities |
1,770 | | ||||||
|
|
|
|
|
|||||
| Net increase (decrease) in cash and cash equivalents |
(2,701 | ) | 6,312 | |||||
| CASH AND CASH EQUIVALENTSbeginning of period |
15,718 | 11,879 | ||||||
|
|
|
|
|
|||||
| Effects of exchange rate changes on cash and cash equivalents |
(1,617 | ) | (514 | ) | ||||
|
|
|
|
|
|||||
| CASH AND CASH EQUIVALENTSend of period |
$ | 11,400 | $ | 17,677 | ||||
|
|
|
|
|
|||||
| Supplemental disclosure of non-cash investing and financing activity: |
||||||||
| Initial measurement of operating lease right-of-use assets and liabilities |
$ | (579 | ) | $ | | |||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these consolidated financial statements.
F-29
Table of Contents
BENITEC BIOPHARMA LIMITED
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1NATURE OF OPERATIONS AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Nature of Operations
Benitec Biopharma Limited (the Company, we, us or our) was incorporated under the laws of Australia in 1995 and was listed on the Australian Securities Exchange, or ASX, since 1997. Since then, the Company has focused on the development of novel genetic medicines. The proprietary platform, called DNA-directed RNA interference, or ddRNAi, combines RNA interference, or RNAi, with gene therapy to create medicines that facilitate sustained silencing of disease-causing genes.
In October 2012, the Company acquired Tacere Therapeutics, Inc. (Tacere), an RNA interference therapeutics company based in California with a development program focused on Hepatitis C and Age Related Macular Degeneration (AMD). As consideration for the acquisition, we issued a total of 4,092,854 ordinary shares (taking into account a 25:1 share consolidation that became effective in July 2013), representing 9.8% of our issued capital immediately after the transaction, having an aggregate value of $1.5m.
On July 9, 2018, the Company entered into a License and Collaboration Agreement (the Agreement) with Axovant Sciences GMBH (Axovant). The Agreement with the Company granted Axovant an exclusive worldwide license to develop, manufacture, and commercialize products containing the Companys product known as BB-301, which was designed for the potential treatment of Oculopharyngeal Muscular Dystrophy. On June 6, 2019, the termination of the Agreement with Axovant was announced. The termination of the Agreement is effective as of September 3, 2019. The termination discharges all future performance obligations at termination date under the contract.
On November 27, 2019, Benitec Biopharma Limited announced its intention to re-domicile from Australia to the United States of America. Benitec Biopharma Limited implemented a Scheme of Arrangement pursuant to which Benitec Biopharma Inc., a company newly incorporated for the purpose of effecting the re-domiciliation to acquire all Benitec Biopharma Limited shares. Benitec Biopharma Limited shareholders received 1 Benitec Biopharma Inc. share for every 300 Benitec Biopharma Limited shares. In accordance with the U.S. Securities and Exchange Commissions (SEC) Staff Accounting Bulletin Topic 4C, all issued and outstanding shares of the Companys common stock have been retroactively adjusted in these consolidated financial statements to reflect the 300:1 ratio and share consolidation as if it occurred on July 1, 2018.
Benitec Biopharma Limited announced that the transaction to re-domicile from Australia to the United States was fully implemented on April 15, 2020. The re-domiciliation was approved by Benitec Biopharma Limited shareholders at a Scheme Meeting held on March 26, 2020, and confirmed by the Supreme Court of Queensland on March 30, 2020.
The shares of Benitec Biopharma, Inc. common stock issued in connection with the re-domiciliation trade on The Nasdaq Capital Market LLC (Nasdaq) under the symbol BNTC, and Benitec Biopharma Inc. will continue to be subject to the reporting requirements of the SEC and applicable corporate governance rules of Nasdaq.
Benitec Biopharma Limited became a wholly owned subsidiary of Benitec Biopharma Inc. Benitec Biopharma Limited delisted from the ASX on April 15, 2020.
The consolidated financial statements of Benitec Biopharma Limited consist of Benitec Biopharma Limited and the entities it controlled at the end of, or during, the three and nine months ended March 31, 2020, and 2019, and the years ended June 30, 2019 and 2018. The consolidated financial statements are presented in United States dollars.
F-30
Table of Contents
The Companys fiscal year-end is June 30. References to a particular fiscal year are to our fiscal year ended June 30 of that calendar year.
Basis of Presentation
The Companys consolidated financial statements included in this report have been prepared in accordance with accounting principles generally accepted in the United States of America (US GAAP) and pursuant to the rules and regulations of the U.S. Securities and Exchange Commission (SEC). In the opinion of the Companys management, all adjustments (consisting of normal recurring adjustments and reclassifications and non-recurring adjustments) necessary to present fairly our financial position, results of operations, and cash flows for the three and nine months ended March 31, 2020 and 2019, and the years ended June 30, 2019 and 2018 have been made.
Reference is frequently made herein to the Financial Accounting Standards Board (the FASB) Accounting Standards Codification (ASC). This is the source of authoritative US GAAP recognized by the FASB to be applied to non-governmental entities.
Principles of Consolidation
The consolidated financial statements include the Companys accounts and the accounts of its wholly-owned subsidiaries. All intercompany transactions and balances have been eliminated.
Use of Estimates
The preparation of financial statements in conformity with US GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from these estimates. Significant estimates in the accompanying consolidated financial statements include the estimates of useful lives for depreciation, valuation of the operating lease liability and related right-of-use asset, allowance for uncollectable receivables, valuation of equity based instruments issued for other than cash, the valuation allowance on deferred tax assets and foreign currency translation due to certain average exchange rates applied in lieu of spot rates on transaction dates.
Foreign Currency Translation and Other Comprehensive Income (Loss)
The Companys functional currency is the Australian dollar (AUD). For financial reporting purposes, the Australian dollar has been translated into United States dollar ($) and/or (USD) as the reporting currency. Assets and liabilities are translated at the exchange rate in effect at the balance sheet date. Revenues and expenses are translated at the average rate of exchange prevailing during the reporting period. Equity transactions are translated at each historical transaction date spot rate. Translation adjustments arising from the use of different exchange rates from period to period are included as a component of stockholders equity as Accumulated other comprehensive income (loss). Gains and losses resulting from foreign currency transactions are included in the statements of operations and comprehensive income (loss) as other comprehensive income (loss).
Other Comprehensive Income (Loss) for all periods presented includes only foreign currency translation gains (losses).
As of March 31, 2020, June 30, 2019, and March 31, 2019, the exchange rates used to translate amounts in Australian dollars into USD for the purposes of preparing the consolidated financial statements were as follows:
| March 31, 2020 |
June 30, 2019 |
March 31, 2019 |
||||||||||
| Exchange rate on balance sheet dates |
||||||||||||
| USD: AUD Exchange Rate |
0.6140 | 0.70136 | 0.7097 | |||||||||
| Average exchange rate for the period |
||||||||||||
| USD: AUD Exchange Rate |
0.6757 | 0.71486 | 0.7201 | |||||||||
F-31
Table of Contents
Fair Value of Financial Instruments and Fair Value Measurements
The Company measures its financial assets and liabilities in accordance with US GAAP using ASC 820, Fair Value Measurements. For certain financial instruments, including cash and cash equivalents, accounts receivable, accounts payable and accrued liabilities, the carrying amounts approximate fair value due to their short maturities.
The Company follows accounting guidance for financial assets and liabilities. ASC 820 defines fair value, provides guidance for measuring fair value and requires certain disclosures. This standard does not require any new fair value measurements, but rather applies to all other accounting pronouncements that require or permit fair value measurements. This guidance does not apply to measurements related to share-based payments. This guidance discusses valuation techniques, such as the market approach (comparable market prices), the income approach (present value of future income or cash flow), and the cost approach (cost to replace the service capacity of an asset or replacement cost).
The guidance utilizes a fair value hierarchy that prioritizes the inputs to valuation techniques used to measure fair value into three broad levels. The following is a brief description of those three levels:
Level 1: Observable inputs such as quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level 2: Inputs, other than quoted prices that are observable, either directly or indirectly. These include quoted prices for similar assets or liabilities in active markets and quoted prices for identical or similar assets or liabilities in markets that are not active.
Level 3: Unobservable inputs in which little or no market data exists, therefore developed using estimates and assumptions developed by us, which reflect those that a market participant would use.
Cash and Cash Equivalents
Cash and cash equivalents include cash on hand and at banks, short-term deposits with an original maturity of three months or less with financial institutions, and bank overdrafts. Bank overdrafts are reflected as a current liability on the balance sheets.
Trade and Other Receivables
As amounts become uncollectible, they will be charged to an allowance and operations in the period when a determination of uncollectability is made. Any estimates of potentially uncollectible customer accounts receivable will be made based on an analysis of individual customer and historical write-off experience. The Companys analysis includes the age of the receivable account, creditworthiness of the customer and general economic conditions.
Property and Equipment
Property and equipment are stated at cost, net of accumulated depreciation and amortization. Expenditures for maintenance and repairs are expensed as incurred; additions, renewals, and improvements are capitalized. When property and equipment are retired or otherwise disposed of, the related cost and accumulated depreciation and amortization are removed from the respective accounts, and any gain or loss is included in operations. Depreciation and amortization of property and equipment is calculated using the straight-line basis over the following estimated useful lives:
| Leasehold improvements | shorter of the lease term or estimated useful lives | |
| Lab equipment | 3-7 years | |
| Furniture and fixtures | 3-7 years | |
| Computer hardware | 3-5 years | |
| Software | 3-4 years |
F-32
Table of Contents
Impairment of Long-Lived Assets
Property and equipment are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability of long-lived assets to be held and used is measured by a comparison of the carrying amount of an asset to the estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated undiscounted future cash flows, an impairment charge is recognized by the amount by which the carrying amount of the asset exceeds the fair value of the assets. Fair value is generally determined using the assets expected future discounted cash flows or market value, if readily determinable.
Trade and other payables
These amounts represent liabilities for goods and services provided to the Company prior to the end of the period and which are unpaid. Due to their short-term nature, they are measured at amortized cost and are not discounted. The amounts are unsecured and are usually paid within 30 days of recognition.
Australian Goods and Services Tax (GST) and other similar taxes
Revenues, expenses, and balance sheet items are recognized net of the amount of GST, except payable and receivable balances which are shown inclusive of GST. The GST incurred is payable on revenues to, and recoverable on purchases from, the Australian Taxation Office.
Cash flows are presented in the statements of cash flow on a gross basis, except for the GST component of investing and financing activities, which are disclosed as operating cash flows.
Commitments and contingencies are disclosed net of the amount of GST recoverable from, or payable to, the tax authority.
Income Taxes
The Company is governed by Australia and United States income tax laws, which are administered by the Australian Taxation Office and the United States Internal Revenue Service, respectively. The Company follows ASC 740 Accounting for Income Taxes, when accounting for income taxes, which requires an asset and liability approach to financial accounting and reporting for income taxes. Deferred income tax assets and liabilities are computed annually for temporary differences between the financial statements and tax bases of assets and liabilities that will result in taxable or deductible amounts in the future based on enacted tax laws and rates applicable to the periods in which the differences are expected to affect taxable income. Valuation allowances are established when necessary to reduce deferred tax assets to the amount more likely than not to be realized. Income tax expense is the tax payable or refundable for the period plus or minus the change during the period in deferred tax assets and liabilities. As of the date of these consolidated financial statements, the tax years for 2015 to 2019 remain open to examination by tax authorities.
For uncertain tax positions that meet a more likely than not threshold, the Company recognizes the benefit of uncertain tax positions in the consolidated financial statements. The Companys practice is to recognize interest and penalties, if any, related to uncertain tax positions in income tax expense in the consolidated statements of operations.
Share-Based Compensation
The Company records share-based compensation in accordance with ASC 718, Stock Compensation. ASC 718 requires the fair value of all share-based employee compensation awarded to employees to be recorded as an expense over the shorter of the service period or the vesting period. The Company values employee and non-employee share-based compensation at fair value using the Black-Scholes Option Pricing Model.
F-33
Table of Contents
The Company adopted FASB Accounting Standard Update (ASU) 2018-07 and accounts for non-employee share-based awards in accordance with the measurement and recognition criteria of ASC 718 and recognizes the fair value of such awards over the service period. The Company used the modified prospective method of adoption. There was no cumulative effect of adoption on July 1, 2019.
Basic and Diluted Net Loss Per Share
Basic net loss per share is calculated by dividing net loss by the weighted-average number of common shares outstanding during the period, Diluted net loss per share is calculated by dividing net loss by the weighted-average number of common shares outstanding plus potential common shares. Stock options, warrants and convertible instruments are considered potential common shares and are included in the calculation of diluted net loss per share using the treasury stock method when their effect is dilutive. Potential common shares are excluded from the calculation of diluted net loss per share when their effect is anti-dilutive. As of March 31, 2020 and 2019, there were 252,516 and 38,326 potential common shares, respectively, that were excluded from the calculation of diluted net loss per share because their effect was anti-dilutive.
Revenue Recognition
The Company adopted and implemented on July 1, 2018, ASC 606Revenue from Contracts with Customers (ASC 606). ASC 606 did not have a material impact on the consolidated financial statements.
Upon implementation of ASC 606, the Company recognizes revenue in accordance with that core principle by applying the following steps:
Step 1: Identify the contract(s) with a customer.
Step 2: Identify the performance obligations in the contract.
Step 3: Determine the transaction price.
Step 4: Allocate the transaction price to the performance obligations in the contract.
Step 5: Recognize revenue when (or as) the entity satisfies a performance obligation.
The Company applies judgement in determining whether contracts entered into fall within the scope of ASC 606. In doing so, management considers the commercial substance of the transaction and how risks and benefits of the contract accrue to the various parties to the contract. In determining the accounting treatment of the contract with Axovant, management assessed that the contract was within the scope of ASC 606.
Management has also made the judgement that the grant of the license and transfer of associated know-how and materials are accounted for as one performance obligation as they are not considered to be distinct; they are highly interrelated and could not provide benefits to the customer independently from each other. Judgements were made in relation to the transfer of the license and know-how and whether this should be recognized over time or a point in time. The point in time has been determined with regard to the point at which the transfer of know-how has substantially been completed and the customer has control of the asset and the ability to direct the use of and receive substantially all of the remaining benefits.
Licensing revenues
Revenue from licensees of the Companys intellectual property reflects the transfer of a right to use the intellectual property as it exists at the point in time in which the license is transferred to the customer. Consideration can be variable and is estimated using the most likely amount method. Subsequently, the estimate is constrained until it is highly probable that a significant revenue reversal will not occur when the uncertainty is resolved. Revenue is recognized as or when the performance obligations are satisfied.
The Company recognizes contract liabilities for consideration received in respect of unsatisfied performance obligations and reports these amounts as other liabilities in the statement of financial position. Similarly, if the
F-34
Table of Contents
Company satisfies a performance obligation before it receives the consideration, the Company recognizes either a contract asset or a receivable in its statement of financial position, depending on whether something other than the passage of time is required before the consideration is due.
Royalties
Revenue from licensees of the Companys intellectual property reflect a right to use the intellectual property as it exists at the point in time in which the license is granted. Where consideration is based on sales of product by the licensee, revenue is recognized when the customers subsequent sales of product occurs.
Services revenue
Revenue is earned (constrained by variable considerations) from the provision of research and development services to customers. Services revenue is recognized when performance obligations are either satisfied over time or at a point in time. Generally, the provision of research and development services under a contract with a customer will represent satisfaction of a performance obligation over time where the Company retains the right to payment for services performed but not yet completed.
Government Research and Development Grants
Government grants are recognized at fair value where there is reasonable assurance that the grant will be received and all grant conditions will be met. Grants relating to expense items are recognized as income over the periods necessary to match the grant costs they are compensating.
Grant income is generated through the Australian federal governments Research and Development Tax Incentive program, under which the government provides a cash refund for the 43.5% (2018: 43.5%) of eligible research and development expenditures. This grant is available for our research and development activities in Australia, as well as activities in the United States to the extent such U.S.-based expenses relate to our activities in Australia, do not exceed half the expenses for the relevant activities and are approved by the Australian government. Grants are recorded when a reliable estimate can be made.
Research and development expenses
Research and development costs are expensed when incurred.
Accounting Standards recently adopted
ASU 2016-02In February 2016, the FASB issued ASU No. 2016-02: Leases (Topic 842) whereby lessees will need to recognize most leases on their balance sheet as a right of use asset and a lease liability. This guidance is effective for interim and annual reporting periods beginning after December 15, 2018. The Company adopted this ASU effective July 1, 2019.
The determination of whether an arrangement is or contains a lease is based on the substance of the arrangement and requires an assessment of whether the fulfilment of the arrangement is dependent on the use of a specific asset or assets and the arrangement conveys a right to use the asset.
The Companys only lease is the lease on its research and development facilities. The Companys existing lease commitments are set out in Note 8.
ASU 2017-11In July 2017, the FASB issued ASU 2017-11 Earnings Per Share (Topic 260). The amendments in the update change the classification of certain equity-linked financial instruments (or embedded features) with down round features. The amendments also clarify existing disclosure requirements for equity-
F-35
Table of Contents
classified instruments. For freestanding equity-classified financial instruments, the amendments require entities that present earnings per share (EPS) in accordance with Topic 260, Earnings Per Share, to recognize the effect of the down round feature when it is triggered. That effect is treated as a dividend and as a reduction of income available to common shareholders in basic EPS. Convertible instruments with embedded conversion options that have down round features would be subject to the specialized guidance for contingent beneficial conversion features (in Subtopic 470-20, DebtDebt with Conversion and Other Options), including related EPS guidance (in Topic 260). For public business entities, the amendments in Part I of this update are effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2018. The Company has adopted this ASU effective July 1, 2019.
ASU 2018-07In June 2018, the Financial Accounting Standards Board (the FASB) issued Accounting Standards Update (ASU) 2018-07, CompensationStock Compensation (Topic 718). This update is intended to reduce cost and complexity and to improve financial reporting for share-based payments issued to non-employees (for example, service providers, external legal counsel, suppliers, etc.). The ASU expands the scope of Topic 718, CompensationStock Compensation, which currently only includes share-based payments issued to employees, to also include share-based payments issued to non-employees for goods and services. Consequently, the accounting for share-based payments to non-employees and employees will be substantially aligned. This standard will be effective for financial statements issued by public companies for the annual and interim periods beginning after December 15, 2018. Early adoption of the standard is permitted. The standard will be applied in a retrospective approach for each period presented. The Company has adopted this ASU, effective July 1, 2019.
New Accounting Standards and Interpretations not yet mandatory or early adopted
Certain ASUs that are not effective until after March 31, 2020 are not expected to have a significant effect on the Companys financial position or results of operations.
ASU 2016-13In June 2016, the FASB issued ASU No. 2016-13: Financial InstrumentsCredit Losses (Topic 326). This ASU represents a significant change in the accounting for credit losses model by requiring immediate recognition of managements estimates of current expected credit losses (CECL). Under the prior model, losses were recognized only as they were incurred, which FASB has noted delayed recognition of expected losses that might not yet have met the threshold of being probable. This guidance is effective for interim and annual reporting periods beginning after December 15, 2019. The Company has determined that it has met the criteria of a smaller reporting company (SRC) as of November 15, 2019. As such, ASU 2019-10: Financial Instruments-Credit Losses, Derivatives and Hedging, and Leases: Effective Datesamended the effective date for the Company to be for reporting periods beginning after December 15, 2022. The Company will adopt this ASU effective July 1, 2023.
NOTE 2GOING CONCERN (US$000)
The accompanying consolidated financial statements have been prepared in conformity with US GAAP, which contemplate continuation of the Company as a going concern. For the three months ended March 31, 2020, and 2019, the Company had a net loss of $2,102 and $1,704, and net cash used in operations of $1,700 and $1,605, respectively. For the nine months ended March 31, 2020, and 2019, the Company had a net (loss) income of ($5,339) and $4,078, and net cash (used in) provided by operations of ($4,390) and $6,605, respectively.
The Company has performed a review of the cash flow forecasts, considering the cash flow needs of the Company, and believes that funding will be sufficient for a period of at least twelve months from the date of this report.
The financial report does not contain any adjustments to the amounts or classifications of recorded assets or liabilities that might be necessary if the Company does not continue as a going concern.
F-36
Table of Contents
The financial statements take no account of the consequences, if any, of the effects of unsuccessful product development or commercialization, nor of the inability of the Company to obtain adequate funding in the future.
NOTE 3REVENUES
For the three months ended:
| Revenues from customers |
March 31, 2020 (US$000) |
March 31, 2019 (US$000) |
||||||
| Licensing revenue* |
| 76 | ||||||
| Royalty revenue* |
28 | 33 | ||||||
| Service revenue* |
| 340 | ||||||
|
|
|
|
|
|||||
| 28 | 449 | |||||||
|
|
|
|
|
|||||
For the nine months ended:
| Revenues from customers |
March 31, 2020 (US$000) |
March 31, 2019 (US$000) |
||||||
| Licensing revenue* |
60 | 9,909 | ||||||
| Royalty revenue* |
73 | 127 | ||||||
| Service revenue* |
4 | 919 | ||||||
|
|
|
|
|
|||||
| 137 | 10,955 | |||||||
|
|
|
|
|
|||||
| * | On July 9, 2018, the Company entered into an Agreement with Axovant. The Agreement granted Axovant an exclusive worldwide license to develop, manufacture, and commercialize products containing the Companys product known as BB-301, which was designed for the potential treatment of Oculopharyngeal Muscular Dystrophy. Service revenue consists of payments for services provided to Axovant during the license agreement signed in July 2018. On June 6, 2019, the termination of the Agreement with Axovant was announced. The termination of the Agreement is effective as of September 3, 2019. The termination discharges all future performance obligations at termination date under the contract. |
For the three months ended:
| Other revenues |
March 31, 2020 (US$000) |
March 31, 2019 (US$000) |
||||||
| Government research and development grants |
| 111 | ||||||
|
|
|
|
|
|||||
| | 111 | |||||||
|
|
|
|
|
|||||
For the nine months ended:
| Other revenues |
March 31, 2020 (US$000) |
March 31, 2019 (US$000) |
||||||
| Government research and development grants |
| 601 | ||||||
|
|
|
|
|
|||||
| | 601 | |||||||
|
|
|
|
|
|||||
F-37
Table of Contents
| Disaggregated revenue |
Three months ended March 31, 2020 (US$000) | |||||||||||||||
| Licensing | Royalties | Development activities |
Total | |||||||||||||
| Services transferred at a point in time |
| | | | ||||||||||||
| Services transferred over time |
| 28 | | 28 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| | 28 | | 28 | |||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Nine months ended March 31, 2020 (US$000) | ||||||||||||||||
| Licensing | Royalties | Development activities |
Total | |||||||||||||
| Services transferred at a point in time |
73 | | | 73 | ||||||||||||
| Services transferred over time |
| 60 | 4 | 64 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| 73 | 60 | 4 | 137 | |||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Three months ended March 31, 2019 (US$000) | ||||||||||||||||
| Licensing | Royalties | Development activities |
Total | |||||||||||||
| Services transferred at a point in time |
76 | 33 | | 109 | ||||||||||||
| Services transferred over time |
| | 340 | 340 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| 76 | 33 | 340 | 449 | |||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Nine months ended March 31, 2019 (US$000) | ||||||||||||||||
| Licensing | Royalties | Development activities |
Total | |||||||||||||
| Services transferred at a point in time |
9,909 | | | 9,909 | ||||||||||||
| Services transferred over time |
| 127 | 919 | 1,046 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| 9,909 | 127 | 919 | 10,955 | |||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
NOTE 4CASH AND CASH EQUIVALENTS
| March 31, 2020 (US$000) |
June 30, 2019 (US$000) |
|||||||
| Cash at Bank |
6,095 | 10,779 | ||||||
| Term Deposit |
5,305 | 4,939 | ||||||
|
|
|
|
|
|||||
| 11,400 | 15,718 | |||||||
|
|
|
|
|
|||||
NOTE 5OTHER ASSETS
| March 31, 2020 (US$000) |
June 30, 2019 (US$000) |
|||||||
| Prepaid expenses |
428 | 375 | ||||||
| Security deposit |
61 | 103 | ||||||
| Other deposit |
20 | 23 | ||||||
| Market value of listed shares |
1 | 1 | ||||||
|
|
|
|
|
|||||
| 510 | 502 | |||||||
|
|
|
|
|
|||||
F-38
Table of Contents
NOTE 6TRADE AND OTHER RECEIVABLES
| March 31, 2020 (US$000) |
June 30, 2019 (US$000) |
|||||||
| Research & development grants |
557 | 636 | ||||||
| Other receivables* |
261 | 1,900 | ||||||
|
|
|
|
|
|||||
| 818 | 2,536 | |||||||
|
|
|
|
|
|||||
| * | The Company had a contract asset of approximately $1.7M as of June 30, 2019, related to the contract with Axovant which is included in other receivables on the consolidated balance sheet. On June 6, 2019, the termination of the Agreement with Axovant was announced. The termination of the Agreement is effective as of September 3, 2019. |
NOTE 7PROPERTY AND EQUIPMENT, NET
| March 31, 2020 (US$000) |
June 30, 2019 (US$000) |
|||||||
| Leasehold improvements |
70 | 77 | ||||||
| Lab equipment |
1,096 | 1,014 | ||||||
| Furniture and fixtures |
| 6 | ||||||
| Computer hardware |
18 | 32 | ||||||
| Software |
16 | 11 | ||||||
|
|
|
|
|
|||||
| Total property and equipment, gross |
1,200 | 1,140 | ||||||
| Accumulated depreciation and amortization |
(798 | ) | (670 | ) | ||||
|
|
|
|
|
|||||
| Total property and equipment, net |
402 | 470 | ||||||
|
|
|
|
|
|||||
NOTE 8LEASES
On July 1, 2019, the Company adopted ASC 842, Leases (ASC 842). ASC 842 primarily requires lessees to recognize at the lease commencement date a lease liability, which is the lessees obligation to make lease payments arising from a lease, measured on a discounted basis, and a right-of-use asset, which is an asset that represents the lessees right to use, or control the use of, a specified asset for the lease term. Lessees and lessors must either (i) apply a modified retrospective transition approach for leases existing at, or entered into after, the beginning of the earliest comparative period presented in the financial statements or (ii) recognize a cumulative-effect adjustment to the opening balance of retained earnings in the period of adoption. Applying a full retrospective transition approach is not allowed. The Company elected to use the cumulative-effect transition method upon adoption.
ASC 842 also allows lessees and lessors to elect certain practical expedients. The Company elected the following practical expedients:
Transitional practical expedients:
| | The Company need not reassess whether any expired or existing contracts are or contain leases. |
| | The Company need not reassess the lease classification for any expired or existing leases (that is, all existing leases that were classified as operating leases in accordance with the previous guidance will be classified as operating leases, and all existing leases that were classified as capital leases in accordance with the previous guidance will be classified as finance leases). |
| | The Company need not reassess initial direct costs for any existing leases. |
F-39
Table of Contents
Hindsight practical expedient:
| | The Company elected the hindsight practical expedient in determining the lease term (that is, when considering lessee options to extend or terminate the lease and to purchase the underlying asset) and in assessing impairment of the Companys right-of-use assets. |
The Company has entered into an operating lease for office space under an agreement that expires in 2022. The lease requires the Company to pay utilities, insurance, taxes and other operating expenses. The Companys lease does not contain any residual value guarantees or material restrictive covenants.
Upon adoption of ASC 842, the Company recognized on its consolidated balance sheet as of July 1, 2019 an initial measurement of approximately $579k of operating lease liabilities, and approximately $579k of corresponding operating right-of use assets, net of tenant improvement allowances. There was also no cumulative effect adjustment to retained earnings as a result of the transition to ASC 842. The Company recorded the initial recognition of the operating lease as a supplemental noncash financing activity on the accompanying consolidated statement of cash flows. The adoption of ASC 842 did not have a material impact on the Companys consolidated statement of operations.
The tables below show the beginning balances of the operating lease right-of-use assets and liabilities as of June 30, 2019 and the ending balances as of March 31, 2020, including the changes during the periods.
| Operating lease right-of- use assets (US$000) |
||||
| Operating lease right-of-use asset at July 1, 2019 |
$ | 579 | ||
| Amortization right of use asset |
(138 | ) | ||
|
|
|
|||
| Operating lease right-of-use asset at March 31, 2020 |
$ | 441 | ||
|
|
|
|||
| Operating lease liabilities (US$000) |
||||
| Operating lease liabilities at July 1, 2019 |
$ | 579 | ||
| Principal payments on operating lease liabilities |
(129 | ) | ||
|
|
|
|||
| Operating lease liabilities at March 31, 2020 |
450 | |||
| Less: non-current portion |
263 | |||
|
|
|
|||
| Current portion at March 31, 2020 |
$ | 187 | ||
|
|
|
|||
As of March 31, 2020, the Companys operating lease has a remaining lease term of 2.21 years and a discount rate of 4.67%. The maturities of the operating lease liabilities are as follows:
| March 31, 2020 (US$000) |
||||
| 2020 |
50 | |||
| 2021 |
207 | |||
| 2022 |
218 | |||
| Total operating lease payments |
475 | |||
| Less imputed interest |
(25 | ) | ||
|
|
|
|||
| Present value of operating lease liabilities |
450 | |||
|
|
|
|||
F-40
Table of Contents
For the three and nine months ended March 31, 2020 and 2019, total lease expense under operating leases was approximately $52k, $156k and $126k, $155k, respectively, and was recorded in general and administrative expenses.
NOTE 9TRADE AND OTHER PAYABLES
| Trade and other payables |
March 31, 2020 (US$000) |
June 30, 2019 (US$000) |
||||||
| Trade payable |
288 | 1,474 | ||||||
| Accrued license fees |
43 | 385 | ||||||
| Accrued research and development fees |
| 498 | ||||||
| Accrued professional fees |
205 | 61 | ||||||
| Other payables |
77 | 76 | ||||||
|
|
|
|
|
|||||
| 613 | 2,494 | |||||||
|
|
|
|
|
|||||
NOTE 10STOCKHOLDERS EQUITY
Common Stock
On September 30, 2019, Benitec entered into a securities purchase agreement (SPA) with certain sophisticated and professional investors (Investors) in the United States to issue 186,666 shares of common stock at a purchase price of US$10.50 per share, in a registered direct offering.
Warrants
On September 30, 2019, the Investors were issued 4 Pre-Funded Warrants that are exercisable into 27,525 fully paid shares of common stock should the Pre-Funded Warrants be exercised in full (Pre-Funded Warrants). The exercise price for the Pre-Funded Warrants is US$10.50 per share issued on exercise of a Pre-Funded Warrant. The Pre-Funded Warrants may be exercised at any time from issue, in whole or in part, provided that the beneficial ownership of the relevant Investor in the total number of shares on issue may not exceed 9.99%. All of the Pre-Funded Warrants have been exercised.
On December 6, 2019, the Investors were issued 4 Purchase Warrants that are exercisable into 214,190 fully paid shares of common stock should the Purchase Warrants be exercised in full (Purchase Warrants). The exercise price for the Purchase Warrants is US$10.50 per share issued on exercise of a Purchase Warrant. The Purchase Warrants are exercisable, in whole or in part, any time from the date of issue until the fifth anniversary of the date of issue (December 6, 2024).
The activity related to warrants during the three months ended March 31, 2020, is summarized as follows:
| Common Stock from Warrants |
Weighted-average Exercise Price (per share) |
|||||||
| Outstanding at December 31, 2019 |
252,516 | $ | 21.43 | |||||
| Granted |
| |||||||
| Exercised |
| |||||||
| Forfeited |
| |||||||
|
|
|
|||||||
| Outstanding at March 31, 2020 |
252,516 | $ | 21.43 | |||||
|
|
|
|||||||
| Exercisable at March 31, 2020 |
252,516 | $ | 21.43 | |||||
|
|
|
|||||||
F-41
Table of Contents
The activity related to warrants during the nine months ended March 31, 2020, is summarized as follows:
| Common Stock from Warrants |
Weighted-average Exercise Price (per share) |
|||||||
| Outstanding at June 30, 2019 |
38,326 | $ | 82.50 | |||||
| Granted |
241,715 | $ | 10.50 | |||||
| Exercised |
(27,525 | ) | $ | 10.50 | ||||
| Forfeited |
| |||||||
|
|
|
|||||||
| Outstanding at March 31, 2020 |
252,516 | $ | 21.43 | |||||
|
|
|
|||||||
| Exercisable at March 31, 2020 |
252,516 | $ | 21.43 | |||||
|
|
|
|||||||
Equity Incentive Plan
Employee Share Option Plan
The Company is authorized to grant options under the Benitec Biopharma Limited Employee Share Option Plan (the Plan).
Equity Awards
The activity related to equity awards, which comprised of stock options during the three months ended March 31, 2020, is summarized as follows:
| Stock Options |
Weighted-average Exercise Price |
Weighted-average Remaining Contractual Term |
Aggregate Intrinsic Value |
|||||||||||||
| Outstanding at December 31, 2019 |
72,384 | $ | 63.87 | |||||||||||||
| Granted |
| |||||||||||||||
| Exercised |
| |||||||||||||||
| Forfeited |
| |||||||||||||||
|
|
|
|||||||||||||||
| Outstanding at March 31, 2020 |
72,384 | $ | 63.87 | 3.08 years | | |||||||||||
|
|
|
|||||||||||||||
| Exercisable at March 31, 2020 |
30,300 | $ | 85.08 | 2.42 years | | |||||||||||
|
|
|
|||||||||||||||
The activity related to equity awards, which comprised of stock options during the nine months ended March 31, 2020 is summarized as follows:
| Stock Options |
Weighted-average Exercise Price |
Weighted-average Remaining Contractual Term |
Aggregate Intrinsic Value |
|||||||||||||
| Outstanding at June 30, 2019 |
86,467 | $ | 64.20 | |||||||||||||
| Granted |
| |||||||||||||||
| Exercised |
| |||||||||||||||
| Forfeited |
(14,083 | ) | $ | 91.62 | ||||||||||||
|
|
|
|||||||||||||||
| Outstanding at March 31, 2020 |
72,384 | $ | 63.87 | 3.08 years | | |||||||||||
|
|
|
|||||||||||||||
| Exercisable at March 31, 2020 |
30,300 | $ | 85.08 | 2.42 years | | |||||||||||
|
|
|
|||||||||||||||
F-42
Table of Contents
Share-Based Compensation Expense
The classification of share-based compensation expense for the three months ended:
| March 31, 2020 (US$000) |
March 31, 2019 (US$000) |
|||||||
| Research and development |
18 | 12 | ||||||
| General and administrative |
70 | 270 | ||||||
|
|
|
|
|
|||||
| Total share-based compensation expense |
88 | 282 | ||||||
|
|
|
|
|
|||||
The classification of share-based compensation expense for the nine months ended:
| March 31, 2020 (US$000) |
March 31, 2019 (US$000) |
|||||||
| Research and development |
53 | 42 | ||||||
| General and administrative |
127 | 525 | ||||||
|
|
|
|
|
|||||
| Total share-based compensation expense |
180 | 567 | ||||||
|
|
|
|
|
|||||
As of March 31, 2020, there was $284k of unrecognized share-based compensation expense related to shares of common stock issued under the Plan.
NOTE 11INCOME TAXES
For the three and nine months ended March 31, 2020 and 2019, the Company did not recognize a provision for income taxes due to having recorded a full valuation allowance against its deferred tax assets. As of March 31, 2020 and June 30, 2019, the Company established a full valuation allowance against its deferred tax assets due to the uncertainty surrounding the realization of such assets. As of March 31, 2020 and June 30, 2019, the Company had no unrecognized tax benefits. The Company does not anticipate there will be a significant change in unrecognized tax benefits within the next 12 months.
NOTE 12COMMITMENTS AND CONTINGENCIES
On December 18, 2012, the Company announced the appointment of Synteract, Inc. as its Clinical Research Organization responsible for the progression of TT-034 into Phase I/IIa clinical trials in the U.S. The Company has negotiated a contract for Synteract to continue to manage the Phase I/IIa clinical trial and the long-term patient follow-up through 2016 and beyond. The Company announced on February 20, 2016 that it was terminating the HCV program, and at the end of the 2018 financial year had assumed all patients would remain in the study and the follow-up would continue to 2021 at a maximum cost of $462k. However, in July 2018, Benitec applied to the FDA for, and the FDA approved, the discontinuation of the study which will result in minimal costs being incurred in the future.
There are no contingent liabilities as of March 31, 2020.
NOTE 13SUBSEQUENT EVENTS
The Company is closely monitoring the impact of the pandemic of the novel strain of coronavirus COVID-19 (COVID-19) on all aspects of its business and geographies, including how it will impact its team members, suppliers, vendors and business partners. While the Company did experience some disruption from COVID-19 through the delay of the initiation of the pre-clinical trials through the date of issuance, it is unable to predict the impact that COVID-19 will have on its financial position and operating results due to uncertainties.
F-43
Table of Contents
Shares of Common Stock
Pre-funded Warrants to Purchase Shares of Common Stock
Shares of Common Stock underlying the Pre-funded Warrants

PRELIMINARY PROSPECTUS
H.C. Wainwright & Co.
, 2020
Table of Contents
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
| Item 13. | Other Expenses of Issuance and Distribution |
The following table sets forth the costs and expenses, other than the underwriting discounts and commissions, payable by the registrant in connection with the sale of common stock and pre-funded warrants being registered. All amounts are estimates except for the SEC registration fee and the Financial Industry Regulatory Authority, or FINRA, filing fee.
| Item |
Amount to be paid |
|||
| SEC registration fee |
$ | 2,596 | ||
| FINRA filing fee |
* | |||
| Printing and engraving expenses |
* | |||
| Legal fees and expenses |
* | |||
| Accounting fees and expenses |
* | |||
| Transfer agent fees and expenses |
* | |||
| Miscellaneous fees and expenses |
* | |||
|
|
|
|||
| Total |
$ | * | ||
|
|
|
|||
| * | To be provided by amendment. |
| Item 14. | Indemnification of Directors and Officers |
We are incorporated under the laws of the State of Delaware. Section 102 of the Delaware General Corporation Law permits a corporation to eliminate the personal liability of directors of a corporation to the corporation or its stockholders for monetary damages for a breach of fiduciary duty as a director, except where the director breached his or her duty of loyalty, failed to act in good faith, engaged in intentional misconduct or knowingly violated a law, authorized the payment of a dividend or approved a stock repurchase in violation of Delaware corporate law or obtained an improper personal benefit.
Section 145 of the Delaware General Corporation Law provides that a corporation has the power to indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action or suit by or in the right of the corporation to procure a judgment in its favor by reason of the fact that the person is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise against expenses (including attorneys fees) actually and reasonably incurred by him in connection with the defense or settlement of such action or suit if the person acted in good faith and in a manner the person reasonably believed to be in or not opposed to the best interests of the corporation and except that no indemnification shall be made with respect to any claim, issue or matter as to which such person shall have been adjudged to be liable to the corporation unless and only to the extent that the Court of Chancery or the court in which such action or suit was brought shall determine upon application that, despite the adjudication of liability but in view of all the circumstances of the case, such person is fairly and reasonably entitled to indemnity for such expenses which the Court of Chancery or such other court shall deem proper.
As permitted by the Delaware General Corporation Law, our amended and restated certificate of incorporation includes a provision to eliminate the personal liability of our directors for monetary damages for breach of their fiduciary duties as directors, subject to certain exceptions. In addition, our amended and restated certificate of incorporation and amended and restated bylaws provide that we are required to indemnify our officers and directors under certain circumstances, including those circumstances in which indemnification would otherwise be discretionary, and we are required to advance expenses to our officers and directors as incurred in connection with proceedings against them for which they may be indemnified.
II-1
Table of Contents
We have entered into indemnification agreements with our directors and executive officers. These indemnification agreements are not intended to deny or otherwise limit third-party or derivative suits against us or our directors or officers, but to the extent a director or officer were entitled to indemnity or contribution under our bylaws, the financial burden of a third-party suit would be borne by us, and we would not benefit from derivative recoveries against the director or officer. Such recoveries would accrue to our benefit but would be offset by our obligations to the director or officer under the indemnification agreement.
The underwriting agreement provides that the underwriter is obligated, under certain circumstances, to indemnify our directors, officers and controlling persons against certain liabilities, including liabilities under the Securities Act. Reference is made to the form of underwriting agreement to be filed as Exhibit 1.1 hereto.
We maintain directors and officers liability insurance for the benefit of our directors and officers.
| Item 15. | Recent Sales of Unregistered Securities |
Over the past three years, the Company and Benitec Limited have issued and sold to third parties the securities listed below without registering the securities under the Securities Act of 1933, as amended (the Securities Act). None of these transactions involved any public offering. All the securities listed below were sold through private placement either (i) outside the United States or (ii) in the United States to a limited number of investors in transactions not involving any public offering. As discussed below, we believe that each issuance of these securities was exempt from, or not subject to, registration under the Securities Act.
On March 13, 2017, Benitec Limited issued 29,305,819 ordinary shares (which were converted to 97,686 shares of common stock as part of the Re-domiciliation) to Nant Capital, LLC. The shares were priced at A$0.1859 per share. This issuance was exempt from registration under the Securities Act in reliance on Section 4(a)(2).
On June 4, 2018, Benitec Limited issued 36,442,672 ordinary shares (which were converted to 121,475 shares of common stock as part of the Re-domiciliation) to existing shareholders. The shares were priced at A$0.17 per share. This issuance was exempt from registration under the Securities Act in reliance on Regulation S.
On September 30, 2019, Benitec entered into a securities purchase agreement (SPA) with certain sophisticated and professional investors in the United States to issue 2,800,000 American Depositary Shares (ADSs), with each ADS representing 20 fully paid ordinary shares (which were converted to 186,666 shares of common stock as part of the Re-domiciliation), at a purchase price of US$0.70 per ADS, in a registered direct offering. The Investors were also issued warrants to purchase up to 412,863 ADSs (representing 27,524 shares of common stock after the Re-domiciliation) in aggregate, at a purchase price per warrant equal to US$0.6999 per ADS to be issued on exercise of the warrant (Pre-Funded Warrants). The Pre-Funded Warrants were exercisable at any time from issue, in whole or in part, at an exercise price of US$0.0001 per ADS issued on exercise (subject to certain adjustments), provided that the beneficial ownership of the relevant Investor in the total number of ADSs on issue not exceed 9.99%. The Pre-Funded Warrants have all been exercised. This issuance was exempt from registration under the Securities Act in reliance on Section 4(a)(2).
On April 15, 2020, the Company completed the Re-domiciliation. In connection with the Re-domiciliation, Benitec issued 1,070,957 shares of common stock, on the basis of one share of common stock for every 300 ordinary shares of Benitec Limited issued and outstanding prior to the Re-domiciliation. The Re-domiciliation was effected pursuant to a statutory scheme of arrangement under Australian law (the Scheme). The issuance of Benitecs shares of common stock in the Scheme was exempt from registration under the Securities Act in reliance on Section 3(a)(10).
On April 22, 2020, Benitec issued 37,417 shares of common stock in connection with a cashless exercise of warrants exercisable for 107,095 shares of common stock. The issuance was exempt from registration under the Securities Act in reliance on Section 3(a)(9).
II-2
Table of Contents
| Item 16. | Exhibits and financial statement schedules |
| (a) | Exhibits |
The exhibits to the registration statement are listed in the Exhibit Index to this registration statement and are incorporated herein by reference.
| Item 17. | Undertakings |
The undersigned Registrant hereby undertakes:
(1) To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
(i) To include any prospectus required by section 10(a)(3) of the Securities Act of 1933;
(ii) To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Securities and Exchange Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20% change in the maximum aggregate offering price set forth in the Calculation of Registration Fee table in the effective registration statement; and
(iii) To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement.
(2) That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
(3) To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
(4) That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser:
(A) Each prospectus filed by the registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the date the filed prospectus was deemed part of and included in the registration statement; and
(B) Each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use.
(5) That for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of securities, the undersigned registrant undertakes that in a primary offering
II-3
Table of Contents
of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser:
(i) Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424;
(ii) Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant;
(iii) The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and
(iv) Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
(6) Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to any charter provision, by law or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act of 1933 and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
(7) The undersigned registrant hereby undertakes that:
(i) For purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(I) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective; and
(ii) For the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
II-4
Table of Contents
EXHIBIT INDEX
II-5
Table of Contents
| Exhibit |
Exhibit |
|
| 23.1 | Consent of Grant Thornton Audit Pty Ltd | |
| 23.2* | Consent of Proskauer Rose LLP (included in Exhibit 5.1) | |
| 24.1 | Power of Attorney (included on signature page hereto) | |
| * | To be filed by amendment. |
| | Indicates a management contract or compensatory plan. |
II-6
Table of Contents
Pursuant to the requirements of the Securities Act of 1933, as amended, the Registrant has duly caused this Registration Statement on Form S-1 to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Hayward, State of California, on August 14, 2020.
| BENITEC BIOPHARMA INC. | ||
| By: | /s/ Dr. Jerel Banks |
|
| Dr. Jerel Banks Chief Executive Officer |
||
We, the undersigned officers and directors of Benitec Biopharma Inc., hereby severally constitute and appoint Dr. Jerel Banks and Megan Boston, and each of them singly (with full power to each of them to act alone), our true and lawful attorneys-in-fact and agents, with full power of substitution and resubstitution in each of them for him and in his name, place and stead, and in any and all capacities, to sign any and all amendments (including post-effective amendments) to this registration statement (or any other registration statement for the same offering that is to be effective upon filing pursuant to Rule 462(b) under the Securities Act of 1933), and to file the same, with all exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite or necessary to be done in and about the premises, as full to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents or any of them, or their or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act of 1933, as amended, this Registration Statement on Form S-1 has been signed by the following persons in the capacities and on the dates indicated.
| Signature |
Title |
Date |
||
| /s/ Dr. Jerel Banks Dr. Jerel Banks |
Chief Executive Officer, Director (principal executive officer) |
August 14, 2020 | ||
| /s/ Megan Boston Megan Boston |
Executive Director, Director (principal accounting and financial officer) |
August 14, 2020 | ||
| /s/ J. Kevin Buchi J. Kevin Buchi |
Director | August 14, 2020 | ||
| /s/ Peter Francis Peter Francis |
Director | August 14, 2020 | ||
| /s/ Edward Smith Edward Smith |
Director | August 14, 2020 | ||
II-7